Abstract
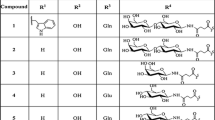
It is demonstrated that gonadotropin-releasing hormone (GnRH) analogs can directly inhibit the proliferation of reproductive tissue cancer cells, but the poor pharmacokinetic properties still restrict their application in treating hormone-dependent diseases. Modifications in position 6 and 10 of natural GnRH can improve the metabolic stability. In order to study the effect of incorporation Cys6 substitution with C-terminal Pro9-NHEt modification and dimerization of linear peptides on metabolic stability and antiproliferative activity of GnRH analogs, two new GnRH analogs [l-Cys6, desGly10, Pro9-NHEt]-GnRH (1) and [d-Cys6, desGly10, Pro9-NHEt]-GnRH (2), and their corresponding dimer derivatives ([l-Cys6, desGly10, Pro9-NHEt]-GnRH)2 (3) and ([d-Cys6, desGly10, Pro9-NHEt]-GnRH)2 (4) were synthesized. Incubation of these analogs with human serum was carried out to evaluate their metabolic stability, and direct growth inhibitory effect of the two dimer derivatives 3 and 4 on MCF-7 human breast cancer cell line was examined by MTT assay. The metabolic stability of dimer derivatives 3 and 4 was remarkably improved in comparison with natural GnRH. The d-Cys6 substituted dimer derivative 4 exhibited higher inhibitory effect (29.6–39.7% growth reduction) on cell growth than its corresponding counterpart 3 (21.8–26.2% growth reduction) at concentration range of 50, 100 and 200 μΜ. The cell growth inhibition of leuprolide was 16.4–27.2% at the tested concentrations. The dimer derivative 4 was the most stable and active GnRH analog in this study and has the potential for future preclinical investigations as promising antitumor drug candidate.




Similar content being viewed by others
Abbreviations
- GnRH:
-
Gonadotropin-releasing hormone
- LHRH:
-
Luteinizing hormone-releasing hormone
- LH:
-
Luteinizing hormone
- FSH:
-
Follicle-stimulating hormone
- DIC:
-
N,N′-Diisopropylcarbodiimide
- HOBt:
-
1-Hydroxy-benzotriazol
- PyBOP:
-
(Benzotriazol-1-yloxy) tripyrrolidinophosphonium hexafluorophosphate
- DIPEA:
-
N,N-Diisopropylethylamine
- TFA:
-
Trifluoroacetic acid
- EDT:
-
1, 2-Ethanedithiol
- DCM:
-
Dichloromethane
- HPLC:
-
High performance liquid chromatography
- MALDI-TOF MS:
-
Matrix-assisted laser desorption/ionization time of flight mass spectrometry
References
Bahk JY, Hyun JS, Lee H, Kim MO, Cho GJ, Lee BH, Choi WS (1998) Expression of gonadotropin-releasing hormone (GnRH) and GnRH receptor mRNA in prostate cancer cells and effect of GnRH on the proliferation of prostate cancer cells. Urol Res 26(4):259–264
Chatzaki E, Bax CM, Eidne KA, Anderson L, Grudzinskas JG, Gallagher CJ (1996) The expression of gonadotropin-releasing hormone and its receptor in endometrial cancer, and its relevance as an autocrine growth factor. Cancer Res 56(9):2059–2065
Dai T, Yin ZF, Zhao HL, Gao Y, Li YH, Li ST, Wang LY (2014) Synthesis of buserelin. Chin J New Drugs 23(15):1806–1810
Eidne KA, Flanagan CA, Millar RP (1985) Gonadotropin-releasing hormone binding sites in human breast carcinoma. Science 229(4717):989–991
Emons G, Ortmann O, Becker M, Irmer G, Springer B, Laun R (1993) High affinity binding and direct antiproliferative effects of LHRH analogues in human ovarian cancer cell lines. Cancer Res 53(22):5439–5446
Flanagan CA, Millar RP, Illing N (1997) Advances in understanding gonadotrophin-releasing hormone receptor structure and ligand interactions. Rev Reprod 2(2):113–120
Fujino M, Kobayashi S, Obayashi M, Shinagawa S, Fukuda T (1972) Structure-activity relationships in the C-terminal part of luteinizing hormone releasing hormone (LH-RH). Biochem Biophys Res Commun 49(3):863–869
Goodwin D, Simerska P, Chang CH, Mansfeld FM, Varamini P, D’Occhio MJ, Toth I (2014) Active immunisation of mice with GnRH lipopeptide vaccine candidates: Importance of T helper or multi-dimer GnRH epitope. Bioorg Med Chem 22(17):4848–4854
Goodwin D, Varamini P, Simerska P, Toth I (2015) Stability, permeability and growth-inhibitory properties of gonadotropin-releasing hormone liposaccharides. Pharm Res 32(5):1570–1584
Gründker C, Emons G (2017) The role of gonadotropin-releasing hormone in cancer cell proliferation and metastasis. Front Endocrinol (Lausanne) 8:187–196
Gründker C, Föst C, Fister S, Nolte N, Günthert AR, Emons G (2010) Gonadotropin-releasing hormone type II antagonist induces apoptosis in MCF-7 and triple-negative MDA-MB-231 human breast cancer cells in vitro and in vivo. Breast Cancer Res 12(4):R49
Harrison GS, Wierman ME, Nett TM, Glode LM (2004) Gonadotropin-releasing hormone and its receptor in normal and malignant cells. Endocr Relat Cancer 11(4):725–748
Karten MJ, Rivier JE (1986) Gonadotropin-releasing hormone analog design. Structure-function studies toward the development of agonists and antagonists: rationale and perspective. Endocr Rev 7(1):44–66
