Abstract
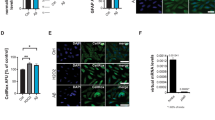
Temporal lobe epilepsy (TLE), the most common form of epilepsy is often resistant to pharmacological treatment. Neuronal loss observed in epileptic brain may be result of an overproduction of free radicals (oxidative stress). Oxidative stress is characterized by an imbalance between antioxidant defenses and oxidizing agents (free radicals), which can lead to tissue injury. The n-3 PUFAs are important for the development and maintenance of central nervous system functions. Research by our group has shown that chronic treatment with fish oil, immediately after status epilepticus (SE), exhibits both neuroprotective effects and effects on neuroplasticity. The main purpose of this research was to evaluate if fish oil exhibits a protective effect against oxidative stress. Animals were subjected to TLE model by pilocarpine administration. After 3 h of SE they were randomly divided into the following groups: control animals treated daily with vehicle or with 85 mg/kg of fish oil and animals with epilepsy treated daily with vehicle or with 85 mg/kg of fish oil. After 90 days, superoxide anion production, enzymatic activity of superoxide dismutase (SOD) and catalase (CAT) and protein expression of NAD(P)H oxidase subunits (p47PHOX and gp91PHOX) were analyzed. Our results showed evidences that reactive oxygen species are increased in animals with epilepsy and that fish oil supplementation could counteract it. Fish oil supplementation promoted protection against oxidative stress by multiple ways, which involved the reduction of activity and expression of NAD(P)H oxidase subunits and increased the activity and expression of antioxidants enzymes, contributing to well-known neuroprotective effect in epilepsy.



Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. doi:10.1016/S0076-6879(84)05016-3
Babior BM (2004) NADPH oxidase. Curr Opin Immunol 16:42–47. doi:10.1016/j.coi.2003.12.001
Bellissimo MI, Amado D, Abdalla DS, Ferreira EC, Cavalheiro EA, Naffah-Mazzacoratti MG (2001) Superoxide dismutase, glutathione peroxidase activities and the hydroperoxide concentration are modified in the hippocampus of epileptic rats. Epilepsy Res 46:121–128. doi:10.1016/S0920-1211(01)00269-8
Bondy SC, Lee DK (1993) Oxidative stress induced by glutamate receptors agonists. Brain Res 610:229–233. doi:10.1016/0006-8993(93)91405-H
Bonfoco E, Krainic D, Ankarcrona M, Nocotera P, Lipton AS (1995) Apoptosis and necrosis: two distinct events induced, respectively, by mild and intense insults with N- methyl-d-aspartate or nitric oxide/superoxide in cortical cell cultures. Proc Natl Acad Sci 92:7162–7166
Bradford MM (1976) A rapid and sensitive method for quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1016/0003-2697(76)90527-3
Cavalheiro EA (1995) The pilocarpine model of epilepsy. Ital J Neurol Sci 16:33–37
Curia G, Longo D, Biagini G, Jones RS, Avoli M (2008) The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods 172(2):143–157. doi:10.1016/j.jneumeth.2008.04.019
Ferrari D, Cysneiros RM, Scorza CA, Arida RM, Cavalheiro EA, de Almeida ACG, Scorza FA (2008) Neuroprotective activity of omega-3 fatty acids against epilepsy-induced hippocampal damage: quantification with immunohistochoemical for calcium-binding proteins. Epilepsy Behav 13:36–42. doi:10.1016/j.yebeh.2008.01.001
Fisher M, Levine PH, Weiner BH (1990) Dietary n-3 fatty acid supplementation reduces superoxide production and chemiluminescence in a monocyte-enriched preparation of leukocytes. Am J Clin Nutr 51:804–808
Freitas RM, Vasconcelos SM, Souza FC, Viana GS, Fonteles MM (2005) Oxidative stress in the hippocampus after pilocarpine-induced status epilepticus in Wistar rats. FEBS J 272:1307–1312. doi:10.1111/j.1742-4658.2004.04537.x
Fujikawa DG (2005) Prolonged seizures and cellular injury: understanding the connection. Epilepsy Behav 7(3):S3–S11. doi:10.1016/j.yebeh.2005.08.003
