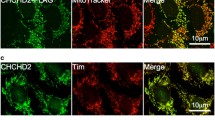
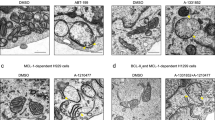
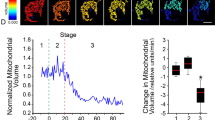
Abstract
The regulation of mitochondrial permeability transition (MPT) is essential for cell survival. Un-controlled opening of the MPT pore is often associated with cell death. Anti-death protein Bcl-2 can block MPT as assessed by the enhanced capacity of mitochondria to accumulate and retain Ca2+. We report here that two proteins of the mitochondrial fission machinery, dynamin-related protein (Drp1) and human mitochondrial fission protein (hFis1), have an antagonistic effect on Bcl-2. Drp1, with the assistance of hFis1, sensitizes cells to MPT by reducing the mitochondrial Ca2+ retention capacity (CRC). While the reduction of CRC by Drp1/hFis1 is linked to mitochondrial fission, the antagonism between Bcl-2 and Drp1 appears to be mediated by mutually exclusive interactions of the two proteins with hFis1. The complexity of protein–protein interactions demonstrated in the present study suggests that in addition to the previously described role of Bcl-2 in the control of apoptosis, Bcl-2 may also participate directly or indirectly in the regulation of mitochondrial fission.
Similar content being viewed by others
References
Orrenius S, Nicotera P: Regulation of cell death: The calcium-apoptosis link. Nat Rev Mol Cell Biol 4: 552–565, 2003
Jiang S, Chow SC, Nicotera P, Orrenius S: Intracellular Ca2+ signals activate apoptosis in thymocytes: Studies using the Ca2+-ATPase inhibitor Thapsigargin. Exp Cell Res 212: 84–92,1994
McGinnis KM, Gnegy ME, Wang KKW: Alterations of extracellular calcium elicit selective modes of cell death and protease activation in SH-SY5Y human neuroblastoma cells. J Neurochem 72: 1853–1863, 1999
Kuo TH, Kim H-RC, Zhu Lp, Yu Y, Lin HM, Tsang W: Modulation of endoplasmic reticulum calcium pump Bcl-2. Oncogene 17: 1903–1910, 1998
Zhu LP, Yu XD, Ling S, Brown RA, Kuo TH: Mitochondrial Ca2+ homeostasis in the regulation of apoptotic and necrotic cell deaths. Cell Calcium 28: 107–117, 2000
Zhu LP, Yu Y, Chua BHL, Ho YS, Kuo TH: Regulation of sodium–calcium exchange and mitochondrial energetics by Bcl-2 in the heart of transgenic mice. J Mol Cell Cardio 33: 2135–2144, 2001
Hansford RG: Physiological role of mitochondrial transport. J Bioenerg Biomembr 26: 495–508, 1994
Di Lisa F, Menabo R, Canton M, Barile M, Bernardi P: Opening of the mitochondriral permeability transition pore causes depletion of mitochondrial and cytosolic NAD and is a causative event in the death of myocytes in postischemic reperfusion of the heart. J Biol Chem 276: 2571–2575, 200
Crompton M: Bax, Bid and the permeabilization of the mitochondrial outer membrane in apoptosis. Curr Opin Cell Biol 12: 414–419, 2000
Scorrano L, Ashiya M, Buttle K, Weiler S, Oakes SA., Mannella CA., Korsmeyer SJ: A distinct pathway remodels mitochondrial cristae and mobilizes cytochrome c during apoptosis. Dev Cell 2: 55–67, 2002
Kuwana T, Mackey MR, Perkins G, Ellisman MR, Latterich M, Schneiter R, Green DR, Newmeyer DD: Bid, Bax, and lipids cooperate to form supramolecular openings in the outer mitochondrial membrane. Cell 111: 331–342, 2002
Annis MG, Zamzami N, Zhu W, Penn LZ, Kroemer G, Leber B, Andrews DW: Endoplasmic reticulum localized Bcl-2 prevents apoptosis when redistribution of cytochrome c is a late event. Oncogene 20: 1939–1952, 2001
Green RG, Kroemer G: The pathophysiology of mitochondrial cell death. Science ([0-9]+): 626–629, 2004
Bernardi P: Mitochondrial transport of cations: Channels, exchangers and permeability transition. Physiol Rev 79: 1127–1155, 1999
Murphy AN, Bredesen DE, Cortopassi G, Wang E, Fiskum G: Bcl-2 potentiates the maximal calcium uptake capacity of neural cell mitochondria. Proc Nat Acad Sci USA 93: 9893–9898, 1996
Zhu L, Ling S, Yu XD, Venkatesh LK, Subramanian T, Chinnadurai G, Kuo TH: Modulation of mitochondrial Ca2+ homeostasis by Bcl-2. J Biol Chem 274: 33267–33273, 1999
Bereiter-Hahn J, Voth M: Dynamics of mitochondria in living cells: Shape changes, dislocation, fusion and fission of mitochondria. Microsc Res Technol ([0-9]+): 198–219, 1994
