Abstract
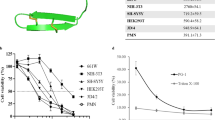
Eosinophil cationic protein (ECP) is a ribonuclease secreted from activated eosinophils that may cause tissue injure as a result of eosinophilic inflammation. ECP possesses bactericidal, antiviral and helminthotoxic activity and inhibits mammalian cell growth. The mechanism by which ECP exerts its toxicity is not known but it has been related to the ability of the protein to destabilise lipid bilayers. We have assessed the involvement of some cationic and aromatic surface exposed residues of ECP in the inhibition of proliferation of mammalian cell lines. We have constructed ECP mutants for the selected residues and assessed their ability to prevent cell growth. Trp10 and Trp35 together with the adjacent stacking residue are critical for the damaging effect of ECP on mammalian cell lines. These residues are also crucial for the membrane disruption activity of ECP. Other exposed aromatic residues packed against arginines (Arg75-Phe76 and Arg121-Tyr122) and specific cationic amino acids (Arg101and Arg104) of ECP play a secondary role in the cell growth inhibition. This may be related to the ability of the protein to bind carbohydrates such as those found on the surface of mammalian cells.
Similar content being viewed by others
References
Peters MS, Rodriguez M, Gleich GJ: Localization of human eosinophil granule major basic protein, eosinophil cationic protein, and eosinophil-derived neurotoxin by immunoelectron microscopy. Lab Investig 54: 656–662, 1986
Giembycz MA, Lindsay MA: Pharmacology of the eosinophil. Pharmacol Rev 51: 213–339, 1999
Barker RL, Loegering DA, Ten RM, Hamann KJ, Pease LR, Gleich GJ: Eosinophil cationic protein cDNA. Comparison with other toxic cationic proteins and ribonucleases. J Immunol 143: 952–955, 1989
Boix E, Leonidas DD, Nikolovski Z, Nogués MV, Cuchillo CM, Acharya, KR: Crystal structure of eosinophil cationic protein at 2.4 Å resolution. Biochemistry 38: 16794–16801, 1999
Sorrentino S, Glitz DG: Ribonuclease activity and substrate preference of human eosinophil cationic protein (ECP). FEBS Lett 288: 23–26, 1991
Boix E, Nikolovski Z, Moiseyev GP, Rosenberg HF, Cuchillo CM, Nogués MV: Kinetic and product distribution analysis of human eosinophil cationic protein indicates a subsite arrangement that favors exonuclease-type activity. J Biol Chem 274: 15605–15614, 1999
Venge P: Monitoring the allergic inflammation. Allergy 59: 26–32, 2004
Venge P, Byström J, Carlson M, Håkansson L, Karawacjzyk M, Peterson C, Sevéus L, Trulson A: Eosinophil cationic protein (ECP): Molecular and biological properties and the use of ECP as a marker of eosinophil activation in disease. Clin Exp Allergy 29: 1172–1186, 1999
Tai PC, Hayes DJ, Clark JB, Spry CJF: Toxic effects of human eosinophil products on isolated rat heart cells in vitro. Biochem J 204: 75–80, 1982
Tai PC, Ackerman SJ, Spry CJF, Dunnette S, Olsen EGJ, Gleich GJ: Toxic effects of human eosinophil products on isolated rat heart cells in vitro. Lancet 21: 643–647, 1987
Fredens K, Dybdahl H, Dahl R, Baandrup U: Extracellular deposit of the cationic proteins ECP and EPX in tissue infiltrations of eosinophils related to tissue damage. APMIS 96: 711–719, 1988
Motojima S, Frigas E, Loegering DA, Gleich GJ: Toxicity of eosinophil cationic proteins for guinea pig tracheal epithelium in vitro. Am Rev Respir Dis 139: 801–805, 1989
Fredens K, Dahl R, Venge P: The Gordon phenomenon induced by the eosinophil cationic protein and eosinophil protein X. J Allergy Clin Immunol 70: 361–366, 1982
Lehrer RI, Szklarek D, Barton A, Ganz T, Hamann KJ, Gleich GJ: Antibacterial properties of eosinophil major basic protein and eosinophil cationic protein. J Immunol 142: 4428–4434, 1989
McLaren DJ, Peterson CGB, Venge P: Schistosoma mansoni: Further studies of the interaction between schistosomula and granulocyte-derived cationic proteins in vitro. Parasitology 88: 491–503, 1984
Molina HA, Kierszenbaum F, Hamann KJ, Gleich GJ: Toxic effects produced or mediated by human eosinophil granule components on Trypanosoma cruzi. Am J Trop Med Hyg 38: 327–384, 1988
Hamann KJ, Gleich GJ, Checkel JL, Loegering DA, McCall JW, Barker RL: In vitro killing of microfilariae of Brugia pahangi and Brugia malayi by eosinophil granule proteins. J Immunol 144: 3166–3173, 1990
Domachowske JB, Dyer KD, Adams AG, Leto TL, Rosenberg HF: Eosinophil cationic protein/RNase 3 is another RNase A-family ribonuclease with direct antiviral activity. Nucleic Acids Res 24: 3507–3513, 1998
Peterson CGB, Skoog V, Venge P: Human eosinophil cationic proteins (ECP and EPX) and their suppressive effects on lymphocyte proliferation. Immunobiology 171: 1–13, 1986
Kimata H, Yoshida A, Ishioka C, Jiang Y, Mikawa H: Eosinophil cationic protein inhibits immunoglobulin production and proliferation in vitro in human plasma cells. Cell Immunol 141: 422–432, 1992
Maeda T, Kitazoe M, Tada H, Llorens R, Salomon DS, Ueda M, Yamada H, Seno M: Growth inhibition of mammalian cells by eosinophil cationic protein. Eur J Biochem 269: 307–316, 2002
Slifman NR, Loegering DA, McKean DJ, Gleich GJ: Ribonuclease activity associated with human eosinophil-derived neurotoxin and eosinophil cationic protein. J Immunol 137: 2913–2917, 1986
Gullberg U, Widegren B, Arnason U, Egesten A, Olsson I: The cytotoxic eosinophil cationic protein (ECP) has ribonuclease activity. Biochem Biophys Res Commun 139: 1239–1242, 1986.