Hossain MS, Hashimoto M, Gamoh S, Masumura S (1999) Antioxidative effects of docosahexaenoic acid in the cerebrum versus cerebellum and brainstem of aged hypercholesterolemic rats. J Neurochem 72(3):1133–1138. doi:10.1046/j.1471-4159.1999.0721133.x
Inni SM (2007) Dietary (n-3) fatty acids and brain development. J Nutr 137(4):855–859. doi:10.1016/j.brainres.2008.08.078
Liu SH, Chang CD, Chen PH, Su JR, Chen CC, Chaung HC (2012) Docosahexaenoic acid and phosphatidylserine supplementations improve antioxidant activities and cognitive functions of the developing brain on pentylenetetrazol-induced seizure model. Brain Res 1451:19–26. doi:10.1016/j.brainres.2012.02.060
McCann JC, Ames BN (2005) Is docosahexaenoic acid, an n-3 long-chain polyunsaturated fatty acid, required for development of normal brain function? An overview of evidence from cognitive and behavioral tests in humans and animals. Am J Clin Nutr 82(2):281–295
Nevander G, Ingvar M, Auer R, Siesjö BK (1985) Status epilepticus in well-oxygenated rats causes neuronal necrosis. Ann Neurol 18(3):281–290. doi:10.1002/ana.410180303
Patel M, Li QY, Chang LY, Crapo J, Liang LP (2005) Activation of NADPH oxidase and extracellular superoxide production in seizure-induced hippocampal damage. J Neurochem 92:123–131. doi:10.1111/j.1471-4159.2004.02838.x
Paxinos JP, Watson C (1998) The rat brain in stereotaxic coordinates. Academic, San Diego
Pestana RF, Kinjo ER, Hernandes MS, Britto LRG (2010) Reactive oxygen species generated by NAD(P)H oxidase are involved in neurodegeneration in the pilocarpine model of temporal lobe epilepsy. Neurosci Lett 484:187–191. doi:10.1016/j.neulet.2010.08.049
Rumià J, Marmol F, Sanchez J, Giménez-Crouseilles J, Carreño M, Bargalló N, Boget T, Pintor L, Setoain X, Donaire A, Saez GT, Ribalta T, Ferrer E, Puig-Parellada P (2013) Oxidative stress markers submitted to epilepsy surgery. Epilepsy Res 107(1–2):75–81. doi:10.1016/j.eplepsyres.2013.08.020
Schlanger S, Shinitzky M, Yam D (2002) Diet enriched with omega–3 fatty acids alleviates convulsion symptoms in epilepsy patients. Epilepsia 43:103–104. doi:10.1046/j.1528-1157.2002.13601.x
Shulz JN, Henshaw DR, Siwek D, Jenkins BG, Ferrante RJ, Cipolloni PB, Kowall NW, Rosen BR, Beal MF (1995) Involvement of free radicals in excitotoxicity in vivo. J Neurochem 64:2239–2247. doi:10.1046/j.1471-4159.1995.64052239.x
Takahashi M, Tsuboyama-Kasaoka N, Nakatani T (2002) Fish oil feeding alters liver gene expressions to defend against PPAR alpha activation and ROS production. Am J Physiol Gastrointest Liver Physiol 282:G338–G348. doi:10.1152/ajpgi.00376.2001
Turski WA, Cavalheiro EA, Bortolotto ZA, Mello LM, Schwarz M, Turski L (1984) Seizures produced by pilocarpine in mice: a behavioral, electroencephalographic and morphological analysis. Brain Res 321(2):237–253
Valko M, Rhodes CJ, Izakovic MM (2006) Free radicals metals and antioxidants on oxidative stress-induced cancer. Chem Biol Interact 160:1–40. doi:10.1016/j.cbi.2005.12.009
Vincent AM, Maiese K (1999) Nitric oxide induction of neuronal endonuclease activity in programmed cell death. Exp Cell Res 246:290–300. doi:10.1006/excr.1998.4282
Vincent P, Mulle C (2009) Kainate receptors in epilepsy and excitotoxicity. Neuroscience 158:309–323. doi:10.1016/j.neuroscience.2008.02.066
Wu A, Ying Z, Gomez-Pinilla F (2004) Dietary omega-3 fatty acids normalize BDNF levels, reduce oxidative damage, and counteract learning disability after traumatic brain injury in rats. J Neurotrauma 21(10):1457–1467. doi:10.1089/neu.2004.21.1457
Acknowledgments
This work was supported financially by FAPESP, CInAPCe-FAPESP, FAPESP/CNPq/MCT-Instituto Nacional de Neurociência Translacional and CNPq.
Conflict of interest
The authors declare that there is no conflict of interest with any financial organization.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nejm, M.B., Haidar, A.A., Marques, M.J.G. et al. Fish oil provides protection against the oxidative stress in pilocarpine model of epilepsy. Metab Brain Dis 30, 903–909 (2015). https://doi.org/10.1007/s11011-015-9666-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-015-9666-0