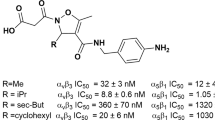
Abstract
We report about the preparation of novel protected Nα(ω-drug) amino acid building units and their straightforward incorporation in solid phase synthesis for the preparation of peptide-drug conjugates. These building units were synthesized applying various coupling methods between anticancer drugs and the side chains of different Nα protected amino acids. Subsequent incorporation of these amino acid-drug motifs into linear and cyclic integrin RGD and NGR containing ligands enabled a non-linear/divergent synthetic pathway of medicinally potential peptide-drug conjugates. The synthetic routes reported in this work are both general and applicable, and significantly expand the scope of the conjugation capabilities for peptide drug conjugates. For the preliminary in vitro evaluation of the novel peptide-drug conjugates reported herein, selective cytotoxicity of two representatives—one linear and one cyclic RGD—camptothecin conjugates were evaluated on αvβ3 integrin overexpressed cancer cell lines.
Graphical Abstract










Similar content being viewed by others
Abbreviations
- AABU:
-
Amino acid building units
- BU:
-
Building unit
- DCC:
-
N,N′-dicyclohexylcarbodiimide
- DCM:
-
Dichloromethane
- DIPEA:
-
N,N′-diisopropylethylamine
- DMF:
-
Dimethylformamide
- EDC:
-
1-Ethyl-3-(3-Dimethylaminopropyl) Carbodiimide Hydrochloride
- Fmoc:
-
N-(9-Fluorenylmethoxycarbonyl)
- IBCF:
-
Isobutyl Chloroformate
- NE:
-
Not evaluated
- PNFC:
-
para-Nitro phenyl chloroformate
- SPPS:
-
Solid phase peptide synthesis
- TFA:
-
Trifluoroacetic acid
- THF:
-
Tetrahydrofuran
- TMS:
-
Trimethylsilyl
References
Arap W, Pasqualini R, Ruoslahti E (1998) Cancer treatment by targeted drug delivery to tumor vasculature in a mouse model. Science 279:377–380. doi:10.1126/science.279.5349.377
Bagnato JD, Eilers AL, Horton RA, Grissom CB (2004) Synthesis and characterization of a cobalamin–colchicine conjugate as a novel tumor-targeted cytotoxin. J Org Chem 69:8987–8996. doi:10.1021/jo049953w
Bellmann-Sickert K, Beck-Sickinger AG (2010) Peptide drugs to target G protein-coupled receptors. Trends Pharmacol Sci 31:434–441. doi:10.1016/j.tips.2010.06.003
Chen K, Chen X (2011) Integrin targeted delivery of chemotherapeutics. Theranostics 1:189–200
Cheng NC, van Zandwijk N, Reid G (2014) Cilengitide inhibits attachment and invasion of malignant pleural mesothelioma cells through antagonism of integrins αvβ3 and αvβ5. PLoS One 9:e90374. doi:10.1371/journal.pone.0090374
Cohen S, Cahan R, Ben-Dov E, Nisnevitch M, Zaritsky A, Firer MA (2007) Specific targeting to murine myeloma cells of Cyt1Aa toxin from bacillus thuringiensis subspecies israelensis. J Biol Chem 282:28301–28308. doi:10.1074/jbc.M703567200
Colombo G et al (2002) Structure-activity relationships of linear and cyclic peptides containing the NGR tumor-homing motif. J Biol Chem 277:47891–47897. doi:10.1074/jbc.M207500200
Corti A, Curnis F (2011) Isoaspartate-dependent molecular switches for integrin–ligand recognition. J Cell Sci 124:515–522. doi:10.1242/jcs.077172
Corti A, Curnis F, Arap W, Pasqualini R (2008) The neovasculature homing motif NGR: more than meets the eye. Blood 112:2628–2635. doi:10.1182/blood-2008-04-150862
Curnis F, Longhi R, Crippa L, Cattaneo A, Dondossola E, Bachi A, Corti A (2006) Spontaneous formation of l-isoaspartate and gain of function in fibronectin. J Biol Chem 281:36466–36476. doi:10.1074/jbc.M604812200
Dai X, Su Z, Liu JO (2000) An improved synthesis of a selective αvβ3-integrin antagonist cyclo(-RGDfK-). Tetrahedron Lett 41:6295–6298. doi:10.1016/S0040-4039(00)01060-1
Dal Pozzo A et al (2010) Novel tumor-targeted RGD peptide–camptothecin conjugates: synthesis and biological evaluation. Bioorg Med Chem 18:64–72. doi:10.1016/j.bmc.2009.11.019
Danhier F, Breton AL, Préat V (2012) RGD-based strategies to target alpha(v) beta(3) integrin in cancer therapy and diagnosis. Mol Pharm 9:2961–2973. doi:10.1021/mp3002733
Firer M, Gellerman G (2012) Targeted drug delivery for cancer therapy: the other side of antibodies. J Hematol Oncol 5:70
Friligou I et al (2013) Divergent and convergent synthesis of polymannosylated dibranched antigenic peptide of the immunodominant epitope MBP(83–99). Bioorg Med Chem 21:6718–6725. doi:10.1016/j.bmc.2013.08.008
