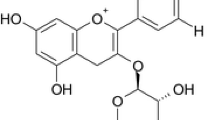
Abstract
A series of 1,3,5-triazines were synthesized and their UV absorption properties were tested. The computational chemistry methods were used to construct quantitative structure-property relationship (QSPR), which was used to computer aided design of new 1,3,5-triazines ultraviolet rays absorber compounds. The experimental UV absorption data are in good agreement with those predicted data using the Time-dependent density functional theory (TD-DFT) [B3LYP/6–311 + G(d,p)]. A suitable forecasting model (R > 0.8, P < 0.0001) was revealed. Predictive three-dimensional quantitative structure-property relationship (3D-QSPR) model was established using multifit molecular alignment rule of Sybyl program, which conclusion is consistent with the TD-DFT calculation. The exceptional photostability mechanism of such ultraviolet rays absorber compounds was studied and confirmed as principally banked upon their ability to undergo excited-state deactivation via an ultrafast excited-state proton transfer (ESIPT). The intramolecular hydrogen bond (IMHB) of 1,3,5-triazines compounds is the basis for the excited state proton transfer, which was explored by IR spectroscopy, UV spectra, structural and energetic aspects of different conformers and frontier molecular orbitals analysis.









Similar content being viewed by others
References
Pei KM, Cui ZH, Chen WG (2013) An adduct of cl-substituted benzotriazole and hydroxy benzophenone as a novel UVA/UVB absorber: theory-guided design, synthesis, and calculations. J Mol Struct 1032:100–104
Cui ZH, Li X, Wang XD, Pei KM, Chen WG (2013) Structure and properties of N-heterocycle-containing benzotriazoles as UV absorbers. J Mol Struct 1054-1055:94–99
Santa MD, Claramunt RM, Bobosik V, Torralba MC, Torres MR, Alkorta I, Elguero J (2013) Synthesis and structural study of 2-arylbenzotriazoles related to Tinuvins. Tetrahedron 69:3027–3038
Liang YP, Xiang B, Liu C, Zhou XD, Wang D, Song S, Bai GH, An Q, Huang QH, Chen SJ, Wei XF (2014) Ultraviolet light absorber with low surface energy: synthesis and characterization. Tetrahedron 70:6585–6593
Damiani E, Baschong W, Greci L (2007) UV-filter combinations under UV-A exposure: concomitant quantification of over-all spectral stability and molecular integrity. J Photochem Photobiol B Biol 87:95–104
Dobashi Y, Yuyama T, Ohkatsu Y (2007) Interaction of ultraviolet absorbers. Polym Degrad Stab 92:1227–1233
Phatangare KR, Gupta VD, Tathe AB, Padalkar VS, Patil VS, Ramasami P, Sekar N (2013) ESIPT inspired fluorescent 2-(4-benzo[d]oxazol-2-yl)naphtho[1,2-d] oxazol-2-yl)phenol: experimental and DFT based approach to photophysical properties. Tetrahedron 69:1767–1777
Barua N, Sarmah P, Hussain I, Deka RC, Buragohain AK (2012) DFT-based QSAR models to predict the antimycobacterial activity of chalcones. Chem Biol Drug Des 79:553–559
Mahmood U, Rashid S, Ali SI, Parveen R, Zaheer-ul-Haq, Ambreen N, Khan KM, Perveen S, Voelter W (2011) 3D-QSPR method of computational technique applied on red reactive dyes by using CoMFA strategy. Int J Mol Sci 12:8862–8877
Liu HX, Shi JQ, Liu H, Wang ZY (2013) Improved 3D-QSPR analysis of the predictive octanoleair partition coefficients of hydroxylated and methoxylated polybrominated diphenyl ethers. Atmos Environ 77:840–845
Gu CG, Ju XH, Jiang X, Yu K, Yang SG, Sun C (2010) Improved 3D-QSAR analyzes for the predictive toxicology of polybrominated diphenyl ethers with CoMFA/CoMSIA and DFT. Ecotoxicol Environ Saf 73:1470–1479
Yang J, Ma M, Wang XD, Jiang XJ, Zhang YY, Yang WQ, Li ZC, Wang XH, Yang B, Ma ML (2015) Synthesis and quantitative structure-activity relationships study for phenylpropenamide derivatives as inhibitors of hepatitis B virus replication. Eur J Med Chem 99:82–91
Ma M, Jiang XJ, Wang XD, Zou H, Yang WQ, Zhang YY, Peng CR, Li ZC, Yang J, Du Q, Ma ML (2016) Synthesis and quantitative structure-activity relationships study for arylpropenamide derivatives as inhibitors of hepatitis B virus replication. Chem Biol Drug Des 88:451–459
