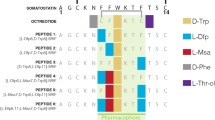
We describe the synthesis and anticancer activities of octapeptide analogs of somatostatin incorporating α,α-dialkylated amino acids. The designed analogs of somatostatin are: d-Phe1-Cys2-Tyr3-d-Trp4-Orn5-Xxx6-Pen7-Thr8-NH2 where Xxx=α-Aminoisobutyric acid (Aib), Diethyl glycine (Deg), 1-Aminocyclopentane carboxylic acid (Ac5c), and, d-Phe1-Cys2-Tyr3-d-Trp4-Lys5-Ac5c6-Pen7-Thr8-NH2 (disulphide bond between Cys2 and Pen7 in all analogs). The conformational studies two of the designed analogs were carried out by NMR techniques and the experimental results suggest a β-turn structure for one of the designed analog. In vivo tumor regression study of two designed analogs on human primary colon tumor xenografts in nude mice demonstrates the anticancer potential of the synthesized analogs.



Similar content being viewed by others
Abbreviations
- Orn:
-
Ornithine
- Pen:
-
Penicillamine
- Aib:
-
α-Aminoisobutyric acid
- Deg:
-
α,α-Di-ethylglycine
- Ac5c:
-
1-Aminocyclopentane carboxylic acid
References
Bauer W., Briner U., Doepfner W., Haller R., Huguenin R., Marbach P., Petcher T. J., Pless M.. (1982) Life Sci 31:1133–1140
Cai R. Z., Karashima T., Guoth J., Szoke B., Olsen D., Schally A. V., (1987) Proc. Natl. Acad. Sci. USA 84:2502–2506
Cai R. Z., Szoke B., Lu R., Fu D., Redding T. W., Schally A. V. (1986). Proc. Natl. Acad. Sci. USA 83:1896–1900
Capello A., Krenning E. P., Bernard B. F., Breeman W. A., van Hagen M. P., de Jong M., (2004) J. Nucl. Med 45:1716–1720
Dasgupta P., Singh A., Mukherjee R., (2002) Biol. Pharm. Bull 25:29–36
Grimberg A., (2004) Cancer Biol. Ther 3:731–733
Hruby V. J., (1985) Trends Pharm. Sci 6:259–262
Janecka A., Zubrzycka M., Janecki T., (2001) J. Pept. Res 58:91–107
Karle I. L., Balaram P., (1990) Biochemistry 29:6747–6756
Karle I. L., Rao R. B., Prasad S., Kaul R., Balaram P., (1994) J. Amer. Chem. Soc 116:10335–10361
Karle I. L., Gurunath R., Prasad S., Kaul R., Rao R.B., Balaram P. (1995) J. Amer. Chem. Soc 117:9632–9637
Kazmierski W. M., Ferguson R. D., Lipkowski A. W., Hruby V. J., (1995) Int. J. Pept. Protein Res 46:265–278
Lamberts S. W. J., de Herder W. W., Hofland L. J., (2002) Trends Endocrinol. Metab 13:451–457
Mattern R. H., Tran T. A., Goodman M., (1999) J. Pept. Res 53:146–160
Mukherjee, R. and Jaggi, M.: 2000. Drug for the treatment of cancer US Patent 6,156,725
Murphy W. A., Heiman M. L., Lance V. A., Mezo I., Coy D. H., (1985) Biochem. Biophys. Res. Commun 132:922–928
Pelton J. T., Whalon M., Cody W. L., Hruby V. J., (1988) Int. J. Pept. Protein Res 31:109–115
Pless M., Waldherr C., Maecke H., Buitrago C., Herrmann R., Mueller-Brand J., (2004) Lung Cancer 45:365–371
Prasad S., BalajiRao R., Bergsstand H., Lindquist B., Becker E. L., Balaram P (1996) Int. J. Pept. Protein Res 48:312–318
Prasad S., Rao R. B., Balaram P., (1995) Biopolymers 35:11–20
Scarpignato C., Pelosini I., (2001) Chemotherapy 47:1–29
Schally A. V., Szepeshazi K., Nagy A., Comaru-Schally A. M., Halmos G., (2004) Cell Mol. Life Sci 61:1042–1068
Schmitt A., Bernhardt P., Nilsson O., Ahlman H., Kolby L., Maecke H. R., Forssell-Aronsson E., (2004) J. Nucl. Med 45:1542–1548
Toniolo C., Crisma M., Valle G., Bonora G. M., Polinelli S., Becker E. L., Freer R. J., Prasad S., Rao R. B., Balaram P., (1989) Peptide Res 2:275–281
Tran T. A., Mattern R. H., Morgan B. A., Taylor J. E., Goodman M., (1999) J. Peptide Res 53:134–145
Veber D. F., Freidlinger R. M., Perlow D. S., Paleveda W. J. Jr., Holly F. W., Strachan R. G., Nutt R. F., Arison B. H., Homnick C., Randall W. C., Glitzer M. S., Saperstein R., Hirschmann R., (1981) Nature 292:55–58
Veber D. F., Saperstein R., Nutt R. F., Freidinger R. M., Brady S. F., Curley P., Perlow D. S., Paleveda W. J., Colton C. D., Zacchei A. G., (1984) Life Sci 34:1371–1378
Weckbecker G., Lewis I., Albert R., Schmid H. A., Hoyer D., Bruns C., (2003) Nat. Rev. Drug Discov 12:999–1017
Zatelli M. C., Piccin D., Bottoni A., Ambrosio M. R., Margutti A., Padovani R., Scanarini M., Taylor J. E., Culler M. D., Cavazzini L., degli Uberti E. C., (2004) J. Clin. Endocrinol. Metab 89:5181–5188
Acknowledgments
We are thankful to Dr. Anand C. Burman, Chairman, Dabur Research Foundation for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prasad, S., Mathur, A., Sharma, R. et al. Octapeptide Analogs of Somatostatin Containing α,α-Dialkylated Amino Acids with Potent Anticancer Activity. Int J Pept Res Ther 12, 179–185 (2006). https://doi.org/10.1007/s10989-005-9005-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-005-9005-0