Abstract
The absorption and emission spectra of six purine derivatives: adenine (I), N(9)-hydroxyethyladenine (II), N(6)-acetyladenine (III), N(6)-isobutyryladenine (IV), guanine (V), and N(2),N(9)-diacetylguanine (VI) have been investigated. The effects of solvent and pH on the positions of λ max (absorption) and λ max (emission) of these compounds were determined. Correlations between the absorption wavelength (λ max ) of these organic compounds and the solvent parameters (D,n,E) or (K,M,N) show that the peak position is affected mainly by specific- and non-specific types of interactions between the solvent and solute. Solvent effects on the electronic absorption band shifts are indicative of the extent of charge reorganization of the solute molecules upon electronic excitation. The Stokes shift (ν abs−ν em) was correlated with the orientation polarizability (Δf) and was found to depend mainly on the dielectric constant and the refractive index of the solvents. This shift reflects the influence of the equilibrium solvent arrangement around the excited solute molecule, which rearranges inertially due to the instantaneous charge redistribution upon radiative deactivation to the electronic ground state. A spectrofluorometric analysis technique was applied for the quantitative analysis of the components of a ternary mixture of compounds (I–III).
Similar content being viewed by others
References
Cadet, J., Vigny, P.: The photochemistry of nucleic acids. In: Morrison, H. (ed.) Photochemistry and the Nucleic Acids. Wiley, New York (1990)
Williams, M., Kowaluk, E.A., Arneric, S.P.: Emerging molecular approaches to pain therapy. J. Med. Chem. 42, 1481–500 (1999)
Lipinski, C.A.: Drug-like properties and the causes of poor solubility and poor permeability. J. Pharmacol. Toxicol. Methods 44, 235–249 (2000)
Edamura, K., Sasai, H.: No self-injurious behavior was found in HPRT-deficient mice treated with 9-ethyladenine. Pharmacol. Biochem. Behav. 61, 175–179 (1998)
Hecht, S.M., McDonald, J.J.: Mass spectra of some 6-substituted ureidopurines and N6-acyladenines. Anal. Biochem. 47, 157–173 (1972)
Lang, P., Magnin, G., Mathis, G., Burger, A., Biellmann, J.F.: Synthesis of 8-(ω-hydroxyalkyl)-, 8-(ω-hydroxyalk-1-enyl)-, and 8-(ω-hydroxyalk-1-ynyl)adenines using the tert-butyldimethylsilyloxymethyl group, a new and versatile protecting group of adenine. J. Org. Chem. 65, 7825–7832 (2000)
Schaeffer, H.J., Bhargava, P.S.: Enzyme inhibitors, V: the syntheses of 6-substituted-(9-hydroxyalkyl)purines and their evaluation as inhibitors of adenosine deaminase. Biochemistry 4, 71–76 (1965)
Kitade, Y., Nakata, Y., Hirota, K., Maki, Y., Pabuccuoglu, A., Torrence, P.F.: 8-Methyladenosine-substituted analogues of 2-5A: synthesis and their biological activities. Nucl. Acids Res. 19, 4103–4108 (1991)
Martin, J.H., Fox, J.E., McChesney, J.D.: Synthesis and cytokinin activity of some N6-acylaminopurines. Phytochemistry 12, 749–752 (1973)
Saparbaev, M., Kleibl, K., Laval, J.: Escherichia coli, Saccharomyces cerevisiae, rat and human 3-methyladenine DNA glycosylases repair 1,N6-ethenoadenine when present in DNA. Nucl. Acids Res. 23, 3750–3755 (1995)
Nagase, H., Haga, A., Kito, H., Sasaki, K., Sato, T.: Enhancing effect of metallothionein on tumor cells invasion in vitro. Cancer Res. Ther. Control 4, 301–307 (1995)
Luhrs, D.C., Viallon, J., Fischer, I.: Excited state spectroscopy and dynamics of isolated adenine and 9-methyladenine. Phys. Chem. Chem. Phys. 3, 1827–1831 (2001)
Mason, S.F.: Purine studies, part II: the ultra-violet absorption spectra of some mono- and poly-substituted purines. J. Chem. Soc. 2071–2081 (1954)
Mason, S.F.: The Electronic Spectra of N-Heteroarornatic Systems, Part VI: The π→π Transitions of Monocyclic Amino- and Mercaptoazines, pp 219–224 (1960)
Stewart, R.F., Davidson, N.: Polarized absorption spectra of purines and pyrimidines. J. Chem. Phys. 39, 255–266 (1963)
Mason, S.: The Pyrimidines. Interscience, New York (1962), Chap. 13
Andréasson, J., Holmén, A., Albinsson, B.: The photophysical properties of the adenine chromophore. Phys. Chem. B 103, 9782–9789 (1999)
Sinsheimer, R.L., Scott, J.F., Loofbourow, J.R.: Ultraviolet absorption spectra at reduced temperatures, II: pyrimidines and purines. J. Biol. Chem. 187, 313–324 (1950)
