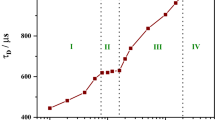
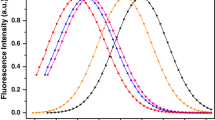
Abstract
The polarity sensitive photo-induced intra-molecular charge transfer (ICT) fluorescence probe (E)-3-(4-methylamino-phenyl)-acrylic acid ethyl ester (MAPAEE) has been used to study the model protein Bovine Serum Albumin (BSA) in its native and thermal and urea induced denatured states. The interaction between BSA and the regular surfactant Sodium Dodecyl Sulphate (SDS) as well as the biologically relevant steroid-based amphiphile Sodium Deoxycholate (NaDC) has also been very keenly followed using this ICT probe. The variation of micellar properties of both SDS and NaDC with increasing ionic strengths and in presence of the chaotrope urea has also been well documemted by the same probe. Steady-state spectroscopy, FRET, and fluorescence anisotropy measurements have been used to gain better insight into these processes and the molecule MAPAEE to be a full-bodied fluorescent probe for studying such intricate biological systems, their properties and interactions.










Similar content being viewed by others
References
Peters T Jr (1985) Serum albumin. Adv Protein Chem 37:161–245
Brown JR (1977) In: Rosenoer VM, Oratz M, Rothschild MA (eds) Albumin structure, function and uses. Pergamon, Oxford, pp 27–51
Peters TJ (1996) All about albumin. Academic, San Diego
He XM, Carter DC (1992) Atomic structure and chemistry of human serum albumin. Nature 358:209–215
Helms MK, Paterson CE, Bhagavan NV, Jameson DM (1997) Time-resolved fluorescent studies on site-directed mutants of human serum albumin. FEBS Lett 408:67–70
Sun C, Yang J, Wu X, Huang X, Wang F, Liu S (2005) Unfolding and refolding of Bovine Serum Albumin induced by cetylpyridinium bromide. Biophys J 88:3518–3524
Vasilescu M, Angelescu D, Almgren M, Valstar A (1999) Interactions of globular proteins with surfactants studied with fluorescence probe methods. Langmuir 15:2635–2643
Valstar A, Almgren M, Brown W, Vasilescu M (2000) The interaction of Bovine Serum Albumin with surfactants studied by light scattering. Langmuir 16:922–927
Deep S, Ahluwalia JC (2001) Interaction of Bovine Serum Albumin with anionic surfactants. Phys Chem Chem Phys 3:4583–4591
Das R, Guha D, Mitra S, Kar S, Lahiri S, Mukherjee S (1997) Intramolecular charge transfer as probing reaction: fluorescence monitoring of protein-surfactant interaction. J Phys Chem A 101:4042–4047
Ruiz-Peñaa M, Oropesa-Nuñeza R, Ponsb T, Louroc SRW, Pérez-Gramatgesa A (2010) Physico-chemical studies of molecular interactions between non-ionic surfactants and Bovine Serum Albumin. Colloids Surf, B Biointerfaces 75:282–289
De S, Das S, Girigoswami A (2007) Spectroscopic probing of bile salt–albumin interaction. Colloids Surf, B 54:74–81
Mukhopadhyay S, Maitra U (2004) Chemistry and biology of bile salts. Curr Sci 87:1666–1683
Reis S, Moutinho CG, Matosa C, de Castro B, Gameiroc P, Lima JLFC (2004) Noninvasive methods to determine the critical micelle concentration of some bile acid salts. Anal Biochem 334:117–126
Coello A, Meijide F, Rodriguez Nunez E, Vaazquez Tatox J (1996) Aggregation behavior of bile salts in aqueous solutions. J Pharm Sci 85:9–15
Mallick A, Halder B, Chattopadhyay N (2005) Spectroscopic investigation on the interaction of ICT probe 3-Acetyl-4-oxo-6, 7-dihydro-12H Indolo-[2, 3-a] quinolizine with serum albumin. J Phys Chem B 109:14683–14690
Grabowski Z, Rotkiewicz K, Rettig W (2003) Structural changes accompanying intramolecular electron transfer: focus on twisted intramolecular charge-transfer states and structures. Chem Rev 103:3899–4032
Poklar N, Lah J, Salobir M, Maek P, Vesnaver G (1997) pH and temperature-induced molten globule-like denatured states of equinatoxin II: a study by UV-Melting, DSC, far- and near-UV CD spectroscopy, and ANS fluorescence. Biochemistry 36:14345–14352
Haskard CA, Li-Chan ECY (1998) Hydrophobicity of Bovine Serum Albumin and Ovalbumin Determined Using Uncharged (PRODAN) and Anionic (ANS-) Fluorescent Probes. J Agric Food Chem 46:2671–2677
Cardamone M, Puri NK (1992) Spectrofluorimetric assessment of the surface hydrophobicity of proteins. Biochem J 282:589–593
