Abstract
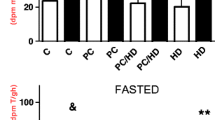
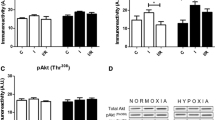
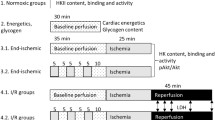
For both, cardioplegia (CP) and ischemic preconditioning (IP), increased ischemic tolerance with reduction in infarct size is well documented. These cardioprotective effects are related to a limitation of high energy phosphate (HEP) depletion. As CP and IP have to be assumed to act by different mechanisms, their effects on myocardial HEP metabolism cannot be assumed to be identical. Therefore, a systematic analysis of myocardial HEP metabolism for both procedures and their combination was performed, addressing the question whether there are different effects on myocardial HEP metabolism by IP and CP. In this study, metabolic control analysis was used to analyze the regulation of HEP metabolism. In open chest pigs subjected to 45 min LAD occlusion (index ischemia), CP and IP preserved myocardial ATP (control (C) 0.14 ± 0.05 μmol/g wwt; CP: 0.95 ± 0.14, IP: 0.61 ± 0.12; p<0.05 C vs. CP and IP) and reduced myocardial necrosis (infarct size IA/RA: C: 90.0 ± 3.0%; CP: 0.0 ± 0.0% but patchy necroses; IP: 5.05 ± 2.1%; p<0.05 C vs. CP and IP). The effects on HEP metabolism, however, were different: CP acted predominantly by slowing down the breakdown of phosphocreatine (PCr) during early phases of ischemia (C: ΔPCr 0–2 min: 5.24 ± 0.32 μmol/g wwt; CP: ΔPCr 0–2 min: 3.38 ± 0.23 μmol/g wwt, p<0.05 vs. C), leaving ATP breakdown during later stages unaffected (C: ΔATP 5–45 min: 1.77 ± 0.11 μmol/g wwt CP: ΔATP 5–45 min: 1.59 ± 0.28 μmol/g wwt, n.s. vs. C). In contrast to CP, in IP PCr breakdown was even increased (IP: ΔPCr 0–2 min: 7.06 ± 0.34 μmol/g wwt, p<0.05 vs. C), but ATP depletion greatly attenuated (IP: ΔATP 5–45 min: 0.48 ± 0.10 μmol/g wwt, p<0.05 vs. C and CP). Combining IP and CP yielded an additive effect with slowing down the breakdown of both PCr (IP+CP: ΔPCr 0–2 min: 5.09± 0.35 μmol/g wwt, p<0.05 vs. C and IP) and ATP (IP+CP: ΔATP 5–45 min: 0.56 ± 0.48 μmol/g wwt, p<0.05 vs. C and CP), resulting in a higher ATP content at the end of index ischemia (1.86 ± 0.46 μmol/g wwt, p<0.05 vs. C, CP and IP). Compared to IP, combining IP+CP achieved also a further reduction in infarct size (IA/RA: 0.0 ± 0.0%, p<0.05 vs IP) and—compared to CP—a disappearance of the patchy necroses. {The concept of major differences in myocardial HEP metabolism during CP and IP is further supported at a molecular level by metabolic control analysis. CP but not IP slowed down the CK reaction velocity at high PCr levels. In contrast to CP exerting a continuous decline in vATPase for any given ATP level, in IP myocardium ATPase reaction velocity was even increased at higher ATP contents, whereas a marked decrease in ATPase reaction velocity was found if ATP levels decreased. The equilibrium of the CK-reaction remained unchanged following CP, whereas IP induced a changing CK equilibrium, which was the more shifted towards PCr the more myocardial HEP content decreased. The data demonstrate different effects of CP and IP on myocardial HEP metabolism, i.e. PCr and ATP breakdown as well as the apparent equilibrium of the creatine kinase (CK)-reaction. For these reasons the combination of the two protective interventions has an additive effect. (Mol Cell Biochem 278: 222–232, 2005)
Similar content being viewed by others
References
Reimer K: The slowing of ischemic energy demand in preconditioned myocardium. Ann N Y Acad Sci 793: 13–26, 1996
Kübler W: Tierexperimentelle Untersuchungen zum Myokardstoff-wechsel im Angina-pectoris-Anfall und beim Herzinfarkt. S. Karger, Basel, 1969
Fell D: Understanding the control of metabolism, Portland Press, London, 1997
Vogt A, Ando H, Arras M, Elsässer A: Lacking adenosine causes myocardial refractoriness. J Am Coll Cardiol 31: 1134–1141, 1998
Kupriyanov V, St Jean M, Xiang B, Butler K, Deslauriers R: Contractile dysfunction caused by normothermic ischaemia and KCl arrest in the isolated pig heart: A 31-P NMR study. J Mol Cell Cardiol 27: 1715–1730, 1995
Schott R, Rohmann S, Braun E, Schaper W: Ischemic preconditioning reduces infarct size in swine myocardium. Circ Res 66: 1133–1142, 1990
Sellevold O, Jynge P, Aarstad K: High performance liquid chromatography: A rapid isocratic method for determination of creatine compounds and adenine nucleotides in myocardial tissue. J Mol Cell Cardiol 18: 517–527, 1986
