Abstract
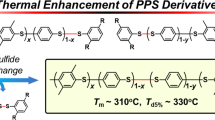
Poly(phenylene sulfide ether) (PPSE) with increased molecular weight was synthesized by means of the quantitative conversion of a poly(phenylene sulfoxide ether sulfide ether) (PPSOESE) precursor via a modified reduction method. PPSOESE precursor was prepared in good yield using polycondensation reaction between bis(4-fluorophenyl) sulfoxide (BFPSO) and bis(4-hydroxyphenyl) sulfide (BHPS). The effects of reaction conditions on the synthesis of PPSOESE and its conversion to PPSE were examined and the conditions were optimized. The results showed that the solvent, reaction temperature, and total monomer concentration are important factors affecting the yield and molecular weight of PPSOESE. For the reduction of PPSOESE to PPSE, the solvents and the feeding rate of the reducing reagent are key factors controlling the quantitative conversion of sulfoxide to sulfide. Structural characterization displayed that PPSOESE is amorphous and has the desired linear structure, and the derived PPSE is semicrystalline with low crystallinity. The study indicated that alternative introduction of ether units induces a gauche conformation relative to that of poly(phenylene sulfide) (PPS), resulting in crystallinity depression. PPSE with a number-average molecular weight of 1.7 × 104 g mol−1 was obtained in 92.4 % yield, which was much higher than that previously reported for PPSE. Thermal analyses showed that the glass transition temperature (Tg) and the melting temperature (Tm) of the obtained PPSE are 92 °C and 192 °C, respectively. Thermogravimetric analysis indicated that PPSE has high thermal stability with a degradation onset temperature above 470 °C.







Similar content being viewed by others
References
Berti C, Celli A, Marianucci E, Vannini M (2007) Preparation and characterization of novel random copoly(arylene ether-thioether ketone)s containing 2,2-bis(4-phenylene)propane units. Eur Polymer J 43:2453–2461
Yang YQ, Duan HJ, Zhang G, Long SR, Yang J, Wang XJ (2013) Effect of the contribution of crystalline and amorphous phase on tensile behavior of poly(phenylene sulfide). J Polym Res 20:198–205
Abbas S, Shahram MA, Ebrahim AL, Ali Y (2006) Sulfide and sulfoxide based poly(ether-amide)s: Synthesis and characterization. Eur Polymer J 42:133–139
Huang XH, Huang W, Fu LC, Yan DY (2012) Synthesis and characterization of thioether-containing polyimides with high refractive indices. J Polym Res 19:9790–9798
Berti C, Celli A, Marianucci E, Vannini M (2005) Sulfur-containing polymers. Synthesis and properties of novel poly(arylene thioether)s based on 2,2-bis(4-mercaptophenyl)propane. Eur Polymer J 41:1812–1820
Lee KS, Kim JP, Lee JS (2010) Synthesis and characterization of low-birefringent crosslinkable fluorinated poly(arylene ether sulfide)s containing pedant phenyl moiety. Polymer 51:632–638
Knauss DM, Edson JB (2006) Poly(aryl ether sulfide)s by sulfide-activated nucleophilic aromatic substitution polymerization. Polymer 47:3996–4003
Berti C, Cell A, Marianucci E, Vannini M (2006) Novel random coploy(arylene ether-thioehter keton)s based on 2,2-bis(4-mercaptophenyl) propane and 4,4′-dihydroxybiphenyl: synthesis and properties. Eur Polymer J 42:2562–2572
Zhang G, Huang GS, Wang XJ, Long SR, Yang J (2011) Synthesis of high refractive index polyamides containing thioether unit. J Polym Res 18:1261–1268
Guo R, McGrath JE (2012) In: Matyjaszewski K, Möller M (eds) Polymer Science: A comprehensive reference, 2nd edn. Holland, Elsevier Science Ltd
Edmonds JT, Hill HW, Okla B (1967) Production of polymers from aromatic compounds. US Pat 3354129
Seo KH, Park LS, Baek JB, Brostow W (1993) Thermal behaviour of poly(phenylene sulfide) and its derivatives. Polymer 34:2524–2527
Jennings, Every G (1968) Polythioethers. GB Pat 1160666
Gu AQ, Xie MJ, Yu ZL (2006) The synthesis and characterization of high molecular weight poly(phenylene sulfide/ether). Chinese Chem Lett 17:1373–1376
Gu AQ, Yu ZL, Li YB (2008) Influence of Polymerization condition on the viscosity and yield of Poly(phenylene sulfide ether). J Appl Polym Sci 110:61–65
Montaudo G, Bruno G, Maravigna P, Finocchiaro P (1973) Bridged polyaromatics synthesis and isomorphous properties of polymers and oligomers containing O, S, CH2 bridges. J Polym Sci: Polym Chem Ed 11:65–79
Wang YF, Hay AS (1997) Macrocylic arylene ether sulfide oligomers: new intermediates for the synthesis of high-performance poly(arylene ether ether sulfide)s. Macromolecules 30:182–193
Percec V, Clough RS (1994) Reductive dehalogenation vs substitution in the polyetherification of bis(ary chloride)s activated by carbonyl groups with hydroquinoners: a potential competition between SET and polar pathways. Macromolecules 27:1535–1547
Gu AQ, Li ZC, Yu ZL, Xu LJ (2013) Molecular weight determination of poly (phenylene sulfide ether). J Appl Polym Sci 129:3682–3686
Park LS, Lee SC, Nam J III (1996) Synthesis and thermal properties of random-copoly(p-phenylene /biphenylene sulfide)s. Polymer 37:1339–1343
Hsiao SH, Lin KH (2004) Soluble aromatic polyamides bearing asymmetrical diary ether groups. Polymer 45:7877–7885
Babu JR, Brink AE, Konas M, Riffle JS (1994) High performance polymer particles for composite matrices: poly(arylene ether ether sulfide)s. Polymer 35:4949–4955
Wang YZ, Lin CH, Chan BW, Hsieh KH (2004) Synthesis and properties of thio-containing poly(ether ether ketone)s. Polym Int 53:320–325
Tabor BJ, Magre EP, Boon J (1971) The crystal structure of poly-p-phenylene sulphide. Euro Polym J 7:1127–1128
Acknowledgments
The authors gratefully acknowledge financial support from the National Natural Science Foundation of China (No. 20974070).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gu, A., Li, Z., Liu, S. et al. Synthesis and characterization of poly(phenylene sulfide ether) by reduction of poly(phenylene sulfoxide ether sulfide ether) precursor. J Polym Res 20, 327 (2013). https://doi.org/10.1007/s10965-013-0327-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-013-0327-x