Abstract
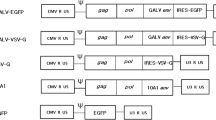
Malignant glioma can be treated with radioiodine following transfection with human sodium iodide symporter (hNIS) gene. Ad-Tp-E1A-Gp-NIS is engineered with human telomerase reverse transcriptase (hTERT) and glial fibrillary acidic protein (GFAP) promoters to express early region 1A (E1A) and hNIS genes, which may be useful in targeted gene therapy. The Ad-Tp-E1A-Gp-NIS was constructed and purified using the E1A and hNIS genes regulated by the hTERT and GFAP promoters, respectively. Glioma cells were infected by Ad-Tp-E1A-Gp-NIS. Selective replication ability of Ad-Tp-E1A-Gp-NIS was then evaluated by plaque forming assay, transgene expression by Western blot, 125I-iodide uptake and efflux, clonogenicity following 131I-iodide treatment in the tumor cells, and radioiodine therapy using nude mouse model. The Ad-Tp-E1A-Gp-NIS could selectively replicate; the hNIS gene was successfully expressed under the GFAP promoter. Western blot analyses using E1A- and hNIS-specific antibodies revealed two bands of approximately 40 and 70 kDa. In addition, the cells showed about 93.4 and 107.1 times higher 125I uptake in U251 and U87 cells than in the control cells, respectively. Clonogenic assay indicated that >90 % of cells transfected with Ad-Tp-E1A-Gp-NIS were killed. The Ad-Tp-E1A-Gp-NIS-transfected and 2 mCi 131I-injected U87 xenograft nude mice survived the longest among the three groups. Ad-Tp-E1A-Gp-NIS has a good ability of selective replication and strong antitumor selectivity. An effective therapy of 131I was achieved activity in malignant glioma cells after induction of tumor-specific iodide uptake activity by GFAP promoter-directed hNIS gene expression in vitro and in vivo.





Similar content being viewed by others
References
Rowe WP, Huebner RJ, Gilmore LK, Parrott RH, Ward TG (1953) Isolation of a cytopathogenic agent from human adenoids undergoing spontaneous degeneration in tissue culture. Proc Soc Exp Biol Med 84:570–573
Gallimore PH, Turnell AS (2001) Adenovirus E1A: remodelling the host cell, a life or death experience. Oncogene 20:7824–7835. doi:10.1038/sj.onc.1204913
Kirn D (2000) Replication-selective oncolytic adenoviruses: virotherapy aimed at genetic targets in cancer. Oncogene 19:6660–6669. doi:10.1038/sj.onc.1204094
Chen L, Altmann A, Mier W, Eskerski H, Leotta K, Guo L, Zhu R, Haberkorn U (2006) Radioiodine therapy of hepatoma using targeted transfer of the human sodium/iodide symporter gene. J Nucl Med 47:854–862
Brenner M, Kisseberth WC, Su Y, Besnard F, Messing A (1994) GFAP promoter directs astrocyte-specific expression in transgenic mice. J Neurosci 14:1030–1037
Nolte C, Matyash M, Pivneva T, Schipke CG, Ohlemeyer C, Hanisch UK, Kirchhoff F, Kettenmann H (2001) GFAP promoter-controlled EGFP-expressing transgenic mice: a tool to visualize astrocytes and astrogliosis in living brain tissue. Glia 33:72–86
Besnard F, Brenner M, Nakatani Y, Chao R, Purohit HJ, Freese E (1991) Multiple interacting sites regulate astrocyte-specific transcription of the human gene for glial fibrillary acidic protein. J Biol Chem 266:18877–18883
Cho JY, Shen DH, Yang W, Williams B, Buckwalter TL, La Perle KM, Hinkle G, Pozderac R, Kloos R, Nagaraja HN, Barth RF, Jhiang SM (2002) In vivo imaging and radioiodine therapy following sodium iodide symporter gene transfer in animal model of intracerebral gliomas. Gene Ther 9:1139–1145. doi:10.1038/sj.gt.3301787