Santhosh, C., Mishra, P.C.: Electronic spectra of adenine: interaction with dissolved oxygen in solution, oscillation and intensification of n–π * transition. J. Mol. Struct. 220, 25–41 (1990)
Scheibe, G., Felger, E., Robler, G.: Beeinflussung von Absorptionsspektrum, Reaktionsgeschwindigkeit und Gleichgewicht durch Lösungsmittel. Ber. Dtsch. Chem. Ges. 60, 1406–1419 (1927)
Sheppard, S.E.: The effects of environment and aggregation on the absorption spectra of dyes. Rev. Mod. Phys. 14, 303–340 (1942)
Reichardt, C.: Solvents and Solvent Effects in Organic Chemistry, 2nd edn. VCH, Weinheim (1990)
Kamlet, M.J., Abboud, J.L., Taft, R.W.: An examination of linear solvation energy relationships. Prog. Phys. Org. Chem. 13, 485–630 (1981)
Griffiths, T.R., Pugh, D.C.: Correlations among solvent polarity scales, dielectric constant and dipole moment, and a means to reliable predictions of polarity scale values from cu. Coord. Chem. Rev. 29, 129–211 (1979)
Hammud, H.H., Ghannoum, A.M., Masoud, M.S.: Spectral regression and correlation coefficients of some benzaldimines and salicylaldimines in different solvents. Spectrochim. Acta Part A 63, 255–265 (2006)
George, T.F.: Laser-stimulated molecular dynamics and rate processes. J. Phys. Chem. 86, 10–21 (1982)
Albinsson, B.: Dual fluorescence from N6,N6-dimethyladenosine. J. Am. Chem. Soc. 119, 6369–6375 (1997)
Wierzchowski, J., Sepioł, J., Sulikowski, D., Kierdaszuk, B., Shugar, D.: Fluorescence emission properties of 8-azaxanthine and its N-alkyl derivatives: excited-state proton transfer, and potential applications in enzymology. J. Photochem. Photobiol. A: Chem. 179, 276–282 (2006)
Masoud, M.S., Haggag, S.S., El-Nahas, H.M., Abd El-Hi, N.: Electronic transitions and computerized electronic spectral coefficients related to the solvent effects of some azo compounds of barbituric acid. Acta Chim. Hung. 130, 783–804 (1993)
Masoud, M., Khalil, E., Hindawy, A., Ramadan, A.: Structural chemistry of some pyrimidines. Can. J. Anal. Sci. Spectrosc. 50, 207–220 (2005)
Masoud, M.S., Mostafa, M.A., Ahmed, R.H., Abd El Moneim, N.H.: Chemical equilibria and ionization of some compounds containing amide linkage. Molecules 8, 430–438 (2003)
Hammud, H.H., Ghannoum, A.M., Fares, F.A., Abramian, L.K., Bouhadir, K.H.: New 1,6-heptadienes with pyrimidine bases attached—syntheses and spectroscopic analyses. J. Mol. Struct. (2007, in press)
Call, P.R.: Electronic states and luminescence of nucleic acid systems. Ann. Rev. Phys. Chem. 34, 329–357 (1983)
Kahmann, R., Seiler, A., Wulczyn, F.G., Pfaff, E.: The mom gene of bacteriophage Mu: a unique regulatory scheme to control a lethal function. Gene 39, 61–70 (1985)
Wang, Y., Liu, Z., Dixon, C.: Major adenine products from 2-methyl-1,4-naphthoquinone-sensitized photoirradiation at 365 nm. Biochem. Biophys. Res. Commun. 291, 252–1257 (2002)
Hsieh, L.L., Hsu, S.W., Chen, D.S., Santella, R.M.: Immunological detection of aflatoxin B1-DNA adducts formed in vivo. Cancer Res. 48, 6328–6331 (1988)
Weston, A., Bowman, E., Manchester, D., Harris, C.: Fluorescence detection of lesions in DNA, In: Sutherland, B.M., Woodhead, A.D. (eds.) DNA Damage and Repair in Human Tissues, pp. 63–81. Plenum, New York (1990)
Shuker, D., Prevost, V., Friesen, M., Lin, D., Ohshima, H., Bartsch, H.: Urinary markers for measuring exposure to endogenous and exogenous alkylating agents and precursors. Environ. Health Perspect. 99, 33–37 (1993)
Reed, E., Sauerhoff, S., Poirier, M.C.: Quantitation of platinum-DNA binding after therapeutic levels of drug exposure–a novel use of graphite furnace spectrometry. At. Spectrosc. 9, 93–95 (1988)
Wold, S., Sjostrom, M., Eriksson, L.: PLS-regression: a basic tool of chemometrics. Chemom. Intell. Lab. Syst. 58, 109–130 (2001)
Dinç, E., Yücesoy, C., Onur, F.: Simultaneous spectrophotometric determination of mefenamic acid and paracetamol in a pharmaceutical preparation using ratio spectra derivative spectrophotometry and chemometric methods. J. Pharm. Biomed. Anal. 28, 1091–1100 (2002)
Blanco, M., Gene, J., Iturriaga, H., Maspoch, S., Riba, J.: Diode-array detectors in flow-injection analysis mixture resolution by multi-wavelength analysis. Talanta 34, 987–993 (1987)
