Abstract
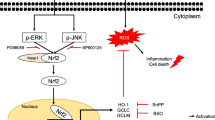
Chemical modification of chitosan is a promising method for the improvement of biological activity. In this study, chitosan-caffeic acid (CCA) was prepared and its in vitro hepatoprotective ability against hydrogen peroxide-induced hepatic damage in liver cells was evaluated. Treatment with CCA (50–400 µg/mL) did not show cytotoxicity and also significantly (p < 0.05) recovered cell viability against 650 µM hydrogen peroxide-induced hepatotoxicity. CCA treatment attenuated reactive oxygen species generation and lipid peroxidation in addition to increasing cellular glutathione level in cultured hepatocytes. To validate the underlying mechanism, antioxidant and phase II detoxifying enzyme expressions, which are mediated by NF-E2-related factor 2 (Nrf2) activation, were analyzed and CCA treatment was found to increase the expression of superoxide dismutase-1 (SOD-1), glutathione reductase (GR), heme oxygenase-1 (HO-1), and NAD(P)H:quinine oxidoreductase 1 (NQO1). CCA treatment resulted in increased Nrf2 nuclear translocation. The phosphorylation of extracellular regulated kinase (ERK), c-Jun NH2-terminal kinase (JNK), and p38 mitogen-activated protein kinase (MAPK) by CCA treatment contributed to Nrf2 activation. Pharmacological blockade of ERK, JNK, and p38 MAPK revealed that SP600125 (JNK inhibitor) and PD98059 (ERK inhibitor) treatment reduced Nrf2 translocation into the nucleus while SB203580 (p38 inhibitor) exhibited weak inhibition. Collectively, CCA protects liver cells against hydrogen peroxide-induced injury and this ability is attributed to the induction of antioxidants and phase II detoxifying enzymes that are mediated by Nrf2 translocation via JNK/ERK signaling.






Similar content being viewed by others
Abbreviations
- CCA:
-
Chitosan-caffeic acid
- SOD-1:
-
Superoxide dismutase-1
- GR:
-
Glutathione reductase
- HO-1:
-
Heme oxygenase-1
- NQO1:
-
NAD(P)H:quinine oxidoreductase 1
- Nrf2:
-
NF-E2-related factor 2
- GSH:
-
Glutathione
- MAPK:
-
Mitogen-activated protein kinase
References
Yang CM, Huang SM, Liu CL et al (2012) Apo-8′-lycopenal induces expression of HO-1 and NQO-1 via the ERK/p38-Nrf2-ARE pathway in human HepG2 cells. J Agric Food Chem 60(6):1576–1585
Farombi EO, Tahnteng JG, Agboola AO et al (2000) Chemoprevention of 2-acetylaminofluorene-induced hepatotoxicity and lipid peroxidation in rats by kolaviron-a Garcinia kola seed extract. Food Chem Toxicol 38(6):535–541
Fang YZ, Yang S, Wu G (2002) Free radicals, antioxidants, and nutrition. Nutrition 18(10):872–879
Muriel P (2009) Role of free radicals in liver diseases. Hepatol Int 3(4):526–536
Surh YJ (2003) Cancer chemoprevention with dietary phytochemicals. Nat Rev Cancer 3(10):768–780
Kim J, Cha YN, Surh YJ (2010) A protective role of nuclear factor-erythroid 2-related factor-2 (Nrf2) in inflammatory disorders. Mut Res 690(1–2):12–23
Hwang YP, Choi JH, Han EH et al (2008) Protective mechanisms of Aralia continentalis extract against tert-butyl hydroperoxide-induced hepatotoxicity: in vivo and in vitro studies. Food Chem Toxicol 46(11):3512–3521
Subramaniam SR, Ellis EM (2011) Esculetin-induced protection of human hepatoma HepG2 cells against hydrogen peroxide is associated with the Nrf2-dependent induction of the NAD(P)H: quinone oxidoreductase 1 gene. Toxicol Appl Pharmacol 250(2):130–136
Senthil Kumar KJ, Liao JW, Xiao JH et al (2012) Hepatoprotective effect of lucidone against alcohol-induced oxidative stress in human hepatic HepG2 cells through the up-regulation of HO-1/Nrf-2 antioxidant genes. Toxicol In Vitro 26(5):700–708
Truong VL, Bak MJ, Jun M et al (2014) Antioxidant defense and hepatoprotection by procyanidins from almond (Prunus amygdalus) skins. J Agric Food Chem 62(34):8668–8678
Yu R, Chen C, Mo YY, Hebbar V, Owuor ED, Tan TH, Kong AN (2000) Activation of mitogen-activated protein kinase pathways induces antioxidant response element-mediated gene expression via a Nrf2-dependent mechanism. J Biol Chem 275:39907–39913