Giralt A, Friedman HC, Caneda-Ferron B, Urban N, Moreno E, Rubio N, Blanco J, Peterson A, Canals JM, Alberch J (2010) BDNF regulation under GFAP promoter provides engineered astrocytes as a new approach for long-term protection in Huntington’s disease. Gene Ther 17:1294–1308. doi:10.1038/gt.2010.71
Huang M, Batra R, Kogai T, Lin Y (2001) Ectopic expression of the thyroperoxidase gene augments radioiodide uptake and retention mediated by the sodium iodide symporter in non-small cell lung cancer. Cancer Gene Ther 8:612–618
Hede SM, Hansson I, Afink GB, Eriksson A, Nazarenko I, Andrae J, Genove G, Westermark B, Nister M (2009) GFAP promoter driven transgenic expression of PDGFB in the mouse brain leads to glioblastoma in a Trp53 null background. Glia 57:1143–1153. doi:10.1002/glia.20837
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL, Coviello GM, Wright WE, Weinrich SL, Shay JW (1994) Specific association of human telomerase activity with immortal cells and cancer. Science 266:2011–2015
Gu J, Kagawa S, Takakura M, Kyo S (2000) Tumor-specific transgene expression from the human telomerase reverse transcriptase promoter enables targeting of the therapeutic effects of the Bax gene to cancers. Cancer Res 60:5359–5364
Komata TKY, Kanzawa T et al (2001) Treatment of malignant glioma cells with the transfer of constitutively active caspase-6 using the human telomerase catalytic subunit (human telomerase reverse transcriptase) gene promoter. Cancer Res 61(15):5796–5802
Koga S, Hirohata S, Kondo Y, Komata T, Takakura M, Inoue M (2001) FADD gene therapy using the human telomerase catalytic subunit (hTERT) gene promoter to restrict induction of apoptosis to tumors in vitro and in vivo. Anticancer Res 21:1937–1943
Lin T, Gu J, Zhang L, Huang X, Stephens L, Curley S, Fang B (2002) Targeted expression of green fluorescent protein/tumor necrosis factor-related apoptosis-inducing ligand fusion protein from human telomerase reverse transcriptase promoter elicits antitumor activity without toxic effects on primary human hepatocytes. Cancer Res 62:3620–3625
de Leeuw B, Su M, ter Horst M, Iwata S, Rodijk M, Hoeben RC, Messing A, Smitt PS, Brenner M (2006) Increased glia-specific transgene expression with glial fibrillary acidic protein promoters containing multiple enhancer elements. J Neurosci Res 83:744–753. doi:10.1002/jnr.20776
Ng P, Cummings DT, Evelegh CM, Graham FL (2000) Yeast recombinase FLP functions effectively in human cells for construction of adenovirus vectors. Biotechniques 29(524–526):528
Doloff JC, Waxman DJ, Jounaidi Y (2008) Human telomerase reverse transcriptase promoter-driven oncolytic adenovirus with E1B-19 kDa and E1B-55 kDa gene deletions. Hum Gene Ther 19:1383–1400. doi:10.1089/hum.2008.056
Yu X, Zhan X, D’Costa J, Tanavde VM, Ye Z, Peng T, Malehorn MT, Yang X, Civin CI, Cheng L (2003) Lentiviral vectors with two independent internal promoters transfer high-level expression of multiple transgenes to human hematopoietic stem-progenitor cells. Mol Ther 7:827–838
Cao X, Yang M, Wei RC, Zeng Y, Gu JF, Huang WD, Yang DQ, Li HL, Ding M, Wei N, Zhang KJ, Xu B, Liu XR, Qian QJ, Liu XY (2011) Cancer targeting Gene-Viro-Therapy of liver carcinoma by dual-regulated oncolytic adenovirus armed with TRAIL gene. Gene Ther 18:765–777. doi:10.1038/gt.2011.16