Vogt AM, Ackermann C, Noe T, Jensen D, Kübler W: Simultaneous detection of high energy phosphates and metabolites of glycolysis and the Krebs cycle by HPLC. Biochem Biophys Res Commun 248: 527–532, 1998
Brand M: Control of oxidative phosphorylation in liver mitochondria and cells: Top-down (C)control (A)nalysis and top-down (E)lasticity (A)nalysis. In: P. Hochacka, P. Lutz, T. Sick, M. Rosenthal, G. van den Thillart (eds). Surviving Hypoxia: Mechanisms of Control and Adaptation. CRC Press, Boca Raton, FL, 1993, pp 295–309
Brand M: Top-down elasticity analysis and its application ti energy metabolism in isolated mitochondria and intact cells. Mol Cell Biochem 184: 13–20, 1998
Sauro H: SCAMP: A general purpose simulator and metabolic control analysis program. Comput Appl Biosci 9: 441–450, 1993
Kübler W, Spieckermann P: Regulation of glycolysis in the ischemic and anoxic myocardium. J Mol Cell Cardiol 1: 351–377, 1970
Reimer K, Jennings R: Myocadial ischemia, hypoxia, and infarction. In: H. Fozzard, E. Haber, R. Jennings, A. Katz, H. Morgan (eds). The Heart and Cardiovascular System: Scientific Foundations, 2nd edn. Raven Press, New York, 1992, pp 1875–1973
Kober IM, Obermayr RP, Brull T, Ehsani N, Schneider B, Spieckermann PG: Comparison of the solutions of Bretschneider, St. Thomas’ Hospital and the National Institutes of Health for cardioplegic protection during moderate hypothermic arrest. Eur Surg Res 30: 243–251, 1998
Schnabel PA, Gebhard MM, Pomykaj T, Schmiedl A, Preusse CJ, Richter J, Bretschneider HJ: Myocardial protection: Left ventricular ultrastructure after different forms of cardiac arrest. Thorac Cardiovasc Surg 35: 148–156, 1987
Mitchell MB, Winter CB, Locke Winter CR, Banerjee A, Harken AH: Cardiac preconditioning does not require myocardial stunning. Ann Thorac Surg 55: 395–400, 1993
Gross G, Fryer R: Sarcolemmal versus mitochondrial ATP-sensitive K+ channels and myocardial preconditioning. Circ Res 84: 973–979, 1999
Grover G, Newburger J, Sleph P, Dzwonczyk S, Taylor S, Ahmed S, Atwal K: Cardioprotective effects of the potassium channel opener cromakalim: Stereoselectivity and effects of myocardial adenine nucleotides. J Pharmacol Exp Ther 257: 156–162, 1991
McPherson P, Pierce G, Cole W: Ischemic cardioprotection by ATP-sensitive K+-channels involves high-energy phosphate preservation. Am J Physiol 265: H1809–1818, 1993
Wikström B, Ronquist G, Waldenström A: Glyburide enhancement of lactate production in ischemic heart is modified by ischemic preconditioning: An in vivo experimental study in pigs by michodialysis technique. J Cardiovasc Pharmacol 27: 622–628, 1996
Kolocassides K, Seymour A, Galinanes M, Hearse D: Paradixical effect of ischemic preconditioning on ischemic contracture? NMR studies of energy metabolism and intracellular pH in rat heart. J Mol Cell Cardiol 28: 1045–1057, 1996
Illes RW, Wright JK, Inners McBride K, Yang CJ, Tristan A: Ischemic preconditioning improves preservation with crystalloid cardioplegia. Ann Thorac Surg 58: 1481–1485, 1994
Lu EX, Chen SX, Hu TH, Xui LM, Yuan MD: Preconditioning enhances myocardial protection in patients undergoing open heart surgery. Thorac Cardiovasc Surg 46: 28–32, 1998
Kolocassides KG, Galinanes M, Hearse DJ: Dichotomy of ischemic preconditioning: Improved postischemic contractile function despite intensification of ischemic contracture. Circulation 93: 1725–1733, 1996
Kemp GJ, Manners DN, Clark JF, Bastin ME, Radda GK: Theoretical modelling of some spatial and temporal aspects of the mitochondrion/creatine kinase/myofibril system in muscle. Mol Cell Biochem 184: 249–289, 1998
Golding E, Teague W, Dobson G: Adjustment of K′ to varying pH and pMg for the creatine kinase, adenylate kinase and ATP hydrolysis equilibria permitting quantitative bioenergetic assessment. J Exp Biol 198: 1775–1782, 1995
Soboll S, Conrad A, Hebisch S: Influence of mitochondrial creatine kinase on the mitochondrial/extramitochondrial distribution of high energy phosphates in muscle tissue: Evidence for a leak in the creatine shuttle. Mol Cell Biochem 133/134: 105–113, 1994
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vogt, A.M., Elsässer, A., Pott-Beckert, A. et al. Myocardial energy metabolism in ischemic preconditioning and cardioplegia: A metabolic control analysis. Mol Cell Biochem 278, 223–232 (2005). https://doi.org/10.1007/s11010-005-7576-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-7576-x