Abstract
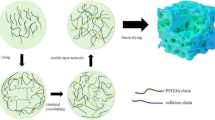
Most gel polymer electrolytes (GPEs) for sodium-ion batteries developed at present have some problems, such as limited absorption uptake of liquid electrolyte (LE) and low ionic conductivity of corresponding GPEs caused by the difficulty of transporting sodium ions with large radius. In view of these problems, hydroxypropyl methyl cellulose (HPMC) is used as raw material to prepare high-performance HPMC-based GPE (HPMC-GPE). When HPMC membrane is added to the LE containing NaClO4 and propylene carbonate (PC) will lead to the ring-opening polymerization of PC, and the products of ring-opening polymerization of PC will be grafted into HPMC side chain. This destroys the dense structure of HPMC membrane and enhances the affinity of HPMC membrane to LE, and then, a large amount of LE (up to 1796 wt.%) can be stored in the molecular chain of HPMC. With the thermal movement of HPMC molecular chain, the volume of HPMC molecular increases significantly in 3D space, providing a large free volume and fast migration channel for sodium ions conduction. The experimental results show that the ionic conductivity of HPMC-GPE is 3.3 × 10–3 S cm−1 and has an electrochemical stability window of 4.72 V. SIB assembled with SnS/rGO working electrode, sodium metal counter electrode and HPMC-GPE has good rate performance and cycle stability.
Graphical Abstract





Similar content being viewed by others
References
Zheng J, Zhao Y, Feng X, Chen W, Zhao Y (2018) J Mater Chem 6:6559. https://doi.org/10.1039/C8TA00530C
Yabuuchi N, Kubota K, Dahbi M, Komaba S (2014) Chem Rev 114:11636. https://doi.org/10.1021/cr500192f
Robinson JB, Finegan DP, Heenan TMM et al (2018). J Electrochem Energy Conversion Storage. https://doi.org/10.1115/1.4038518
Slater MD, Kim D, Lee E, Johnson CS (2013) Adv Func Mater 23:947. https://doi.org/10.1002/adfm.201200691
Lee H, Kim Y-I, Park J-K, Choi JW (2012) Chem Commun 48:8416. https://doi.org/10.1039/C2CC33771A
Yue Y, Binder AJ, Guo B et al (2014) Angew Chem Int Ed 53:3134. https://doi.org/10.1002/anie.201310679
Zeng Z, Jiang X, Li R et al (2016) Adv Sci 3:1600066. https://doi.org/10.1002/advs.201600066
Chaudhari AK, Singh VB (2015) J Electrochem Soc 162:D341. https://doi.org/10.1149/2.0271508jes
Kim, Yoon G, Park I et al (2015) Energy Environ Sci 8:3325. https://doi.org/10.1039/C5EE01876E
Scrosati B (2001) Chem Rec 1:173. https://doi.org/10.1002/tcr.7
Zhou D, Fan L-Z, Fan H, Shi Q (2013) Electrochim Acta 89:334. https://doi.org/10.1016/j.electacta.2012.11.090
Yang YQ, Chang Z, Li MX, Wang XW, Wu YP (2015) Solid State Ionics 269:1. https://doi.org/10.1016/j.ssi.2014.11.015
Gao H, Zhou W, Park K, Goodenough JB (2016) Adv Energy Mater 6:1600467. https://doi.org/10.1002/aenm.201600467
Dong Q, Zhou M, Lin X, Shen L, Feng Y (2018) Eur J Pharm Sci 119:147. https://doi.org/10.1016/j.ejps.2018.04.001
Henry N, Clouet J, Fragale A et al (2017) Drug Deliv 24:999. https://doi.org/10.1080/10717544.2017.1340362
Owusu-Ware SK, Boateng JS, Chowdhry BZ, Antonijevic MD (2019) Int J Pharmaceut: X 1:100033. https://doi.org/10.1016/j.ijpx.2019.100033
Barik B, Nayak PS, Achary LSK, Kumar A, Dash P (2020) New J Chem 44:322. https://doi.org/10.1039/C9NJ03945G
Wrona M, Cran MJ, Nerin C, Bigger SW (2017) Carbohydr Polym 156:108. https://doi.org/10.1016/j.carbpol.2016.08.094
Wang M, Xu H, Yang Z et al (2019) ACS Appl Mater Interfaces 11:41363. https://doi.org/10.1021/acsami.9b14098
Xu K (2004) Chem Rev 104:4303. https://doi.org/10.1021/cr030203g
R Kent Murmann, RC Thompson (1970) J Inorgan Nuclear Chem. 32, 1404. Doi: https://doi.org/10.1016/0022-1902(70)80147-6
Aurbach D, Daroux ML, Faguy PW, Yeager E (1987) J Electrochem Soc 134:1611. https://doi.org/10.1149/1.2100722
Zhang XQ, Chen MJ, Liu CF, Zhang AP, Sun RC (2015) Molecules 20:6033. https://doi.org/10.3390/molecules20046033
Zheng J, Engelhard MH, Mei D et al (2017) Nat Energy 2:17012. https://doi.org/10.1038/nenergy.2017.12
Duval A, Avérous L (2016) ACS Sustain Chem Eng 4:3103. https://doi.org/10.1021/acssuschemeng.6b00081
Logan MW, Langevin S, Tan B et al (2020) J Mater Chem A 8:8485. https://doi.org/10.1039/d0ta01901a
Long L, Wang S, Xiao M, Meng Y (2016) J Mater Chem A 4:10038. https://doi.org/10.1039/c6ta02621d
A Manuel Stephan, KS Nahm (2006) Polymer. 47, 5952. Doi:https://doi.org/10.1016/j.polymer.2006.05.069
Hwang J-Y, Myung S-T, Sun Y-K (2017) Chem Soc Rev 46:3529. https://doi.org/10.1039/C6CS00776G
Lu YC, Ma C, Alvarado J, Dimov N, Meng YS, Okada S (2015) J Mater Chem 3:16971. https://doi.org/10.1039/C5TA03893F
Wu L, Lu H, Xiao L et al (2014) J Mater Chem A 2:16424. https://doi.org/10.1039/C4TA03365E
Xia C, Zhang F, Liang H, Alshareef HN (2017) Nano Res 10:4368. https://doi.org/10.1007/s12274-017-1722-0
Luo C, Shen T, Ji H et al (2020) Small 16:e1906208. https://doi.org/10.1002/smll.201906208
Kim JI, Choi Y, Chung KY, Park JH (2017). Adv Func Mater. https://doi.org/10.1002/adfm.201701768
Seh ZW, Sun J, Sun Y, Cui Y (2015) ACS Cent Sci 1:449. https://doi.org/10.1021/acscentsci.5b00328
Yu Q, Lu Q, Qi X et al (2019) Energy Storage Mater 23:610. https://doi.org/10.1016/j.ensm.2019.03.011
Acknowledgements
This work was supported by the Technology innovation research and development project of Chengdu Science and Technology Bureau (2019-YF05-02393-SN).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Mark Bissett.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jiepeng Chen and Chen Luo are co-first authors.
Rights and permissions
About this article
Cite this article
Chen, J., Luo, C., Huang, Y. et al. Hydroxypropyl methyl cellulose-based gel polymer electrolyte provides a fast migration channel for sodium-ion batteries. J Mater Sci 57, 4311–4322 (2022). https://doi.org/10.1007/s10853-022-06920-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-022-06920-7