Abstract
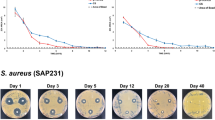
While antibiotic-eluting polymethylmethacrylate space maintainers have shown efficacy in the treatment of bacterial periprosthetic joint infection and osteomyelitis, antifungal-eluting space maintainers are associated with greater limitations for treatment of fungal musculoskeletal infections including limited elution concentration and duration. In this study, we have designed a porous econazole-eluting space maintainer capable of greater inhibition of fungal growth than traditional solid space maintainers. The eluted econazole demonstrated bioactivity in a concentration-dependent manner against the most common species responsible for fungal periprosthetic joint infection as well as staphylococci. Lastly, these porous space maintainers retain compressive mechanical properties appropriate to maintain space before definitive repair of the joint or bony defect.





Similar content being viewed by others
References
Henslee AM, Spicer PS, Shah SR, Tatara AM, Kasper FK, Mikos AG, et al. Use of porous space maintainers in staged mandibular reconstruction. Oral Maxillofac Surg Clin North Am. 2014;26:143–9.
Jakobs O, Schoof B, Klatte TO, Schmidl S, Fensky F, Guenther D, et al. Fungal periprosthetic joint infection in total knee arthroplasty: a systematic review. Orthop Rev. 2015;7:5623 https://doi.org/10.4081/or.2015.5623.
Giannoudis PV, Faour O, Goff T, Kanakaris N, Dimitriou R. Masquelet technique for the treatment of bone defects: tips-tricks and future directions. Injury. 2011;42:591–8. https://doi.org/10.1016/j.injury.2011.03.036.
Anagnostakos K, Furst O, Kelm J. Antibiotic-impregnated PMMA hip spacers: current status. Acta Orthop. 2006;77:628–37. https://doi.org/10.1080/17453670610012719.
Azzam K, Parvizi J, Jungkind D, Hanssen A, Fehring T, Springer B, et al. Microbiological, clinical, and surgical features of fungal prosthetic joint infections: a multi-institutional experience. J Bone Jt Surg Am. 2009;91:142–9. https://doi.org/10.2106/JBJS.I.00574.
Hwang BH, Yoon JY, Nam CH, Jung KA, Lee SC, Han CD, et al. Fungal peri-prosthetic joint infection after primary total knee replacement. J Bone Jt Surg Br. 2012;94:656–9. https://doi.org/10.1302/0301-620X.94B5.28125.
Anagnostakos K, Kelm J, Schmitt E, Jung J. Fungal periprosthetic hip and knee joint infections clinical experience with a 2-stage treatment protocol. J Arthroplast. 2012;27:293–8. https://doi.org/10.1016/j.arth.2011.04.044.
Bariteau JT, Waryasz GR, McDonnell M, Fischer SA, Hayda RA, Born CT. Fungal osteomyelitis and septic arthritis. J Am Acad Orthop Surg. 2014;22:390–401. https://doi.org/10.5435/JAAOS-22-06-390.
Miller AO, Gamaletsou MN, Henry MW, Al-Hafez L, Hussain K, Sipsas NV, et al. Successful treatment of Candida osteoarticular infections with limited duration of antifungal therapy and orthopedic surgical intervention. Infect Dis. 2015;47:144–9. https://doi.org/10.3109/00365548.2014.974207.
Sealy PI, Nguyen C, Tucci M, Benghuzzi H, Cleary JD. Delivery of antifungal agents using bioactive and nonbioactive bone cements. Ann Pharmacother. 2009;43:1606–15. https://doi.org/10.1345/aph.1M143.
Rouse MS, Heijink A, Steckelberg JM, Patel R. Are anidulafungin or voriconazole released from polymethylmethacrylate in vitro? Clin Orthop Relat Res. 2011;469:1466–9. https://doi.org/10.1007/s11999-010-1643-7.
Goss B, Lutton C, Weinrauch P, Jabur M, Gillett G, Crawford R. Elution and mechanical properties of antifungal bone cement. J Arthroplast. 2007;22:902–8. https://doi.org/10.1016/j.arth.2006.09.013.
Spicer PP, Shah SR, Henslee AM, Watson BM, Kinard LA, Kretlow JD, et al. Evaluation of antibiotic releasing porous polymethylmethacrylate space maintainers in an infected composite tissue defect model. Acta Biomater. 2013;9:8832–9. https://doi.org/10.1016/j.actbio.2013.07.018.
Shah SR, Tatara AM, Lam J, Lu S, Scott DW, Bennett GN, et al. Polymer-based local antibiotic delivery for prevention of polymicrobial infection in contaminated mandibular implants. ACS Biomater Sci Eng. 2016;2:558–66.
Kusunoki M, Ikeuchi H, Yanagi H, Noda M, Tonouchi H, Mohri Y, et al. Bioresorbable hyaluronate-carboxymethylcellulose membrane (Seprafilm) in surgery for rectal carcinoma: a prospective randomized clinical trial. Surg Today. 2005;35:940–5. https://doi.org/10.1007/s00595-005-3061-0.
Zawaneh PN, Putnam D. Materials in surgery: a review of biomaterials in postsurgical tissue adhesion and seroma prevention. Tissue Eng Part B Rev. 2008;14:377–91. https://doi.org/10.1089/ten.teb.2008.0226.
Wang L, Yoon DM, Spicer PP, Henslee AM, Scott DW, Wong ME, et al. Characterization of porous polymethylmethacrylate space maintainers for craniofacial reconstruction. J Biomed Mater Res B Appl Biomater. 2013;101:813–25. https://doi.org/10.1002/jbm.b.32885.