Smirnova E, Shurland DL, Ryazantsev SN, van der Bliek AM: A human dynamin-related protein controls the distribution of mitochodria. J Cell Biol ([0-9]+): 351–358, 1998
Pitts KR, Yoon Y, Krueger EW, McNiven MA: The dynamin-like protein DLP1 is essential for normal distribution and morphology of the endoplasmic reticulum and mitochondria in mammalian cells. Mol Biol Cell 10: 4403–4417, 1999
Smirnova E, Griparic L, Shurland DL, van der Bliek, AM: Dynamin-related protein Drp1 is required for mitochondrial division in mammalian cells. Mol Biol Cell 12: 2245–2256, 2001
Bleazard W, McCaffery JM, King EJ, Bale S, Mozdy A, Tieu Q, Nunnari J Shaw JM: The dynamin-related GTPase Dnm1 regulates mitochondrial fission in yeast. Nat Cell Biol 1: 298–304, 1999
Labrousse AM, Zappaterra MD, Rube DA, van der Bliek AM: C. elegans dynamin-related protein DRP-1 controls severing of the mitochondrial outer membrane. Mol Cell 4: 815–826, 1999
Yoon Y, Krueger EW, Oswald BJ, McNiven MA: The mitochondrial protein hFis1 regulates mitochondrial fission in mammalian cells through an interaction with the dynamin-like protein DLP1. Mol Cell Biol 15: 5409–5420, 2003
James DI, Parone PA, Mattenberger Y, Martinou JC: hFis1, a novel component of the mammalian mitochondrial fission machinery. J Biol Chem 278: 36373–36379, 2003
Stojanovski D, Koutsopoulos OS, Okamoto K Ryan MT: Levels of human Fis1 at the mitochondrial outer membrane regulate mitochondrial morphology. J. Cell Sci. ([0-9]+): 1201–1210, 2004
Frank S, Gaume B, Bergmann-Leitner ES, Leitner WW, Robert EG, Catez F, Smith CL, Youle RL: The role of dynamin-related protein 1, a mediator of mitochondrial fission, in apoptosis. Dev Cell 1: 515–525, 2001
Frieden M, James D, Castelbou C, Danckaert A, Martinou JC, Demaurex N: Ca2+ homeostasis during mitochondrial fragmentation and perinucear clustering induced by hFis1. J Biol Chem 279: 22704–22714, 2004
Zhu W, Leber B, Andrews DW: Cytoplasmic O-glycosylation prevents cell surface transport of E-cadherin during apoptosis. EMBO J 20: 5999–6007, 2001
Moreadith RW, Fiskum G: Isolation of mitochondria from ascites tumor cells permeabilized with digitonin. Anal Biochem 137: 360–367, 1984
Evetodienko YV, Teplova VV, Azarashvily TS, Kudin A, Prusakova O, Virtanen I, Saris NEL: The Ca2+ threshold for the mitochondrial permeability transition and the content of proteins related to Bcl-2 in rat liver and Zajdela hepatoma mitochondria. Mol Cell Biochem 194: 251–256, 1999
Liu BF, Xu X, Fridman R, Muallem S, Kuo TH: Consequence of functional expression of the plasma membrane Ca2+ pump isoform 1a. J Biol Chem 271: 5536–5544, 1996
Zhu W, Cowie A, Wasfy GW, Penn LZ, Leber B, Andrews DW: Bcl-2 mutants with restricted subcellular location reveal spatially distinct pathways for apoptosis in different cell types. EMBO J 15: 4130–4141, 1996
Suzuki M, Jeong SY, Karbowski M, Youle RJ, Tjandra N: The solution structure of human mitochondria fission protein Fis1 reveals a novel TPR-like helix bundle. J Mol Biol 334: 445–458, 2003
Karbowski M, Lee YJ, Gaume B, Jeong SY, Frank S, Nechushtan A, Santel A, Fuller M. Smith CL, Youle RJ: Spatial and temporal association of Bax with mitochondrial fission sites, Drp1, and Mfn2 during apoptosis. J Cell Biol 159: 931–938, 2002
Ichas F, Jouaville LS, Mazat JP: Mitochondria are excitable organelles capable of generating and conveying electrical and calcium signals. Cell 89: 1145–1153, 1997
Legros F, Lombes A, Frachon P, Rojo M; Mitochondrial fusion in human cells is efficient, requires the inner membrane potential, and is mediated by mitofusins. Mol Biol Cell 13: 4343–4354, 2002
Lee YJ, Jeong SY, Karbowski M, Smith CL, Youle RJ: Roles of the mammalian mitochondrial fission and fusion mediators Fis1, Drp1, and Opa1 in apoptosis. Mol Biol Cell 15: 5001–5011, 2004
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kong, D., Xu, L., Yu, Y. et al. Regulation of Ca2+-induced permeability transition by Bcl-2 is antagonized by Drp1 and hFis1. Mol Cell Biochem 272, 187–199 (2005). https://doi.org/10.1007/s11010-005-7323-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-7323-3