Gellerman G, Elgavi A, Salitra Y, Kramer M (2001) Facile synthesis of orthogonally protected amino acid building blocks for combinatorial N-backbone cyclic peptide chemistry. J Pept Res 57:277–291. doi:10.1046/j.1397-002x.2000.0780.x
Gilad Y, Firer MA, Rozovsky A, Ragozin E, Redko B, Albeck A, Gellerman G (2014) “Switch off/switch on” regulation of drug cytotoxicity by conjugation to a cell targeting peptide. Eur J Med Chem 85:139–146. doi:10.1016/j.ejmech.2014.07.073
Goldshaid L et al (2010) Novel design principles enable specific targeting of imaging and therapeutic agents to necrotic domains in breast tumors. Breast Cancer Res 12:R29
Goodman SL, Picard M (2012) Integrins as therapeutic targets. Trends Pharmacol Sci 33:405–412. doi:10.1016/j.tips.2012.04.002
Gurrath M, Muller G, Kessler H, Aumailley M, Timpl R (1992) Conformation/activity studies of rationally designed potent anti-adhesive RGD peptides. Eur J Biochem 210:911–921. doi:10.1111/j.1432-1033.1992.tb17495.x
Haubner R, Finsinger D, Kessler H (1997) Stereoisomeric peptide libraries and peptidomimetics for designing selective inhibitors of the αvβ3 integrin for a new cancer therapy. Angew Chem Int Ed 36:1374–1389. doi:10.1002/anie.199713741
Kriek NMAJ, Filippov DV, van den Elst H, Meeuwenoord NJ, Tesser GI, van Boom JH, van der Marel GA (2003) Stepwise solid phase synthesis of uridylylated viral genome-linked peptides using uridylylated amino acid building blocks. Tetrahedron 59:1589–1597. doi:10.1016/S0040-4020(03)00042-5
Lafrenie RM, Gallo S, Podor TJ, Buchanan MR, Orr FW (1994) The relative roles of vitronectin receptor, E-selectin and α4β1 in cancer cell adhesion to interleukin-1-treated endothelial cells. Eur J Cancer 30:2151–2158. doi:10.1016/0959-8049(94)00354-8
Majumdar S, Siahaan TJ (2012) Peptide-mediated targeted drug delivery. Med Res Rev 32:637–658. doi:10.1002/med.20225
Manfredini S et al (2000) Peptide T-araC conjugates: solid-phase synthesis and biological activity of N4-(acylpeptidyl)-araC. Bioorg Med Chem 8:539–547. doi:10.1016/S0968-0896(99)00317-X
Pasqualini R et al (2000) Aminopeptidase N is a receptor for tumor-homing peptides and a target for inhibiting angiogenesis. Cancer Res 60:722–727
Plow EF, Haas TA, Zhang L, Loftus J, Smith JW (2000) Ligand binding to integrins. J Biol Chem 275:21785–21788. doi:10.1074/jbc.R000003200
Reissner KJ, Aswad DW (2003) Deamidation and isoaspartate formation in proteins: unwanted alterations or surreptitious signals? Cell Mol Life Sci 60:1281–1295. doi:10.1007/s00018-003-2287-5
Robinson NE, Robinson ZW, Robinson BR, Robinson AL, Robinson JA, Robinson ML, Robinson AB (2004) Structure-dependent nonenzymatic deamidation of glutaminyl and asparaginyl pentapeptides. J Pept Res 63:426–436. doi:10.1111/j.1399-3011.2004.00151.x
Ruoslahti E (1996) RGD and other recognition sequences for integrins. Annu Rev Cell Dev Biol 12:697–715. doi:10.1146/annurev.cellbio.12.1.697
Ruoslahti E (2002) Specialization of tumour vasculature. Nat Rev Cancer 2:83–90
Shabbir SH, Eisenberg JL, Mrksich M (2010) An inhibitor of a cell adhesion receptor stimulates cell migration. Angew Chem Int Ed 49:7706–7709. doi:10.1002/anie.201002699
Soudy R, Ahmed S, Kaur K (2012) NGR Peptide ligands for targeting CD13/APN identified through peptide array screening resemble fibronectin sequences. ACS Comb Sci 14:590–599. doi:10.1021/co300055s
van Hensbergen Y et al (2002) A doxorubicin-CNGRC-peptide conjugate with prodrug properties. Biochem Pharmacol 63:897–908. doi:10.1016/S0006-2952(01)00928-5
Vig BS, Huttunen KM, Laine K, Rautio J (2013) Amino acids as promoieties in prodrug design and development. Adv Drug Deliv Rev 65:1370–1385. doi:10.1016/j.addr.2012.10.001
Acknowledgments
The authors thank Mrs. Cherna Moskowitz for generous stipend to Y.G. Financial support was provided by Ariel University Internal Research and Development Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gilad, Y., Waintraub, S., Albeck, A. et al. Synthesis of Novel Protected Nα(ω-Drug) Amino Acid Building Units for Facile Preparation of Anticancer Drug-Conjugates. Int J Pept Res Ther 22, 301–316 (2016). https://doi.org/10.1007/s10989-015-9509-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-015-9509-1