Kretlow JD, Shi M, Young S, Spicer PP, Demian N, Jansen JA, et al. Evaluation of soft tissue coverage over porous polymethylmethacrylate space maintainers within nonhealing alveolar bone defects. Tissue Eng Part C Methods. 2010;16:1427–38. https://doi.org/10.1089/ten.tec.2010.0046.
Richter SS, Galask RP, Messer SA, Hollis RJ, Diekema DJ, Pfaller MA. Antifungal susceptibilities of Candida species causing vulvovaginitis and epidemiology of recurrent cases. J Clin Microbiol. 2005;43:2155–62. https://doi.org/10.1128/JCM.43.5.2155-2162.2005.
Wyler R, Murbach A, Mohl H. An imidazole derivative (Econazole) as an antifungal agent in cell culture systems. Vitro. 1979;15:745–50.
Heel RC, Brogden RN, Speight TM, Avery GS. Econazole: a review of its antifungal activity and therapeutic efficacy. Drugs. 1978;16:177–201.
Jiranek WA, Hanssen AD, Greenwald AS. Antibiotic-loaded bone cement for infection prophylaxis in total joint replacement. J Bone Jt Surg Am. 2006;88:2487–500. https://doi.org/10.2106/JBJS.E.01126.
ISO 5833: Implants for surgery—Acrylic resin cements [database on the Internet]. International Organization for Standardization. 2002. Accessed 15 Dec 2017.
Tatara AM, Shah SR, Sotoudeh M, Henslee AM, Wong ME, Ratcliffe A, et al. Effects of electron beam sterilization on mechanical properties of a porous polymethylmethacrylate space maintenance device. J Med Devices. 2015;9:024501.
Watts JL, Clinical Institute LS. Performance standards for antimicrobial disk and dilution susceptibility tests for bacteria isolated from animals: approved standard. National Committee for Clinical Laboratory Standards; 2008.
Morell M, Ramis X, Ferrando F, Serra À. New improved thermosets obtained from diglycidylether of bisphenol A and a multiarm star copolymer based on hyperbranched poly (glycidol) core and poly (methyl methacrylate) arms. Macromol Chem Phys. 2012;213:335–43.
Tatara AM, Wong ME, Mikos AG. In vivo bioreactors for mandibular reconstruction. J Dent Res. 2014;93:1196–202. https://doi.org/10.1177/0022034514547763.
Sud IJ, Feingold DS. Action of antifungal imidazoles on Staphylococcus aureus. Antimicrob Agents Chemother. 1982;22:470–4.
Kokjohn K, Bradley M, Griffiths B, Ghannoum M. Evaluation of in vitro activity of ciclopirox olamine, butenafine HCl and econazole nitrate against dermatophytes, yeasts and bacteria. Int J Dermatol. 2003;42:11–7.
Helmick RA, Fletcher AE, Gardner AM, Gessner CR, Hvitved AN, Gustin MC, et al. Imidazole antibiotics inhibit the nitric oxide dioxygenase function of microbial flavohemoglobin. Antimicrob Agents Chemother. 2005;49:1837–43. https://doi.org/10.1128/AAC.49.5.1837-1843.2005.
Darouiche RO, Hamill RJ, Musher DM, Young EJ, Harris RL. Periprosthetic candidal infections following arthroplasty. Rev Infect Dis. 1989;11:89–96.
Seldes RM, Winiarsky R, Jordan LC, Baldini T, Brause B, Zodda F, et al. Liquid gentamicin in bone cement: a laboratory study of a potentially more cost-effective cement spacer. J Bone Jt Surg Am. 2005;87:268–72. https://doi.org/10.2106/JBJS.C.00728.
Majewicz TG, Erazo‐Majewicz PE, Podlas TJ. Cellulose ethers. Encyclopedia of polymer science and technology. Wiley-Interscience, New York, 2003..
Bauer TW, Smith ST. Bioactive materials in orthopaedic surgery: overview and regulatory considerations. Clin Orthop Relat Res. 2002;395:11–22.
Kaitin KI, Mattison N, Northington FK, Lasagna L. The drug lag: an update of new drug introductions in the United States and in the United Kingdom, 1977 through 1987. Clin Pharmacol Ther. 1989;46:121–38.
Firooz A, Nafisi S, Maibach HI. Novel drug delivery strategies for improving econazole antifungal action. Int J Pharm. 2015;495:599–607. https://doi.org/10.1016/j.ijpharm.2015.09.015.
Acknowledgements
This work was supported by the U.S. Army, Navy, NIH, U.S. Air Force, VA and Health Affairs to support the AFIRM II effort, under Award No. W81XWH-14-2-0004. The U.S. Army Medical Research Acquisition Activity, 820 Chandler Street, Fort Detrick MD 21702-5014 is the awarding and administering acquisition office. In addition, further support has been provided by the John S. Dunn Foundation and the National Institutes of Health (R01 AR068073). E.W. received support from a Ruth L. Kirschstein Fellowship from The National Institute of Dental and Craniofacial Research (F31 DE027586). A.M.T. and E.W. would like to thank the Baylor College of Medicine Medical Scientist Training Program and A.M.T. would like to thank the Barrow Scholars Program.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Tatara, A.M., Rozich, A.J., Kontoyiannis, P.D. et al. Econazole-releasing porous space maintainers for fungal periprosthetic joint infection. J Mater Sci: Mater Med 29, 70 (2018). https://doi.org/10.1007/s10856-018-6073-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10856-018-6073-1