Kottler ML, Starzec A, Carre MC, Lagarde JP, Martin A, Counis R (1997) The genes for gonadotropin-releasing hormone and its receptor are expressed in human breast with fibrocystic disease and cancer. Int J Cancer 71(4):595–599
Kraus S, Levy G, Hanoch T, Naor Z, Seger RM (2004) Gonadotropin-releasing hormone induces apoptosis of prostate cancer cells: role of c-Jun NH2-terminal kinase, protein kinase B, and extracellular signal-regulated kinase pathways. Cancer Res 64(16):5736–5744
Lamari FN, Zompra AA, Pateraki E, Kousidou OC, Magafa V, Karamanos NK, Cordopatis P (2006) Gonadotropin-releasing hormone analogues alter gene expression of metalloproteinases and their tissue inhibitors in human breast cancer epithelial cells. Anticancer Res 26(6B):4615–4621
Limonta P, Moretti RM, Marelli MM, Dondi D, Parenti M, Motta M (1999) The luteinizing hormone-releasing hormone receptor in human prostate cancer cells: messenger ribonucleic acid expression, molecular size, and signal transduction pathway. Endocrinology 140(11):5250–5256
López de Maturana R, Pawson AJ, Lu ZL, Davidson L, Maudsley S, Morgan K, Langdon SP, Millar RP (2008) Gonadotropin-releasing hormone analog structural determinants of selectivity for inhibition of cell growth: support for the concept of ligand-induced selective signaling. Mol Endocrinol 22(7):1711–1722
Matsuo H, Baba Y, Nair RM, Arimura A, Schally AV (1971) Structure of the porcine LH- and FSH-releasing hormone. I. The proposed amino acid sequence. Biochem Biophys Res Commun 43(6):1334–1339
Mezo G, Czajlik A, Manea M, Jakab A, Farkas V, Majer Z, Vass E, Bodor A, Kapuvári B, Boldizsár M, Vincze B, Csuka O, Kovács M, Przybylski M, Perczel A, Hudecz F (2007) Structure, enzymatic stability and antitumor activity of sea lamprey GnRH-III and its dimer derivatives. Peptides 28(4):806–820
Mezo G, Manea M, Szabí I, Vincze B, Kovács M (2008) New derivatives of GnRH as potential anticancer therapeutic agents. Curr Med Chem 15(23):2366–2379
Moradi SV, Varamini P, Toth I (2015) Evaluation of the biological properties and the enzymatic stability of glycosylated luteinizing hormone-releasing hormone analogs. AAPS J 17(5):1135–1143
Moreau JP, Delavault P, Blumberg J (2006) Luteinizing hormone-releasing hormone agonists in the treatment of prostate cancer: a review of their discovery, development, and place in therapy. Clin Ther 28(10):1485–1508
Nikiforovich GV, Marshall GR (1993) Conformation-function relationships in LHRH analogs. Int J Pept Protein Res 42(2):181–193
Pappa EV, Zompra AA, Spyranti Z, Diamantopoulou Z, Pairas G, Lamari FN, Katsoris P, Spyroulias GA, Cordopatis P (2011) Enzymatic stability, solution structure, and antiproliferative effect on prostate cancer cells of leuprolide and new gonadotropin-releasing hormone peptide analogs. Biopolymers 96(3):260–272
Pribylova M, Dvorakova M, Hanusova V, Nemethova I, Skalova L, Vanek T (2011) Paclitaxel conjugation with the analog of the gonadotropin-releasing hormone as a targeting moiety. Int J Pharm 415(1–2):175–180
Rick FG, Block NL, Schally AV (2013) Agonists of luteinizing hormone-releasing hormone in prostate cancer. Expert Opin Pharmacother 14(16):2237–2247
Sealfon SC, Weinstein H, Millar RP (1997) Molecular mechanisms of ligand interaction with the gonadotropin-releasing hormone receptor. Endocr Rev 18(2):180–205
Segal-Abramson T, Kitroser H, Levy J, Schally AV, Sharoni Y (1992) Direct effects of luteinizing hormone-releasing hormone agonists and antagonists on MCF-7 mammary cancer cells. Proc Natl Acad Sci 89(6):2336–2339
Srkalovic G, Wittliff JL, Schally AV (1990) Detection and partial characterization of receptors for [D-Trp6]-luteinizing hormone-releasing hormone and epidermal growth factor in human endometrial carcinoma. Cancer Res 50(6):1841–1846
Tieva A, Stattin P, Wikstrom P, Bergh A, Damber JE (2001) Gonadotropin-releasing hormone receptor expression in the human prostate. Prostate 47(4):276–284
Xu Y, Jiang YF, Wu B (2012) New agonist- and antagonist-based treatment approaches for advanced prostate cancer. J Int Med Res 40(4):1217–1226
Acknowledgements
This study was supported by the Scientific Research Projects in Colleges and Universities of Hebei Province (No. BJ2017055), in part by the Natural Science Foundation of Hebei Province (No. H2017406022), the Undergraduate innovation and entrepreneurship training program of Chengde Medical University (No. 201721) and the Key Discipline Construction Projects of Higher School in Hebei Province ([2013]4).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, S., Zhao, H., Wang, R. et al. Synthesis, Stability and Direct Antiproliferative Effect of New Cysteine Modified GnRH Analogs. Int J Pept Res Ther 25, 1361–1367 (2019). https://doi.org/10.1007/s10989-018-9781-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-018-9781-y