Andrés, J.V., Reig, F.B., Falcó, P.C.: H-point standard additions method for analyte determination in ternary mixtures. Analyst 120, 299–304 (1995)
Dinç, E., Baydan, E., Kanbur, M., Onur, F.: Spectrophotometric multicomponent determination of sunset yellow, tartrazine and allura red in soft drink powder by double divisor-ratio spectra derivative, inverse least-squares and principal component regression methods. Talanta 58, 579–594 (2002)
Afkhami, A., Bahram, M.: Mean centering of ratio spectra as a new spectrophotometric method for the analysis of binary and ternary mixtures. Talanta 66, 712–720 (2005)
Salinas, F., Nevado, J.J., Mansilla, A.: A new spectrophotometric method for quantitative multicomponent analysis resolution of mixtures of salicylic and salicyluric acids. Talanta 37, 347–351 (1990)
El-Yazbi, F.A., Abdine, H.H., Shaalan, R.A., Korany, E.A.: Spectrophotometric determination of ternary mixtures by the derivative ratio spectrum-zero crossing method. Spectrosc. Lett. 31, 1403–1414 (1998)
El-Yazbi, F.A., Kovar, K.A.: Computerized spectrophotometric method for the determination of atenolol and nifedipine in the presence of degradation products of nifedipine. Sci. Pharm. 66, 325–333 (1998)
Mehrotra, B.D., Jain, P., Anand, N.: Acetylation of adenine and 1-deazaadenine to 9(or 3)-acetyladenine. Indian J. Chem. 4, 146–148 (1966)
Goetz-Luthy, N., Lamb, B.: Polarography of some purine derivatives. J. Pharm. Pharmacol. 8, 410–416 (1956)
Lubczak, R., Duliban, J.: A study of the reaction of adenine with ethylene oxide or with ethylene carbonate. React. Funct. Polym. 52, 127–134 (2002)
Masoud, M.S., Hammud, H.H.: Electronic spectral parameters of the azo indicators—methyl red, methyl orange, PAN, and fast black K-salt. Spectrochim. Acta (A) 57, 977–984 (2001)
Lippert, E.: Dipole moment and electronic structure of excited molecules. Z. Naturforsch. 10, 541–546 (1955)
Lippert, E.: Spektroskopische Bestimmungen des Dipolmomentes aromatischer Verbindungen im ersten angeregten Singulettzustand. Electrochemistry 61, 962–975 (1957)
Mataga, N., Kaifu, Y., Koizumi, M.: The solvent effect on fluorescence spectrum, change of solute-solvent interaction during the lifetime of excited solute molecule. Bull. Chem. Soc. Jpn. 28, 690–691 (1955)
Mataga, N., Kaifu, Y., Koizumi, M.: Solvent effects upon fluorescence spectra and the dipole moments of excited molecules. Bull. Chem. Soc. Jpn. 29, 465–470 (1956)
Parker, C.: Photoluminescence of Solutions. Elsevier, Amsterdam (1968)
Swaminathan, M., Dogra, S.K.: Solvent and pH dependence of absorption and fluorescence spectra of 5-aminoindazole: biprotonic phototautomerism of singly protonated species. J. Am. Chem. Soc. 105, 6223–6228 (1983)
O’Connor, D.B., Scott, G.W., Coulter, D.R., Yavrouian, A.: Temperature dependence of electronic energy transfer and quenching in copolymer films of styrene and 2-(2′-hydroxy-5′-vinylphenyl)-2H-benzotriazole. J. Phys. Chem. 95, 10252–10261 (1991)
Lin, G.C., Awad, E.S., El-Sayed, M.A.: Temperature and pH dependence of the deprotonation step L550 .fwdarw. M412 in the bacteriorhodopsin photocycle. J. Phys. Chem. 95, 10442–10447 (1991)
Clark, L.B., Tinoco, I., Jr.: Correlations in the ultraviolet spectra of the purine and pyrimidine bases. J. Am. Chem. Soc. 87, 11–15 (1965)
Ledger, M.B., Suppan, P.: Anomalous spectroscopic shifts and the structure of 1,4-dioxane. Spectrochim. Acta Part A: Mol. Spectrosc. 23, 3007–3011 (1967)
Boerresen, H.: Fluorescence and tautomerism of protonated and methylated adenine derivatives. Acta Chem. Scand. 21, 2463 (1967)
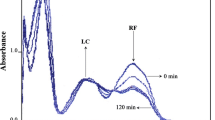
El-Yazbi, F.A., Hammud, H.H., Assi, S.A.: New spectrofluorometric application for the determination of ternary mixtures of drugs. Anal. Chim. Acta 580, 39–46 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hammud, H.H., Bouhadir, K.H., Masoud, M.S. et al. Solvent Effect on the Absorption and Fluorescence Emission Spectra of Some Purine Derivatives: Spectrofluorometric Quantitative Studies. J Solution Chem 37, 895–917 (2008). https://doi.org/10.1007/s10953-008-9289-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-008-9289-8