Abstract
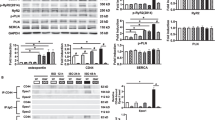
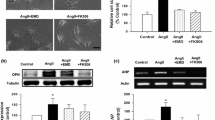
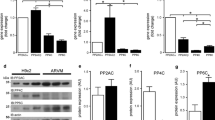
Cluster of differentiation 73 (CD73) is an ecto-5′ nucleotidase which catalyzes the conversion of AMP to adenosine. One of the many functions of adenosine is to suppress the activity of tissue nonspecific alkaline phosphatase (TNAP), an enzyme important in regulating intracellular calcification. Since myocardial calcification is associated with various cardiac disease states, we studied the individual roles and crosstalk between CD73 and TNAP in regulating myocyte responses to the α1 adrenoceptor agonist phenylephrine in terms of calcification and hypertrophy. Cultured neonatal rat cardiomyocytes were treated with 10 µM phenylephrine for 24 h in the absence or presence of the stable adenosine analog 2-chloro-adenosine, the TNAP inhibitor tetramisole or the CD73 inhibitor α,β-methylene ADP. Phenylephrine produced marked hypertrophy as evidenced by significant increases in myocyte surface area and ANP gene expression, as well as calcification determined by Alizarin Red S staining. These responses were associated with reduced CD73 gene and protein expression and CD73 activity. Conversely, TNAP expression and activity were significantly increased although both were suppressed by 2-chloro-adenosine. CD73 inhibition alone significantly reduced myocyte-derived adenosine levels by >50 %, and directly induced hypertrophy and calcification in the absence of phenylephrine. These responses and those to phenylephrine were abrogated by TNAP inhibition. We conclude that TNAP contributes to the hypertrophic effect of phenylephrine, as well as its ability to produce cardiomyocyte calcification. These responses are minimized by CD73-dependent endogenously produced adenosine.






Similar content being viewed by others
References
Laubach VE, French BA, Okusa MD (2011) Targeting of adenosine receptors in ischemia-reperfusion injury. Expert Opin Ther Targets 15:103–118
Liao Y, Takashima S, Asano Y, Asakura M, Ogai A, Shintani Y, Minamino T, Asanuma H, Sanada S, Kim J, Ogita H, Tomoike H, Hori M, Kitakaze M (2007) Activation of adenosine A1 receptor attenuates cardiac hypertrophy and prevents heart failure in murine left ventricular pressure-overload model. Circ Res 93:759–7663
Gan XT, Rajapurohitam V, Haist JV, Chidiac P, Cook MA, Karmazyn M (2005) Inhibition of phenylephrine-induced cardiomyocyte hypertrophy by activation of multiple adenosine receptor subtypes. J Pharmacol Exp Ther 312:27–34
Karmazyn M, Gan XT (2013) Advances in biochemistry in health and disease. In: Ostadal B, Dhalla NS (eds) Cardiac adaptations. Springer, New York, pp 403–414
Schrader WP, West CA (1990) Localization of adenosine deaminase and adenosine deaminase complexing protein in rabbit heart. Implications for adenosine metabolism. Circ Res 66:754–762
Gray RP, Owen KM, Giacomini KM (2004) The concentrative nucleoside transporter family, SLC28. Pflugers Arch 447:728–734
Naito Y, Lowenstein JM (1981) 5’-Nucleotidase from rat heart. Biochemistry 20:5188–5194
Colgan SP, Eltzschig HK, Eckle T, Thompson LF (2006) Physiological roles for ecto-5’-nucleotidase (CD73). Purinergic Signal 2:351–360
St Hilaire C, Ziegler SG, Markello TC, Brusco A, Groden C, Gill F, Carlson-Donohoe H, Lederman RJ, Chen MY, Yang D, Siegenthaler MP, Arduino C, Mancini C, Freudenthal B, Stanescu HC, Zdebik AA, Chaganti RK, Nussbaum RL, Kleta R, Gahl WA, Boehm M (2011) NT5E mutations and arterial calcifications. N Engl J Med 364:432–442
Hessle L, Johnson KA, Anderson HC, Narisawa S, Sali A, Goding JW, Terkeltaub R, Millan JL (2002) Tissue-nonspecific alkaline phosphatase and plasma cell membrane glycoprotein-1 are central antagonistic regulators of bone mineralization. Proc Natl Acad Sci USA 99:9445–9449
Millán JL, Fishman WH (1995) Biology of human alkaline phosphatases with special reference to cancer. Crit Rev Clin Lab Sci 32:1–39
Mota A, Silva P, Neves D, Lemos C, Calhau C, Torres D, Martel F, Fraga H, Ribeiro L, Alçada MN, Pinho MJ, Negrão MR, Pedrosa R, Guerreiro S, Guimarães JT, Azevedo I, Martins MJ (2008) Characterization of rat heart alkaline phosphatase isoenzymes and modulation of activity. Braz J Med Biol Res 41:600–609
Weissen-Plenz G, Nitschke Y, Rutsch F (2008) Mechanisms of arterial calcification: spotlight on the inhibitors. Adv Clin Chem 46:263–293
Schoppet M, Shanahan CM (2008) Role for alkaline phosphatase as an inducer of vascular calcification in renal failure? Kidney Int 73:989–991