Li HG, Niu LY, Xu XF, Zhang ST, Gao F (2011) Therotical investigation of intramolecular proton transfer in the excited states for some newly-designed diphenylethylene derivatives bearing 2-(2-hydroxy-phenyl)-benzotriazole, part. J Fluoresc 21:1721–1728
Farkas R, Lhiaubet-Vallet V, Corbera J, Torincsi M, Gorchs O, Trullas C, Jimenez O, Miranda MA, Novak L (2010) Synthesis of new 2-(2′-hydroxyaryl)benzotriazoles and evaluation of their photochemical behavior as potential UV-filters. Molecules 15:6205–6216
Yin H, Li H, Xia G, Ruan C, Shi Y, Wang H, Jin M, Ding D (2016) A novel non-fluorescent excited state intramolecular proton transfer phenomenon induced by intramolecular hydrogen bonds: an experimental and theoretical investigation. Sci Rep 6:19774–19782
Paul BK, Guchhait N (2011) A computational insight into the photophysics of a potent UV absorber Tinuvin P: critical evaluation of the role of charge transfer interaction and topological properties of the intramolecular hydrogen bonding. Comput Theor Chem 966:250–258
Frisch, MJ, Trucks, GW, Schlegel, HB, Scuseria, GE, Robb, MA, Cheeseman, JR, Scalmani, G, Barone, V, Mennucci, B, Petersson, GA, Nakatsuji, H, Caricato, M, Li, X, Hratchian, HP, Izmaylov, AF, Bloino, J, Zheng, G, Sonnenberg, JL, Hada, M, Ehara, M, Toyota, K, Fukuda, R, Hasegawa, J, Ishida, M, Nakajima, T, Honda, Y, Kitao, O, Nakai, H, Vreven, T, Montgomery, Jr JA, Peralta, JE, Ogliaro, F, Bearpark, M, Heyd, JJ, Brothers, E, Kudin, KN, Staroverov, VN, Kobayashi, R, Normand, J, Raghavachari, K, Rendell, A, Burant, JC, Iyengar, SS, Tomasi, J, Cossi, M, Rega, N, Millam, JM, Klene, M, Knox, JE, Cross, JB, Bakken, V, Adamo, C, Jaramillo, J, Gomperts, R, Stratmann, RE, Yazyev, O, Austin, AJ, Cammi, R, Pomelli, C, Ochterski, JW, Martin, RL, Morokuma, K, Zakrzewski, VG, Voth, GA, Salvador, P, Dannenberg, JJ, Dapprich, S, Daniels, AD, Farkas, O, Foresman, JB, Ortiz, JV, Cioslowski, J, Fox, DJ 2009 Gaussian, Inc, Wallingford CT
Lu T, Chen FW (2012) Multiwfn a multifunctional wavefunction analyzer. J Comput Chem 33:580–592
Clark M, Cramer RD III, Opdenbosch NV (1989) Validation of the general purpose Tripos 5.2 force field. J Comput Chem 10:982–1012
Caballero J, Saavedra M, Fernández M, González-Nilo FD (2007) Agric quantitative structure–activity relationship of rubiscolin analogues as δ opioid peptides using comparative molecular field analysis (CoMFA) and comparative molecular similarity indices analysis (CoMSIA). J Agric Food Chem 55:8101–8104
Tsareva DA, Osolodkin DI, Shulga DA, Oliferenko AA, Pisarev SA, Palyulin VA, Zefirov NS (2011) General purpose electronegativity relaxation charge models applied to CoMFA and CoMSIA study of GSK-3 inhibitors. Mol Inf 30:169–180
Fatemi MH, Shakoori Z, Samghani K (2017) Comparative molecular field analysis of chromatographic hydrophobicity indices for some coumarin analogs. Biomed Chromatogr 31. https://doi.org/10.1002/bmc.3876
Singh RB, Mahanta S, Kar S, Guchhait N (2007) Photo-physical properties of 1-hydroxy-2-naphthaldehyde: a combined fluorescence spectroscopy and quantum chemical calculations. Chem Phys 331:373–383
Paul BK, Samanta A, Guchhait N (2010) Deciphering the photophysics of 5-chlorosalicylic acid: evidence for excited-state intramolecular proton transfer. Photochem Photobiol Sci 9:57–67
Acknowledgements
The financial support of this work was supplied by the NSFC (NO. B020601), Scientific Research Foundation of the Education Department of Sichuan Province (NO. 12ZB128) and Key Research Fund Program (No. Z1123328) of Xihua University. The authors thank the assistant of the students of Prof. Li of College of Chemical engineering, Sichuan University for the work of computational calculations of SYBYL.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X., Xu, Y., Yang, L. et al. Synthesis, Spectra, and Theoretical Investigations of 1,3,5-Triazines Compounds as Ultraviolet Rays Absorber Based on Time-Dependent Density Functional Calculations and three-Dimensional Quantitative Structure-Property Relationship. J Fluoresc 28, 707–723 (2018). https://doi.org/10.1007/s10895-018-2235-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-018-2235-2