Abstract
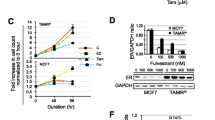
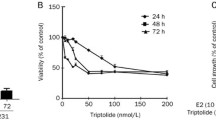
Antiestrogen is one type of the endocrine therapeutic agents for estrogen receptor α (ERα)-positive breast cancer. Unfortunately, this treatment alone is insufficient. Here we reported a novel potential anticancer strategy by using histone deacetylase (HDAC) inhibitor to enhance the action of endocrine therapy in ERα-positive breast cancer cell. The well-described HDAC inhibitor, trichostatin A (TSA), and antiestrogen raloxifene were found to, respectively, inhibit E2-induced proliferation of MCF-7 breast cancer cell in a dose-responsive and time-dependent manner. TSA and raloxifene enhanced the antiproliferative activity of each other by promoting cell death via apoptosis and cell cycle arrest. Thus, they displayed better antiproliferative effects in combined treatment than that with either agent alone. The expression level of estrogen receptor β (ERβ) showed a marked increase after TSA or/and raloxifene treatment. Treatments with TSA or/and raloxifene resulting in the up-regulation of ERβ are in accordance with the antiproliferative effects of the two agents. Furthermore, the over-expression of ERβ by adenovirus delivery could inhibit the proliferation of MCF-7 tumor cells and drastically enhanced the antiproliferative effects of TSA and raloxifene. These results demonstrated that the interference of ERβ on the antiproliferative effects of HDAC inhibitor and antiestrogen constitutes a promising approach for breast cancer treatment.






Similar content being viewed by others
References
Early Breast Cancer Trialists’ Collaborative Group (1992) Systemic treatment of early breast cancer by hormonal, cytotoxic, or immune therapy. 133 randomised trials involving 31,000 recurrences and 24,000 deaths among 75,000 women. Lancet 339:71–85
Early Breast Cancer Trialists’ Collaborative Group (1998) Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet 351:1451–1467
Hodges-Gallagher L, Valentine CD, Bader SE, Kushner PJ (2007) Inhibition of histone deacetylase enhances the antiproliferative action on antiestrogens on breast cancer cells and blocks tamoxifen-induced proliferation of uterine cells. Breast Cancer Res Treat 105:297–309
Clarke R, Leonessa F, Welch JN, Skaar TC (2001) Cellular and molecular pharmacology of antiestrogen action and resistance. Pharmacol Rev 53:25–71
Ring A, Dowsett M (2004) Mechanisms of tamoxifen resistance. Endocr Relat Cancer 11:643–658
Chen SJ, Li H, Durand J, Oparil S, Chen YF (1996) Estrogen reduces myointimal proliferation after balloon injury of rat carotid artery. Circulation 93:577–584
Jacobsson J, Cheng L, Lyke K, Kuwahara M, Kagan E, Ramwell PW, Foegh ML (1992) Effect of estradiol on accelerated atherosclerosis in rabbit heterotopic aortic allografts. J Heart Lung Transplant 11:1188–1193
Adams MR, Kaplan JR, Koritnik DR, Clarkson TB (1987) Pregnancy-associated inhibition of coronary artery atherosclerosis in monkeys. Evidence of a relationship with endogenous estrogen. Arteriosclerosis 7:378–383
Adams MR, Kaplan JR, Manuck SB, Koritnik DR, Parks JS, Wolfe MS, Clarkson TB (1990) Inhibition of coronary artery atherosclerosis by 17-beta estradiol in ovariectomized monkeys. Lack of an effect of added progesterone. Arteriosclerosis 10:1051–1057
Barrett-Connor E (1997) Sex differences in coronary heart disease: why are women so superior? The 1995 Ancel Keys Lecture. Circulation 95:252–264
Stampfer MJ, Colditz GA, Willett WC, Manson JE, Rosner B, Speizer FE, Hennekens CH (1991) Postmenopausal estrogen therapy and cardiovascular disease: ten-year followup from the Nurses’ Health Study. N Engl J Med 325:756–762
