Abstract
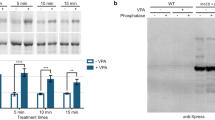
The therapeutic effects of lithium in bipolar disorder are poorly understood. Lithium decreases free inositol levels by inhibiting inositol monophosphatase 1 and myo-inositol 3-phosphate synthase (IPS). In this study, we demonstrate for the first time that IPS can be phosphorylated. This was evident when purified rat IPS was dephosphorylated by lambda protein phosphatase and analyzed by phospho-specific ProQ-Diamond staining and Western blot analysis. These techniques demonstrated a mobility shift consistent with IPS being phosphorylated. Mass spectral analysis revealed that Serine-524 (S524), which resides in the hinge region derived from exon 11 of the gene, is the site for phosphorylation. Further, an antibody generated against a synthetic peptide of IPS containing monophosphorylated-S524, was able to discriminate the phosphorylated and non-phosphorylated forms of IPS. The phosphoprotein is found in the brain and testis, but not in the intestine. The intestinal IPS isoform lacks the peptide bearing S524, and hence, cannot be phosphorylated. Evidences suggest that IPS is monophosphorylated at S524 and that the removal of this phosphate does not alter its enzymatic activity. These observations suggest a novel function for IPS in brain and other tissues. Future studies should resolve the functional role of phospho-IPS in brain inositol signaling.





Similar content being viewed by others
References
Berridge MJ, Irvine RF (1989) Inositol phosphates and cell signaling. Nature 341:197–205
Majerus PW, Kisseleva MV, Norris FA (1999) The role of phosphatases in inositol signaling reactions. J Biol Chem 274:10669–10672
Parthasarathy R, Eisenberg F Jr (1986) The inositol phospholipids: a stereochemical view of biological activity. Biochem J 235:313–322
Fisher SK, Novak JE, Agranoff BW (2002) Inositol and higher inositol phosphates in neural tissues: homeostasis, metabolism and functional significance. J Neurochem 82:736–754
Chakraborty A, Kim S, Snyder SH (2011) Inositol pyrophosphates as mammalian cell signals. Sci Signal 4:re1
Thurston JH, Sherman WR, Hauhart RE, Kloepper RF (1989) Myo-inositol: a newly identified non-nitrogenous osmoregulatory molecule in mammalian brain. Pediatr Res 26:482–485
Eisenberg F Jr, Parthasarathy R (1987) Measurement of biosynthesis of myo-inositol from glucose 6-phosphate. Methods Enzymol 141:127–143
Luo HR, Huang YE, Chen JC, Saiardi A, Iijima M, Ye K, Huang Y, Nagata E, Devreotes P, Snyder SH (2003) Inositol pyrophosphates mediate chemotaxis in Dictyostelium via pleckstrin homology domain-PtdIns(3,4,5)P3 interactions. Cell 114:559–572
Saiardi A, Resnick AC, Snowman AM, Wendland B, Snyder SH (2005) Inositol pyrophosphates regulate cell death and telomere length through phosphoinositide 3-kinase-related protein kinases. Proc Natl Acad Sci U S A 102:1911–1914
Monserrate JP, York JD (2010) Inositol phosphate synthesis and the nuclear processes they affect. Curr Opin Cell Biol 22:365–373
Lee DW, Kim SY, Lee T, Nam YK, Ju A, Woo DC, You SJ, Han JS, Lee SH, Choi CB, Kim SS, Shin HC, Kim HY, Kim DJ, Rhim HS, Choe BY (2012) Ex vivo detection for chronic ethanol consumption-induced neurochemical changes in rats. Brain Res 1429:134–144
Minati L, Aquino D, Bruzzone MG, Erbetta A (2010) Quantitation of normal metabolite concentrations in six brain regions by in vivo H-MR spectroscopy. J Med Phys 35:154–163
Parthasarathy L, Vadnal RE, Parthasarathy R, Devi CS (1994) Biochemical and molecular properties of lithium-sensitive myo-inositol monophosphatase. Life Sci 54:1127–1142
Levine J, Barak Y, Gonzalves M, Szor H, Elizur A, Kofman O, Belmaker RH (1995) Double-blind, controlled trial of inositol treatment of depression. Am J Psychiatry 152:792–794
Vadnal RE, Parthasarathy L, Parthasarathy R (1997) Role of inositol in psychiatric disorders: basic and clinical aspects. CNS Drugs 7:6–16
Vadnal RE, Parthasarathy L, Parthasarathy R (1998) Promising psychotherapeutic effects of the natural sugar: myo-inositol. Nutr Neurosci 1:21–33
Agam G, Shamir A, Shaltiel G, Greenberg ML (2002) Myo-inositol-1-phosphate (MIP) synthase: a possible new target for antibipolar drugs. Bipolar Disord 4(Suppl s1):15–20
Seelan RS, Lakshmanan J, Casanova MF, Parthasarathy RN (2009) Identification of myo-inositol-3-phosphate synthase isoforms: characterization, expression, and putative role of a 16-kDa γc isoform. J Biol Chem 284:9443–9457
Zhuo S, Clemens JC, Hakes DJ, Barford D, Dixon JE (1993) Expression, purification, crystallization and biochemical characterization of a recombinant protein phosphatase. J Biol Chem 268:17754–17761
Lahm HW, Langen H (2000) Mass spectrometry: a tool for the identification of proteins separated by gels. Electrophoresis 21:2105–2114
Imanishi SY, Kochin V, Ferraris SE, de Thonel A, Pallari HM, Corthals GL, Eriksson JE (2007) Reference-facilitated phosphoproteomics: fast and reliable phosphopeptide validation by micro LC-ESI-Q-TOF MS/MS. Mol Cell Proteomics 6:1380–1391
Acknowledgments
This study was supported by the Office of Research and Development, Medical Research Service, Department of Veterans Affairs, Washington, DC, USA to RNP. The contents of this article do not represent the views of the US Department of Veterans Affairs or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Parthasarathy, R.N., Lakshmanan, J., Thangavel, M. et al. Rat brain myo-inositol 3-phosphate synthase is a phosphoprotein. Mol Cell Biochem 378, 83–89 (2013). https://doi.org/10.1007/s11010-013-1597-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1597-7