Young JD, Peterson CG, Venge P, Cohn ZA: Mechanism of membrane damage mediated by human eosinophil cationic protein. Nature 321: 613–616, 1986
Carreras E, Boix E, Rosenberg HF, Cuchillo CM, Nogués MV: Both aromatic and cationic residues contribute to the membrane-lytic and bactericidal activity of eosinophil cationic protein. Biochemistry 42: 6636–6644, 2003
Sorrentino S, Glitz DG, Hamann KJ, Loegering DA, Checkel JL, Gleich GJ: Eosinophil-derived neurotoxin and human liver ribonuclease. Identity of structure and linkage of neurotoxicity to nuclease activity. J Biol Chem 267: 14859–14865, 1992
Rosenberg HF: Recombinant human eosinophil cationic protein. Ribonuclease activity is not essential for cytotoxicity. J Biol Chem 270: 7876–7881, 1995
Hohlfeld, JM, Schmiedl, A, Erpenbeck VJ, Venge P, Krug N: Eosinophil cationic protein alters pulmonary surfactant structure and function in asthma. J Allergy Clin Immunol 113: 496–502, 2004
Laccetti P, Portella G, Mastronicola MR, Russo A, Piccoli R, D’Alessio G, Vecchio G: In vivo and in vitro growth-inhibitory effect of bovine seminal ribonuclease on a system of rat thyroid epithelial transformed cells and tumors. Cancer Res 52: 4582–4586, 1992
Futami J, Maeda T, Kitazoe M, Nukui E, Tada H, Seno M, Kosaka M, Yamada H: Preparation of potent cytotoxic ribonucleases by cationization: enhanced cellular uptake and decreased interaction with ribonuclease inhibitor by chemical modification of carboxyl groups. Biochemistry 40: 7518–7524, 2001
Zhang J, Rosenberg, HF: Sequence variation at two epsinophil-associated ribonuclease loci in humans. Genetics 156: 1949–1958, 2000
Jönsson UB, Byström J, Stålenheim G, Venge P: Polymorphism of the eosinophil cationic protein-gene is related to the expression of allergic symptoms. Clin Exp Allergy 32: 1092–1095, 2002
Hayashida M, Fujii T, Hamasu M, Ishiguro M, Hata Y: Similarity between protein-protein and protein-carbohydrate interactions, revealed by two crystal structures of lectins from the roots of pokeweed. J Mol Biol 334: 551–565, 2003
Zhang J, Rosenbergv HF: Complementary advantageous substitutions in the evolution of an antiviral RNase of higher primates. Proc Natl Acad Sci USA 99: 5486–5491, 2002
Zhang L, Rozek A, Hancock RE: Interaction of cationic antimicrobial peptides with model membranes. J Biol Chem 276: 35714–35722, 2001
Ricard-Blum S, Feraud O, Lortat-Jacob H, Rencurosi A, Fukai N, Dkhissi F, Vittet D, Imberty A, Olsen BR, Van Der Rest M: Characterization of endostatin binding to heparin and heparan sulphate by surface plasmon resonance and molecular modeling: role of divalent cations. J Biol Chem 279: 2927–2936, 2004
Ogawa Y, Iwama M, Ohgi K, Tsuji T, Irie M, Itagaki T, Kobayashi H, Inokuchi N: Effect of replacing the aspartic acid/glutamic acid residues of bullfrog sialic acid binding lectin with asparagine/glutamine and arginine on the inhibition of cell proliferation in murine leukemia P388 cells. Biol Pharm Bull 25: 722–727, 2002
Kraulis PJ: MOLSCRIPT: A program to produce both detailed and schematic plots of protein structures. J Appl Crystallogr 24: 946–950, 1991
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carreras, E., Boix, E., Navarro, S. et al. Surface-exposed amino acids of eosinophil cationic protein play a critical role in the inhibition of mammalian cell proliferation. Mol Cell Biochem 272, 1–7 (2005). https://doi.org/10.1007/s11010-005-4777-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-4777-2