Delmas PD, Bjarnason NH, Mitlak BH, Ravoux AC, Shah AS, Huster WJ, Draper M, Christiansen C (1997) Effects of raloxifene on bone mineral density, serum cholesterol concentrations, and uterine endometrium in postmenopausal women. N Engl J Med 337:1641–1647
Ettinger B, Black DM, Mitlak BH, Knickerbocker RK, Nickelsen T, Genant HK, Christiansen C, Delmas PD, Zanchetta JR, Stakkestad J (1999) Reduction of vertebral fracture risk in postmenopausal women with osteoporosis treated with raloxifene: results from a 3-year randomized clinical trial. Multiple outcomes of raloxifene evaluation (MORE) investigators. JAMA 282:637–645
Jordan VC, Robinson SP (1987) Species specific pharmacology of antiestrogens role of metabolism. Fed Proc 46:1870–1874
Mayo Clinic Women’s HealthSource (2006) Raloxifene works as well as tamoxifen in preventing invasive breast cancer. Mayo Clin Women’s HealthSource 10:3
Viale G, Regan MM, Maiorano E, Mastropasqua MG, Dell`Orto P, Rasmussen BB, Raffoul J, Neven P, Orosz Z, Braye S (2007) Prognostic and predictive value of centrally reviewed expression of estrogen and progesterone receptors in a randomized trial comparing letrozole and tamoxifen adjuvant therapy for postmenopausal early breast cancer: BIG 1–98. J Clin Oncol 25:3846–3852
Levy N, Tatomer D, Herber CB, Zhao X, Tang H, Sargeant T, Ball LJ, Summers J, Speed TP, Leitman DC (2008) Differential regulation of native estrogen receptor-regulatory elements by estrodiol, tamoxifen, and raloxifene. Mol Endocrinol 22:287–303
Joung KE, Kim DK, Sheen YY (2004) Antiproliferative effect of trichostatin A and HC-toxin in T47D human breast cancer cells. Arch Pharm Res 27:640–645
Thomas S, Munster PN (2009) Histone deacetylase inhibitor induced modulation of antiestrogen therapy. Cancer Lett 280:184–191
Hirokawa Y, Arnold M, Nakajima H, Zalcberg J, Maruta H (2005) Signal therapy of breast cancers by the HDAC inhibitor FK228 that blocks the activation of PAK1 and abrogates the tamoxifen-resistance. Cancer Biol Ther 4:956–960
Munster PN, Troso-Sandoval T, Rosen N, Rifkind R, Marks PA, Richon VM (2001) The histone deacetylase inhibitor suberoylanilide hydroxamic acid induces differentiation of human breast cancer cells. Cancer Res 61:8492–8497
Hartman J, Ström A, Gustafsson J-Å (2009) Estrogen receptor beta in breast cancer—diagnostic and therapeutic implications. Steroids 74:635
Bardin A, Boulle N, Lazennec G, Vignonl F, Pujoll P (2004) Loss of ERbeta expression as a common step in estrogen-dependent tumor progression. Endocr Relat Cancer 11:537–551
Margueron R, Duong V, Castet A, Cavailles V (2004) Histone deacetylase inhibition and estrogen signalling in human breast cancer cells. Biochem Pharmacol 68:1239–1246
McKenna NJ, O’Malley BW (2002) Minireview: nuclear receptor coactivators—an update. Endocrinology 143:2461–2465
Marks PA, Richon VM, Rifkind RA (2000) Histone deacetylase inhibitors: inducers of differentiation or apoptosis of transformed cells. J Natl Cancer Inst 92:1210–1216
Lin CY, Strom A, Li Kong S, Kietz S, Thomsen JS, Tee JB, Vega VB, Miller LD, Smeds J, Bergh J, Gustafsson JA, Liu ET (2007) Inhibitory effects of estrogen receptor β on specific hormone-responsive gene expression and association with disease outcome in primary breast cancer. Breast Cancer Res 9:R25
Greco WR, Faessel H, Levasseur L (1996) The search for cytotoxic synergy between anticancer agents: a case of Dorothy and the ruby slippers? J Natl Cancer Inst 88:699–700
Bicaku E, Marchion DC, Schmitt M, Munster PN (2008) Selective inhibition of histone deacetylase 2 silences progesterone receptor mediated signaling. Cancer Res 68:1513–1519
Travaglini L, Vian L, Billi M, Grignani F, Nervi C (2009) Epigenetic reprogramming of breast cancer cells by valproic acid occurs regardless of estrogen receptor status. Int J Biochem Cell Biol 41:225–234
