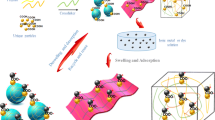
Abstract
A novel and recyclable poly(polyethylene glycol diacrylate/maleamic acid) (p(PEGDA/MALA)) copolymer hydrogel beads were for the first time synthesized through a microfluidic method and used for the removal of heavy mental ions from water. The monodisperse and size-controlled pregel droplets were firstly prepared by a microfluidic device and then polymerized to the hydrogel beads under UV irradiation. The synthesized p(PEGDA/MALA) hydrogel beads were characterized using various techniques and showed good adsorption property for Pb2+. The influencing factors on the adsorption process of Pb2+ were investigated in detail. The experiment results indicated that Pb2+ adsorption process was pH dependent, and agreed with the Langmuir monolayer model and pseudo-second-order equation. The adsorbing mechanism of copolymer hydrogel beads for Pb2+ mainly resulted from the electrostatic interaction and chelation action. The competitive adsorption experiment indicated that the affinity order in multicomponent adsorption was Pb2+>Cu2+>Cd2+. The well-designed p(PEGDA/MALA) copolymer hydrogel beads had excellent adsorption capacity and high recyclability and showed a promising application prospect in the decontamination of heavy metal ions in water.












Similar content being viewed by others
References
Khin MM, Nair AS, Murugan VJ, Babu R, Ramakrishna S (2012) A review on nanomaterials for environmental remediation. Energy Environ Sci 5:8075–8109
Matlock MM, Howerton BS, Atwood DA (2002) Chemical precipitation of heavy metals from acid mine drainage. Water Research 36:4757–4764
Pendergast MTM, Hoek EMV (2011) A review of water treatment membrane nanotechnologies. Energy Environ Sci 4:1946–1971
Ali I (2012) New generation adsorbents for water treatment. Chem Rev 112:5073–5091
Zhang YC, Yang M, Zhang GS, Dionysiou DD (2013) HNO3-involved one-step low temperature solvothermal synthesis of N-doped TiO2 nanocrystals for efficient photocatalytic reduction of Cr(VI) in water. Appl Catal B Environ 142:249–258
Han XG, Kuang Q, Jin MS, Xie ZX, Zheng LS (2009) Synthesis of titania nanosheets with a high percentage of exposed (001) facets and related photocatalytic properties. J Am Chem Soc 131:3152–3153
Ufuk Y, Kemik OF, Hazer B (2010) The removal of heavy metal ions from aqueous solutions by novel pH-sensitive hydrogels. J Hazard Mater 183:521–532
Jing H, Zhao XW, Zhao Y.J, Li J, Xu WY, Wen ZY, Xu M, Gu ZZ (2009) Photonic crystal hydrogel beads used for multiplex biomolecular detection. J Mater Chem 19:5730–5736
Ju XJ, Zhang SB, Zhou MY, Xie R, Yang LH, Chu LY (2009) Novel heavy-metal adsorption material: ion-recognition p(NIPAM-co-BCAm) hydrogels for removal of lead(II) ions. J Hazard Mater 167:114–118
Dragan ES (2014) Design and applications of interpenetrating polymer network hydrogels. A review. Chem Eng J 243:572–590
Shen XP, Shamshina JL, Berton P, Gurau G, Rogers RD (2016) Hydrogels based on cellulose and chitin: fabrication, properties, and applications. Green Chem 18:53–75
Hua R, Li ZK (2014) Sulfhydryl functionalized hydrogel with magnetism: Synthesis, characterization, and adsorption behavior study for heavy metal removal. Chem Eng J 249:189–200
Kasgoz H, Ozgumus S, Orbay M (2003) Modified polyacrylamide hydrogels and their application in removal of heavy metal ions. Polymer 44:1785–1793
Wang WB, Kang YR, Wang AQ (2013) One-step fabrication in aqueous solution of a granular alginate-based hydrogel for fast and efficient removal of heavy metal ions. J Polym Res 20:101–111
Yu L, Liu XK, Yuan WC, Brown LJ, Wang DY (2015) Confined flocculation of ionic pollutants by Poly(L-dopa)-based polyelectrolyte complexes in hydrogel beads for three-dimensional, quantitative, efficient water decontamination. Langmuir 31:6351–6366
Wu NM, Li ZK (2013) Synthesis and characterization of poly(HEA/MALA) hydrogel and its application in removal of heavy metal ions from water. Chem Eng J 215:894–902
Tokarev I, Minko S (2009) Stimuli-responsive hydrogel thin films. Soft Matter 5:511–524
Wang SF, Chen T, Zhang ZL, Shen XC, Lu ZX, Pang DW, Wong KY (2005) Direct electrochemistry and electrocatalysis of heme proteins entrapped in agarose hydrogel films in room-temperature ionic liquids. Langmuir 21:9260–9266
Hund JF, Bertino MF, Zhang GH, Leventis CS, Leventis N, Tokuhiro AT, Farmer J (2003) Formation and entrapment of noble metal clusters in silica aerogel monoliths by γ-radiolysis. J Phys Chem B 107:465–469
