Abstract
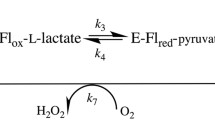
Dihydrolipoamide dehydrogenase (LipDH) transfers two electrons from dihydrolipoamide (DHL) to NAD+ mediated by FAD. Since this reaction is the final step of a series of catalytic reaction of pyruvate dehydrogenase multi-enzyme complex (PDC), LipDH is a key enzyme to maintain the fluent metabolic flow. We reported here the conformational change near the redox center of LipDH induced by NAD+ promoting the access of the DHL to FAD. The increase in the affinity of DHL to redox center was evidenced by the decrease in K M responding to the increase in the concentration of NAD+ in Lineweaver-Burk plots. The fluorescence intensity of FAD transiently reduced by the addition of DHL was not recovered but rather reduced by the binding of NAD+ with LipDH. The fluorescence decay lifetimes of FAD and Trp were prolonged in the presence of NAD+ to show that FAD would be free from the electron transfer from the neighboring Tyrs and the resonance energy transfer efficiency between Trp and FAD lowered. These results consistently reveal that the conformation near the FAD and the surroundings would be so rearranged by NAD+ to allow the easier access of DHL to the redox center of LipDH.







Similar content being viewed by others
Abbreviations
- LipDH:
-
Dihydrolipoamide dehydrogenase
- DHL:
-
Dihydrolipoamide
- FAD:
-
Flavinadenine dinucleotide
- NAD+ :
-
Nicotineamide-adenine dinucleotide
- RET:
-
Resonance excitation energy transfer
References
Hong YS, Kerr DS, Liu T-C, Lusk M, Powell BR, Patel MS (1997) Deficiency of dihydrolipoamide dehydrogenase due to two mutant alleles (E340K and G101del): analysis of a family and prenatal testing. Biochim Biophys Acta 1362(2–3):160–168
Robinson BH, Taylor J, Sherwood WG (1977) Deficiency of dihydrolipoyl dehydrogenase (a component of the pyruvate and α-ketoglutarate dehydrogenase complexes): a cause of congenital chronic lactic acidosis in infancy. Pediatr Res 11:1198–1202
Elpeleg ON, Saada AB, Shaag A, Glustein JZ, Ruitenbeek W, Tein I, Halevy J (1997) Lipoamide dehydrogenase deficiency: a new cause for recurrent myoglobinuria. Muscle Nerve 20(2):238–240
Berger I, Elpeleg ON, Saada A (1996) Lipoamide dehydrogenase activity in lymphocytes. Clin Chim Acta 256(2):197–201
Sakaguchi Y, Yoshino M, Aramaki S, Yoshida I, Yamashita F, Kuhara T, Matsumoto I, Hayashi T (1986) Dihydrolipoyl dehydrogenase deficiency: a therapeutic trial with branched-chain amino acid estriction. Eur J Pediatr 145(4):271–274
Lu HP, Xun L, Xie XS (1998) Single-molecule enzymatic dynamics. Science 282:1877–1882
van den Berg PAW, Feenstra KA, Mark AE, Berendsen HJC, Visser AJWG (2002) Dynamic conformations of flavin adenine dinucleotide: Simulated molecular dynamics of the flavin cofactor related to the time-resolved fluorescence characteristics. J Phys Chem B 106:8858–8869
Liu T-C, Hong YS, Korotchkina LG, Vettakkorumakankav NN, Patel MS (1999) Site-directed mutagenesis of human dihydrolipoamide dehydrogenase: role of lysine-54 and glutamate-192 in stabilizing the thiolate-FAD intermediate. Protein Expr Purif 16:27–39
Mande SS, Sarfaty S, Allen MD, Perham RN, Hol WG (1996) Protein-protein interactions in the pyruvate dehydrogenase multienzyme complex: dihydrolipoamide dehydrogenase complexed with the binding domain of dihydrolipoamide acetyltransferase. Structure 4:277–286
Williams CHJ (1992) Lipoamide dehydrogenase, glutathione reductase, thioredoxin reductase, and mercuric ion reductase-a family of flavoenzyme transhydrogenases. In: Müller F (ed) Chemistry and biochemistry of flavoenzymes, vol 3. CRC Press, Boca Raton, pp 121–211
Reed LJ, Koike M, Levitch ME, Leach FR (1958) Studies on the nature and reactions of protein-bound lipoic acid. J Biol Chem 232:143–158
Willis KJ, Szabo AG (1989) Resolution of tyrosyl and tryptophyl fluorescence emission from subtilisins. Biochemistry 28(11):4902–4908
Zuker M, Szabo AG, Bramall L, Krajcarski DT, Selinger B (1985) Delta function convolution method (DFCM) for fluorescence decay experiments. Rev Sci Instrum 56(1):14–22
Nakashima H, Fukunaga Y, Ueno R, Nishimoto E (2014) Sugar binding effects on the enzymatic reaction and conformation near the active site of pokeweed antiviral protein revealed by fluorescence spectroscopy. J Fluoresc 24(3):951–958
van den Berg PAW, van Hoek A, Walentas CD, Perham RN, Visser AJWG (1998) Flavin fluorescence dynamics and photoinduced electron transfer in Escherichia coli glutathione reductase. Biophys J 74:2046–2058
van den Berg PAW, van Hoek A, Visser AJWG (2004) Evidence for a novel mechanism of time-resolved flavin fluorescence depolarization in glutathione reductase. Biophys J 87(4):2577–2586
Mattevi A, Obmolova G, Sokatch JR, Betzel C, Hol WGJ (1992) The refined crystal structure of Pseudomonas putida lipoamide dehydrogenase complexed with NAD+ at 2.45 Å resolution. Proteins Struct Funct Genet 13:336–351
D’Anna JA, Tollin G (1971) Protein fluorescence and solvent perturbation spectra as probes of flavin-protein interactions in the Shethna flavoprotein. Biochemistry 10:57–64
Nishimoto E, Aso Y, Koga T, Yamashita S (2006) Thermal unfolding process of dihydrolipoamide dehydrogenase Studied by fluorescence spectroscopy. J Biochem 140(3):349–357
Ghisla S, Massey V (1989) Mechanisms of flavoprotein-catalyzed reactions. Eur J Biochem 181(1):1–17
Yang H, Luo G, Karnchanaphanurach P, Louie T-M, Rech I, Cova S, Xun L, Xie XS (2003) Protein conformational dynamics probed by single-molecule electron transfer. Science 302:262–266
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fukamichi, T., Nishimoto, E. Conformational Change Near the Redox Center of Dihydrolipoamide Dehydrogenase Induced by NAD+ to Regulate the Enzyme Activity. J Fluoresc 25, 577–583 (2015). https://doi.org/10.1007/s10895-015-1537-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-015-1537-x