Plotnikov A, Zehorai E, Procaccia S, Seger R (2011) The MAPK cascades: signaling components, nuclear roles and mechanisms of nuclear translocation. Biochim Biophys Acta 1813:1619–1633
Chou TC, Fu E, Shen EC (2003) Chitosan inhibits prostaglandin E2 formation and cyclooxygenase-2 induction in lipopolysaccharide-treated RAW 264.7 macrophages. Biochem Biophys Res Commun 308(2):403–407
Lee DS, Je JY (2013) Gallic acid-grafted-chitosan inhibits foodborne pathogens by a membrane damage mechanism. J Agric Food Chem 61(26):6574–6579
Muzzarelli RAA, Muzzarelli C (2005) Chitosan chemistry: relevance to the biomedical sciences. Adv Polym Sci 186:151–209
Park PJ, Je JY, Kim SK (2004) Free radical scavenging activities of differently deacetylated chitosans using an ESR spectrometer. Carbohydr Polym 55:17–22
Jeon YJ, Kim SK (2001) Potential immuno-stimulating effect of antitumoral fraction of chitosan oligosaccharides. J Chitin Chitosan 6:163–167
Senevirathne M, Ahn CB, Je JY (2001) Hepatoprotective effect of chitooligosaccharides against tert-butylhydroperoxide-induced damage in Chang liver cells. Carbohydr Polym 83:995–1000
Senevirathne M, Jeon YJ, Kim YT et al (2012) Prevention of oxidative stress in Chang liver cells by gallic acid-grafted-chitosans. Carbohydr Polym 87:876–880
Santhosh S, Sini TK, Anandan R et al (2007) Hepatoprotective activity of chitosan against isoniazid and rifampicin-induced toxicity in experimental rats. Eur J Pharmacol 572(1):69–73
Song RJ, Kim JW, Kang KY (2010) Hepatoprotective effect of chitosan oligosaccharides on CCl4-induced liver injury in rats. J Chitin Chitosan 15:80–87
Lee DS, Woo JY, Ahn CB et al (2014) Chitosan-hydroxycinnamic acid conjugates: preparation, antioxidant and antimicrobial activity. Food Chem 148:97–104
Lee DS, Cho YS, Je JY (2013) Antioxidant and antibacterial activities of chitosan-phloroglucinol conjugates. Fish Aquat Sci 16:229–235
Poot M, Verkerk A, Koster JF et al (1986) De novo synthesis of glutathione in human fibroblasts during in vitro ageing and in some metabolic diseases as measured by a flow cytometric method. Biochim Biophys Acta 883(3):580–584
Lee DS, Li B, Kim KS et al (2013) Butein protects human dental pulp cells from hydrogen peroxide-induced oxidative toxicity via Nrf2 pathway-dependent heme oxygenase-1 expressions. Toxicol In Vitro 27(2):874–881
Kamata H, Hirata H (1999) Redox regulation of cellular signalling. Cell Signal 11(1):1–14
Finkel T, Holbrook NJ (2000) Oxidants, oxidative stress and the biology of ageing. Nature 408(6809):239–247
Ji LL, Sheng YC, Zheng ZY et al (2015) The involvement of p62-Keap1-Nrf2 antioxidative signaling pathway and JNK in the protection of natural flavonoid quercetin against hepatotoxicity. Free Radic Biol Med 85:12–23
Chen RC, Sun GB, Wang J et al (2015) Naringin protects against anoxia/reoxygenation-induced apoptosis in H9c2 cells via the Nrf2 signaling pathway. Food Funct 6(4):1331–1344
Nakagawa H, Maeda S (2012) Molecular mechanisms of liver injury and hepatocarcinogenesis: focusing on the role of stress-activated MAPK. Pathol Res Int 2012:172894
Acknowledgments
This work was supported by the Human Resource Training Program for Regional Innovation and Creativity through the Ministry of Education and National Research Foundation of Korea (NRF-2014H1C1A1066586).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that there is no conflict of interests.
Rights and permissions
About this article
Cite this article
Ahn, CB., Je, JY., Kim, YS. et al. Induction of Nrf2-mediated phase II detoxifying/antioxidant enzymes in vitro by chitosan-caffeic acid against hydrogen peroxide-induced hepatotoxicity through JNK/ERK pathway. Mol Cell Biochem 424, 79–86 (2017). https://doi.org/10.1007/s11010-016-2845-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2845-4