Wu XL, Wen T, Guo HL, Yang SB, Wang XK, Xu AW (2013) Biomass-derived sponge-like carbonaceous hydrogels and aerogels for supercapacitors. ACS Nano 7:3589–3597
Merkela TJ, Jonesb SW, Herlihya KP, Kerseyc FR, et al (2011) Using mechanobiological mimicry of red blood cells to extend circulation times of hydrogel microparticles. Proc Natt Acad Sci USA 108:586–591
Helwa Y, Dave N, Froidevaux R, Samadi A, Liu JW (2012) Aptamer-functionalized hydrogel microparticles for fast visual detection of mercury(II) and adenosine. ACS Appl Mater Interfaces 4:2228–2233
Kim J, Serpe MJ, Andrew Lyon L (2004) Hydrogel microparticles as dynamically tunable microlenses. J Am Chem Soc 126:9512–9513
Jin L, Bai RB (2002) Mechanisms of lead adsorption on chitosan/PVA hydrogel beads. Langmuir 18:9765–9770
Dragan ES, Loghin DFA, Cocarta AI (2014) Efficient sorption of Cu2+ by composite chelating sorbents based on potato starch-graft-polyamidoxime embedded in chitosan beads. ACS Appl Mater 6:16577–16592
Farhoudian S, Yadollahi M, Namazi H (2016) Facile synthesis of antibacterial chitosan/CuO bio-nanocompositehydrogel beads. Int J Biol Macromol 82:837–843
Yadollahia M, Farhoudiana S, Barkhordaria S, Gholamali I (2016) Facile synthesis of chitosan/ZnO bio-nanocomposite hydrogel beads as drug delivery systems. Int J Biol Macromol 82: 273–278
Zheng YA, Huang DJ, Wang AQ (2011) Chitosan-g-poly(acrylic acid) hydrogel with crosslinked polymeric networks for Ni2+ recovery. Anal Chim Acta 687:193–200
Li B, Su F, Luo HK, Liang L, Tan B (2011) Hypercrosslinked microporous polymer networks for effective removal of toxic metal ions from water. Microporous Mesoporous Mater 138:207–214
Dragan ES, Lazar MM, Dinu MV, Doroftei F (2012) Macroporous composite IPN hydrogels based on poly(acrylamide) and chitosan with tuned swelling and sorption of cationic dyes. Chem Eng J 204-206: 198–209
Zhu Q, Li ZK (2015) Hydrogel-supported nanosized hydrous manganese dioxide: synthesis, characterization, and adsorption behavior study for Pb2+, Cu2+, Cd2+ and Ni2+ removal from water. Chem Eng J 281:69–80
Febrianto J, Kosasih AN, Sunarso J, Ju YH, Indraswati N, Ismadji S (2009) Equilibrium and kinetic studies in adsorption of heavy metals using biosorbent: a summary of recent studies. J Hazard Mater 162:616–645
Gupta VK, Rastogi A (2008) Biosorption of lead from aqueous solutions by green algae Spirogyra species: kinetics and equilibrium studies. J Hazard Mater 152:407–414
Ho YS, Ng JCY, Mckay G (2000) Kinetics of pollutant sorption by biosorbents: review. Sep Purif Methods 29:189–232
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403
Yan H, Dai J, Yang Z, Yang H, Cheng RS (2011) Enhanced and selective adsorption of copper (II) ions on surface carboxymethylated chitosan hydrogel beads. Chem Eng J 174:586–594
Langmuir I (1916) The constitution and fundamental properties of solids and liquids. J Am Chem Soc 28:2221–2295
Freundlich HMF (1906) Over the adsorption in solution. J Phys Chem 57:385–471
Liu JS, Ma Y, T. Xu W, Shao GQ (2010) Preparation of zwitterionic hybrid polymer and its application for the removal of heavy metal ions from water. J Hazard Mater 178:1021–1029
Liu BJ, Lv X, Meng XH, Yu GL, Wang DF (2013) Removal of Pb(II) from aqueous solution using dithiocarbamate modified chitosan beads with Pb(II) as imprinted ions. Chem Eng J 220:412–419
Krusic MK, Milosavljevic N, Debeljkovic A, Uzum OB, Karadag E (2012) Removal of Pb2+ ions from water by poly(acrylamide-co-sodium methacrylate) hydrogels. Water Air Soil Pollut 223:4355–4368
Yetimoglu EK, Firlak M, Kahraman MV, Deniz S (2011) Removal of Pb2+ and Cd2+ ions from aqueous solutions using guanidine modified hydrogels. Polym Adv Technol 22:612–619
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Nos. 21575125 and 21475116), The Priority Academic Program Development of Jiangsu Higher Education Institution, and University Natural Science Foundation of Jiangsu Province (13KJB150039). Also, the authors thank the Testing Center of Yangzhou University for the characterization data.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Li, J., Tong, J., Li, X. et al. Facile microfluidic synthesis of copolymer hydrogel beads for the removal of heavy metal ions. J Mater Sci 51, 10375–10385 (2016). https://doi.org/10.1007/s10853-016-0258-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-016-0258-0