Li Y, Idamakanti N, Arroyo T, Thorne S, Reid T, Nichols S, VanRoey M, Colbern G, Nguyen N, Tam O, Working P, Yu DC (2005) Dual promoter-controlled oncolytic adenovirus CG5757 has strong tumor selectivity and significant antitumor efficacy in preclinical models. Clin Cancer Res 11:8845–8855. doi:10.1158/1078-0432.CCR-05-1757
Jounaidi Y, Hecht JE, Waxman DJ (1998) Retroviral transfer of human cytochrome P450 genes for oxazaphosphorine-based cancer gene therapy. Cancer Res 58:4391–4401
Spitzweg C, Zhang S, Bergert ER, Castro MR, McIver B, Heufelder AE, Tindall DJ, Young CY, Morris JC (1999) Prostate-specific antigen (PSA) promoter-driven androgen-inducible expression of sodium iodide symporter in prostate cancer cell lines. Cancer Res 59:2136–2141
Boland A, Ricard M, Opolon P, Bidart J, Yeh P, Filetti S (2000) Adenovirus-mediated transfer of the thyroid sodium/iodide symporter gene into tumors for a targeted radiotherapy. Cancer Res 60:3484–3492
Schipper M, Weber A, Béhé M, Göke R, Joba W, Schmidt H, Bert T, Simon B, Arnold R, Heufelder A, Behr T (2003) Radioiodide treatment after sodium iodide symporter gene transfer is a highly effective therapy in neuroendocrine tumor cells. Cancer Res 63:1333–1338
Ma XJ, Huang R, Kuang AR (2009) AFP promoter enhancer increased specific expression of the human sodium iodide symporter (hNIS) for targeted radioiodine therapy of hepatocellular carcinoma. Cancer Invest 27:673–681. doi:10.1080/07357900802620885
Spitzweg C, Baker CH, Bergert ER, O’Connor MK, Morris JC (2007) Image-guided radioiodide therapy of medullary thyroid cancer after carcinoembryonic antigen promoter-targeted sodium iodide symporter gene expression. Hum Gene Ther 18:916–924. doi:10.1089/hum.2007.081
Kim HJ, Jeon YH, Kang JH, Lee YJ, Kim KI, Chung HK, Jeong JM, Lee DS, Lee MC, Chung JK (2007) In vivo long-term imaging and radioiodine therapy by sodium-iodide symporter gene expression using a lentiviral system containing ubiquitin C promoter. Cancer Biol Ther 6:1130–1135
Bauerschmitz GJ, Ranki T, Kangasniemi L, Ribacka C, Eriksson M, Porten M, Herrmann I, Ristimaki A, Virkkunen P, Tarkkanen M, Hakkarainen T, Kanerva A, Rein D, Pesonen S, Hemminki A (2008) Tissue-specific promoters active in CD44+ CD24−/low breast cancer cells. Cancer Res 68:5533–5539. doi:10.1158/0008-5472.CAN-07-5288
Li W, Tan J, Wang P, Li N, Zhang F (2013) The glial fibrillary acidic protein promoter directs sodium/iodide symporter gene expression for radioiodine therapy of malignant glioma. Oncol Lett 5:669–674. doi:10.3892/ol.2012.1055
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (to Jian TAN) (No. 81171372) (to Wei LI) (No. 81301244) and the Tianjin Research Program of Application Foundation and Advanced Technology (to Jian TAN) (No. 12JCZDJC26000) and National Key Clinical Specialty Project of China.
Disclaimer
This paper is our own work. We have no specific disclaimer or conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, W., Tan, J., Wang, P. et al. Glial fibrillary acidic protein promoters direct adenovirus early 1A gene and human telomerase reverse transcriptase promoters direct sodium iodide symporter expression for malignant glioma radioiodine therapy. Mol Cell Biochem 399, 279–289 (2015). https://doi.org/10.1007/s11010-014-2254-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2254-5