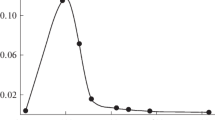
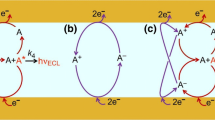
Abstract
Oscillating chemical reactions are complex systems, involving a large number of chemical species. In oscillating chemical reaction, some species, usually a reaction intermediate, exhibit fluctuation in its concentration. In this report, visible oscillating chemiluminescence produced by the addition of luminol (3-aminophthalhydrazide) to the oscillating system of H2O2=KSCN=CuSO4=TMAOH was investigated using spectrofluorimetry method. The effects of ingredient concentration of the oscillating system and complexing agents like citric acid and cysteine on the behavior of the oscillating system were investigated. Moreover, the influence of nonaqueous solvents such as ethanol and ethylene glycol has been studied.
Similar content being viewed by others
References
R. M. Noyes (1989). Some models of chemical oscillators. J. Chem. Educ. 66 (3), 190–191.
R. J. Field and F. W. Schneider (1989). Oscillating chemical reactions and nonlinear dynamic. J. Chem. Educ. 66(3), 195–204.
I. R. Epstein (1989). The role of flow systems in far-from-equilibrium dynamics. J. Chem. Educ. 66(3), 191–195.
R. J. Field, E. Koros, and R. M. Noyes (1972). Oscillating chemical system. II. Through analysis of temporal oscillation in the bromate–cerium–malonic acid system. J. Am. Chem. Soc. 45(25), 8649–8664.
I. R. Wilson and G. M. Harris, (1960). Oxidation of thiocyanate, a pH-independent reaction. J. Am. Chem. Soc. 82(82), 5415–4517.
M. Orbán (1986). Oscillation and bistability in the Cu(II)-catalysed reaction between H2O2 and KSCN. J. Am. Chem. Soc. 108(22), 6893–6898.
R. Jiménes-Prieto, M. Silva, and D. P. Péres-Bendito (1995). Analytical pulse perturbation technique: A tool for analytical determinations in far-from equilibrium dynamic systems. Anal. Chem. 67(4), 729–734.
R. Jiménes-Prieto, M. Silva, and D. P. Péres-Bendito (1998). Approching the use of oscillating reaction for analytical monitoring. Analyst 123, 1R–8R.
S. Sattar and I. R. Epstein (1990). Interaction of luminol with the oscillating system H2O2–KSCN–CuSO4–NaOH. J. Phys. Chem. 94(1), 275–277.
J. Amrehn, A. Resch, and F. W. Schneider (1988). Oscillating chemiluminescence with luminol in the continuous flow stirred tank reactor. J. Phys. Chem. 92(12), 3318–3320.
A. Samadi-Maybodi and S. M. Ourad (2003). Studies of visible oscillating chemiluminescence with a luminol–H2O2–KSCN–CuSO4–NaOH system in batch reactor. Luminescence 18(1), 42–48.
A. Samadi-Maybodi and M. K. Tavakoli-Amiri (2004). Studies of oscillating chemical in the H2O2–KSCN–CuSO4–NaOH system using a conductometry method. Transition Metal Chem. 29(7).
W. C. Bary (1921). A periodic reaction in homogeneous solution and its reaction to catalysis. J. Am. Chem. Soc. 43, 1262–1266.
R. J. Field (1972). A reaction periodic time and space. J. Chem. Educ. 49(5), 308–311.
B. L. Clarke (1992). Stoichiometric network analysis of the oxalate-persulfate-silver oscillator. J. Chem. Phys. 97(4), 2459–2472.
Y. Luo, M. Orbàn, K. Kustin, and I. R. Epstein (1989). Mechanistic study of oscillations and bistability in the Cu(II)-catalyzed reaction between H2O2 and KSCN. J. Am. Chem. Soc. 111(13), 4542–4548.
R. C. Weast (1978). Handbook of Chemistry and Physics, 1st student ed., CRC Press, Boca Raton, p. B-106.
J. A. Dean (1995). Analytical Chemistry Handbook, McGraw-Hill, New York.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Samadi-Maybodi, A., Naseri, N. & Chaichi, M.J. Fluorimetry Studies of Oscillating Chemilumnescence in the Luminol= H2O2= KSCN= CuSO4= TMAOH System. J Fluoresc 15, 117–122 (2005). https://doi.org/10.1007/s10895-005-2518-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10895-005-2518-2