Matulis D, Baumann CG, Bloomfield VA, Lovrien RE (1999) 1-Anilino-8-naphthalene sulfonate as a protein conformational tightening agent. Biopolymers 49:451–458
Daniel E, Weber G (1966) Cooperative effects in binding by Bovine Serum Albumin. I. The binding of 1-Anilino-8-naphthalenesulfonate. Fluorimetric titrations. Biochemistry 5:1893–1900
Chakraborty A, Ghosh S, Kar S, Nath DN, Guchhait N (2009) Dual emission from (E)-3-(4-methylamino-phenyl)-acrylic acid ethyl ester(MAPAEE) and its application as fluorescence probe for studying micellar and protein microenvironment. J Mol Struct 917:148–157
Lakowicz JR (1999) Principles of fluorescence spectroscopy. Plenum, NewYork
Pal SK, Batabyal SK, Ganguly T (2005) Preparation and photophysical properties of nanostructured 9-cyanoanthracene. Chem Phys Lett 406:420–424
Paul BK, Samanta A, Guchhait N (2010) Modulation of excited-state intramolecular proton transfer reaction of 1-Hydroxy-2-naphthaldehyde in different supramolecular assemblies. Langmuir 26:3214–3224
Benesi ML, Hildebrand JH (1949) A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J Am Chem Soc 71:2703–2707
Reichardt C (1994) Solvatochromic dyes as solvent polarity indicators. Chem Rev 94:2319–2358
Kundu S, Maity S, Bera SC, Chattopadhyay N (1997) Twisted intramolecular charge transfer of dimethylaminobenzonitrile in micellar environments A way to look at the orientation of the probe within the apolar microenvironment. J Mol Struct 405:231–238
Kato M, Ozawa S, Hayashi R (1997) Effects of high pressure and temperature on micelle formation of sodium deoxycholate and sodium dodecylsulfate. Lipids 32:1229–1230
Makhatadze GI, Privalov PL (1992) Protein interactions with urea and guanidinium chloride. A calorimetric study. J Mol Biol 226:491–505
Valeur B, Brochan JC (2001) New trends in fluorescence spectroscopy. Springer, Berlin, p 25
Valeur B (2001) Molecular fluorescence: principles and applications. Wiley, New York
Mahanta S, Singh RB, Guchhait N (2009) Study of protein-probe interaction and protective action of surfactant sodium dodecyl sulphate in urea-denatured HSA using charge transfer fluorescence probe methyl ester of N, N-dimethylamino naphthyl acrylic acid. J Fluorescence 19:291–303
Singh RB, Mahanta S, Guchhait N (2009) Study of proteinous and micellar microenvironment using donor acceptor charge transfer fluorosensor N, N-dimethylaminonaphthyl-(acrylo)-nitrile. Spectrochim Acta, Part A: Mol Biomol Spectrosc 72:1103–1111
Ghosh S, Guchhait N (2009) Chemically induced unfolding of Bovine Serum Albumin by urea and sodium dodecyl sulfate: a spectral study with the polarity-sensitive charge-transfer fluorescent probe (E)-3-(4-Methylaminophenyl)acrylic acid methyl ester. Chem Phys Chem 10:1664–1671
Chakrabarty A, Das P, Mallick A, Chattopdhyay N (2008) Effect of surfactant chain length on the binding interaction of a biological photosensitizer with cationic micelles. J Phys Chem B 112:3684–3692
Krishnamoorthy G, Dogra SK (2000) Effect of micelles on dual fluorescence of 2-(4′-N, N-dimethylaminophenyl) pyrido[3, 4-d]imidazole. Chem Phys Lett 323:234–242
Midura RJ, Yanagishita M (1995) Chaotropic solvents increase the critical micellar concentrations of detergents. Anal Biochem 228:318–322
Raghuraman H, Pradhan SK, Chattopadhyay A (2004) Effect of urea on the organization and dynamics of triton X-100 micelles: a fluorescence approach. J Phys Chem B 108:2489–2496
Singh RB, Mahanta S, Guchhait N (2008) Destructive and protective action of sodium dodecyl sulphate micelles on the native conformation of Bovine Serum Albumin: a study by extrinsic fluorescence probe 1-hydroxy-2-naphthaldehyde. Chem Phys Lett 463:183–188
Acknowledgement
This work is supported by CSIR, India (Project no. 01(2161)07/EMR-II) and DST, India (Project no. SR/S1/PC/26/2008). The authors would like to thank Prof. T. Ganguly of IACS, Kolkata for allowing them the fluorescence lifetime measurements
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghosh, S., Jana, S., Nath, D. et al. Fluorescent Probing of Protein Bovine Serum Albumin Stability and Denaturation Using Polarity Sensitive Spectral Response of a Charge Transfer Probe. J Fluoresc 21, 365–374 (2011). https://doi.org/10.1007/s10895-010-0725-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-010-0725-y