van Kruijsdijk RC, van der Heijden JJ, Uijlings R, Otterspoor LC (2011) Sepsis-related myocardial calcification. Circ Heart Fail 4:e16–e18
Kaimoto S, Kawasaki T, Yamano M, Miki S, Kamitani T, Sugihara H (2012) Intramyocardial calcification in a patient with apical hypertrophic cardiomyopathy. Intern Med 51:1523–1526
Lee HU, Youn HJ, Shim BJ, Lee SJ, Park MY, Jeong JU, Gu GM, Jeon HK, Lee JE, Kwon BJ (2012) Porcelain heart: rapid progression of cardiac calcification in a patient with hemodialysis. J Cardiovasc Ultrasound 20:193–196
Joshi PV, Lele VR, Shah HJ, Rao S (2012) Massive calcification in the myocardium post infarction. Heart Views 13:74
Proudfoot D, Skepper JN, Hegyi L, Bennett MR, Shanahan CM, Weissberg PL (2000) Apoptosis regulates human vascular calcification in vitro: evidence for initiation of vascular calcification by apoptotic bodies. Circ Res 87:1055–1062
Pang T, Gan XT, Freeman DJ, Cook MA, Karmazyn M (2010) Compensatory upregulation of the adenosine system following phenylephrine-induced hypertrophy in cultured rat ventricular myocytes. Am J Physiol Heart Circ Physiol 298:H545–H553
Li L, Chang L, Pellet-Rostaing S, Liger F, Lemaire M, Buchet R, Wu Y (2009) Synthesis and evaluation of benzo[b]thiophene derivatives as inhibitors of alkaline phosphatases. Bioorg Med Chem 17:7290–7300
Darvish A, Pomerantz RW, Zografides PG, Metting PJ (1996) Contribution of cytosolic and membrane-bound 5’-nucleotidases to cardiac adenosine production. Am J Physiol 271:H2162–H2167
Fujita M, Asakura M, Sanada S, Funaya H, Tsukamoto O, Komamura K, Asanuma H, Taketani S, Isomura T, Nakamaru K, Furukawa H, Sawa Y, Hori M, Kitakaze M (2008) Activation of ecto-5’-nucleotidase in the blood and hearts of patients with chronic heart failure. J Card Fail 14:426–430
Xu X, Fassett J, Hu X, Zhu G, Lu Z, Li Y, Schnermann J, Bache RJ, Chen Y (2008) Ecto-5’-nucleotidase deficiency exacerbates pressure-overload-induced left ventricular hypertrophy and dysfunction. Hypertension 51:1557–1564
Robin E, Sabourin J, Marcillac F, Raddatz E (2013) Involvement of CD73, equilibrative nucleoside transporters and inosine in rhythm and conduction disturbances mediated by adenosine A1 and A2A receptors in the developing heart. J Mol Cell Cardiol 63:14–25
Millán JL (2006) Alkaline Phosphatases: Structure, substrate specificity and functional relatedness to other members of a large superfamily of enzymes. Purinergic Signal 2:335–341
Markello TC, Pak LK, St Hilaire C, Dorward H, Ziegler SG, Chen MY, Chaganti K, Nussbaum RL, Boehm M, Gahl WA (2011) Vascular pathology of medial arterial calcifications in NT5E deficiency: implications for the role of adenosine in pseudoxanthoma elasticum. Mol Genet Metab 103:44–50
Tonelli M, Curhan G, Pfeffer M, Sacks F, Thadhani R, Melamed ML (2009) Relation between alkaline phosphatase, serum phosphate, and all-cause or cardiovascular mortality. Circulation 120:1784–1792
Dahl R, Sergienko EA, Su Y, Mostofi YS, Yang L, Simao AM, Narisawa S, Brown B, Mangravita-Novo A, Vicchiarelli M, Smith LH, O’Neill WC, Millán JL, Cosford ND (2009) Discovery and validation of a series of aryl sulfonamides as selective inhibitors of tissue-nonspecific alkaline phosphatase (TNAP). J Med Chem 52:6919–6925
Heineke J, Ritter O (2012) Cardiomyocyte calcineurin signaling in subcellular domains: from the sarcolemma to the nucleus and beyond. J Mol Cell Cardiol 52:62–73
Kim YH, Yoon DS, Kim HO, Lee JW (2012) Characterization of different subpopulations from bone marrow-derived mesenchymal stromal cells by alkaline phosphatase expression. Stem Cells Dev 21:2958–2968
Elsherif L, Huang MS, Shai SY, Yang Y, Li RY, Chun J, Mekany MA, Chu AL, Kaufman SJ, Ross RS (2008) Combined deficiency of dystrophin and beta1 integrin in the cardiac myocyte causes myocardial dysfunction, fibrosis and calcification. Circ Res 102:1109–1117
Acknowledgments
This work was supported by a grant from the Heart and Stroke Foundation of Ontario (NA 6028). M Karmazyn holds a Tier 1 Canada Research Chair in Experimental Cardiology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gan, X.T., Taniai, S., Zhao, G. et al. CD73-TNAP crosstalk regulates the hypertrophic response and cardiomyocyte calcification due to α1 adrenoceptor activation. Mol Cell Biochem 394, 237–246 (2014). https://doi.org/10.1007/s11010-014-2100-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-014-2100-9