Zhao C, Lam EW, Sunters A, Enmark E, De Bella MT, Coombes RC, Gustafsson JA, Dahlman-Wright K (2003) Expression of estrogen receptor beta isoforms in normal breast epithelial cells and breast cancer: regulation by methylation. Oncogene 22:7600–7606
Rody A, Holtrich U, Solbach C, Kourtis K, von Minckwitz G, Engels K, Kissler S, Gätje R, Karn T, Kaufmann M (2005) Methylation of estrogen receptor beta promoter correlates with loss of ER-beta expression in mammary carcinoma and is an early indication marker in premalignant lesions. Endocr Relat Cancer 12:903–916
Duong V, Licznar A, Margueron R, Boulle N, Busson M, Lacroix M, Katzenellenbogen BS, Cavaillès V, Lazennec G (2006) ERalpha and ERbeta expression and transcriptional activity are differentially regulated by HDAC inhibitors. Oncogene 25:1799–1806
Lapidus RG, Ferguson AT, Ottaviano YL, Smith HS, Parl FF, Weitzman SA, Baylin SB, Issa JP, Davidson NE (1996) Methylation of estrogen and progesterone receptor gene 5′ CpG islands correlates with lack of estrogen and progesterone receptor gene expression in breast tumors. Clin Cancer Res 2:805–810
Ruh MF, Tian S, Cox LK, Ruh TS (1999) The effects of histone acetylation on estrogen responsiveness in MCF-7 cells. Endocrine 11:157–164
Liu Z, Wong J, Tsai SY, Tsai MJ, O’Malley BW (1999) Steroid receptor coactivator-1 (SRC-1) enhances ligand-dependent and receptor-dependent cell-free transcription of chromatin. Proc Natl Acad Sci USA 96:9485–9490
Minucci S, Horn V, Bhattacharyya N, Russanova V, Ogryzko VV, Gabriele L, Howard BH, Ozato K (1997) A histone deacetylase inhibitor potentiates retinoid receptor action in embryonal carcinoma cells. Proc Natl Acad Sci USA 94:11295–11300
Shang Y, Myers M, Brown M (2002) Formation of the androgen receptor transcription complex. Mol Cell 9:601–610
Fajas L, Egler V, Reiter R, Miard S, Lefebvre AM, Auwerx J (2003) PPARγ controls cell proliferation and apoptosis in an RB-dependent manner. Oncogene 22:4186–4193
Zhao C, Dahlman-Wright K, Gustafsson JA (2008) Estrogen receptor beta: an overview and update. Nucl Recept Signal 6:1–10
Jang ER, Lim SJ, Lee ES, Jeong G, Kim TY, Bang YJ, Lee JS (2004) The histone deacetylase inhibitor trichostatin A sensitizes estrogen receptor alpha-negative breast cancer cells to tamoxifen. Oncogene 23:1724–1736
Morishima S, Shibata MA, Ohmichi M, Otsuki Y (2008) Raloxifene, a selective estrogen receptor modulator, induces mitochondria-mediated apoptosis in human endometrial carcinoma cells. Med Mol Morphol 41:132–138
Fryar-Tita EB, Das JR, Davis JH, Desotos JA, Green S, Southerland WM, Bowen D (2007) Raloxifene and selective cell cycle specific agents: a case for the inclusion of raloxifene in current breast cancer treatment therapies. Anticancer Res 27:1393–1400
Krämer OH, Göttlicher M, Heinzel T (2001) Histone deacetylase as a therapeutic target. Trends Endocrinol Metab 12:294–300
Acknowledgments
We thank Dr. Guowei Gu (Calabria, Italy) for the gift of Plasmid pCMV5-hERβ. We are also grateful to Professor Tao Xi (Nanjing, China) for the supports during this study. This research was supported by the Natural Science Foundation Committee of China (NSFC 30672015, 30700779, 30800257, 30970776, 81000666, 81071194, 31050110123) and the major project from the Ministry of Science and Technology for new drug development (2009ZX09310-004).
Conflict of interest
The authors have no conflicts to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tu, Z., Li, H., Ma, Y. et al. The enhanced antiproliferative response to combined treatment of trichostatin A with raloxifene in MCF-7 breast cancer cells and its relevance to estrogen receptor β expression. Mol Cell Biochem 366, 111–122 (2012). https://doi.org/10.1007/s11010-012-1288-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1288-9