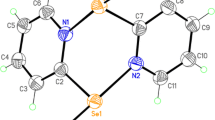
The preparation of the new disulfide compound 4,5-diethylthio-4-cyclopenten-l,3-dione (1) from 4,5-dichloro- 4-cyclopenten-l,3-dione and ethanethiol is described. The title compound has been isolated and characterized in solution by IR and NMR spectroscopies, and the solid-state structure solved by X-ray crystallography. 4,5-Diethylthio-4-cyclopenten-l,3-dione crystallizes in the triclinic space group P-l, a = 7.230(4) Å, b = 8.191(5) Å, c = 10.187(6) Å, α = 108.48(1)°, β = 97.75(1)°, γ = 105.90(1)°, V = 533.8(6) Å3, Z = 2, and d calc = 1.346 Mg/m3; R = 0.0406, R w = 0.0967 for 1394 reflections with I > 2σ(I). The redox properties of 4,5-diethylthio-4-cyclopenten-l,3-dione have been examined by cyclic voltammetry in CH2C12 solution, where a reversible one-electron reduction was found. The reduction behavior of the diethylthio derivative is discussed relative to the closely related 4,5-bis(p-tolylthio)-4-cyclopenten-l,3-dione, whose radical anion exhibits quasi-reversible reduction behavior due to competitive C(dione ring)–S bond cleavage. The differences in the electrochemical behavior of the two disulfides are discussed with respect to the ancillary R group.


Similar content being viewed by others
References
Watson, W.H.; Chen, T.; Richmond, M.G. J. Chem. Crystallogr. 2004, 34, 765.
Watson, W.H.; Wu, G.; Richmond, M.G. J. Chem. Crystallogr. 2004, 34, 709.
See also: Bodige, S.G.; Mendez-Rojas, M.; Watson, W.H. J. Chem. Crystallogr. 1999, 29, 57.
Fenske, D.; Becher, H.J. Chem. Ber. 1974, 107, 117.
Shriver, D.F. The Manipulation of Air-Sensitive Compounds, McGraw-Hill: New York, 1969.
Saint Version 6.02, Bruker Analytical X-ray Systems, Inc., 1997–1999, Madison, WI.
SHELXTL Version 5.1, Bruker Analytical X-ray Systems, Inc., 1998, Madison, WI.
PLATON - A Multipurpose Crystallographic Tool, Utrecht University, Utrecht, The Netherlands, Speck, A. L. (2001).
Bard, A.J.; Faulkner, L.R. Electrochemical Methods; Wiley: New York, 1980.
Dolphin, D.; Wick, A. Tabulation of Infrared Spectral Data; Wiley: New York, 1977.
Adams, R.N. Electrochemistry at Solid Electrodes; Marcel Dekker, New York, 1969.
Eugster, C.H.; Balmer, M.; Prewo, R.; Bieri, J.H. Helv. Chim. Acta 1981, 64, 2636.
Bycroft, B.W.; King, T.J.; Shute, R.E. Tet. Lett. 1983, 24, 601.
Antolini, L.; Borsari, M.; Goldoni, D.; Mucci, A.; Schenetti, L. J. Chem. Soc., Perkin Trans. 1 1999, 3207.
Rizvi, S.K.; Hossain, M.B.; van der Helm, D., Acta Crystallogr. Sect. C 1993, 49,151.
Weast, R..C.; Ed.; Handbook of Chemistry and Physics; CRC Press: Cleveland, OH, 56th ed., 1975.
Acknowledgment
Financial support from the Robert A. Welch Foundation (Grants P-0074-WHW and B-1093-MGR) is appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Watson, W.H., Wu, G., Vadapally, P. et al. Synthesis, redox properties, and molecular structure of 4,5-diethylthio-4-cyclopenten-1,3-dione: Cyclic voltammetric evidence for a kinetically stable radical anion. J Chem Crystallogr 36, 55–60 (2006). https://doi.org/10.1007/s10870-005-9023-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-005-9023-y