Abstract
Background
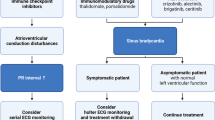
Tyrosine kinase inhibitors (TKIs) have been increasingly used as first-line therapy in hematologic and solid-organ malignancies. Multiple TKIs have been linked with the development of cardiovascular complications, especially atrial arrhythmias, but data on ventricular arrhythmias (VAs) is scarce.
Methods
Herein we describe five detailed cases of VAs related to TKI use in patients with varied baseline cardiovascular risk factors between 2019 and 2022 at three centers. Individual chart review was conducted retrospectively.
Results
Patient ages ranged from 43 to 83 years. Three patients were on Bruton’s TKI (2 ibrutinib and 1 zanubrutinib) at the time of VAs; other TKIs involved were afatinib and dasatinib. Three patients had a high burden of non-sustained ventricular tachycardia (NSVT) requiring interventions, whereas two patients had sustained VAs. While all patients in our case series had significant improvement in VA burden after TKI cessation, two patients required new long-term antiarrhythmic drug therapy, and one had an implantable defibrillator cardioverter (ICD) placed due to persistent VAs after cessation of TKI therapy. One patient reinitiated TKI therapy after control of arrhythmia was achieved with antiarrhythmic drug therapy.
Conclusions
Given the expanding long-term use of TKIs among a growing population of cancer patients, it is critical to acknowledge the association of TKIs with cardiovascular complications such as VAs, to characterize those at risk, and deploy preventive and therapeutic measures to avoid such complications and interference with oncologic therapy. Further efforts are warranted to develop monitoring protocols and optimal treatment strategies for TKI-induced VAs.





Similar content being viewed by others
References
Bohdan M, Kowalczys A, Mickiewicz A, Gruchała M, Lewicka E. Cancer therapy-related cardiovascular complications in clinical practice: current perspectives. J Clin Med Res [Internet]. 2021;10(8). https://doi.org/10.3390/jcm10081647.
Ferguson FM, Gray NS. Kinase inhibitors: the road ahead. Nat Rev Drug Discov. 2018;17(5):353–77.
Wiczer TE, Levine LB, Brumbaugh J, Coggins J, Zhao Q, Ruppert AS, et al. Cumulative incidence, risk factors, and management of atrial fibrillation in patients receiving ibrutinib. Blood Adv. 2017;1(20):1739–48.
Ganatra S, Sharma A, Shah S, Chaudhry GM, Martin DT, Neilan TG, et al. Ibrutinib-Associated Atrial Fibrillation. JACC Clin Electrophysiol. 2018;4(12):1491–500.
Guha A, Derbala MH, Zhao Q, Wiczer TE, Woyach JA, Byrd JC, et al. Ventricular Arrhythmias Following Ibrutinib Initiation for Lymphoid Malignancies. J Am Coll Cardiol. 2018;72(6):697–8.
Lampson B, Yu L, Barrientos J, Jacobsen E, Banerji V, Jones J, et al. Ventricular arrhythmias and sudden death in patients taking ibrutinib [Internet]. Vol. 16, Clinical Lymphoma Myeloma and Leukemia. 2016. S52. https://doi.org/10.1016/j.clml.2016.07.075.
Salem JE, Manouchehri A, Bretagne M, Lebrun-Vignes B, Groarke JD, Johnson DB, et al. Cardiovascular Toxicities associated with ibrutinib. J Am Coll Cardiol. 2019;74(13):1667–78.
Tam CS, Opat S, D’Sa S, Jurczak W, Lee HP, Cull G, et al. A randomized phase 3 trial of zanubrutinib vs ibrutinib in symptomatic Waldenström macroglobulinemia: the ASPEN study. Blood. 2020;136(18):2038–50.
Byrd JC, Hillmen P, Ghia P, Kater AP, Chanan-Khan A, Furman RR, et al. Acalabrutinib versus ibrutinib in previously treated chronic lymphocytic leukemia: results of the first randomized phase III trial. J Clin Oncol. 2021;39(31):3441–52.
Bhat SA, Gambril JA, Azali L, Chen ST, Rosen L, Palettas M, et al. Ventricular arrhythmias and sudden death events following acalabrutinib initiation. Blood [Internet]. 2022; https://doi.org/10.1182/blood.2022016953.
Beyer A, Ganti B, Majkrzak A, Theyyunni N. A Perfect Storm: Tyrosine kinase inhibitor-associated polymorphic ventricular tachycardia. J Emerg Med. 2017;52(4):e123–7.
Wallace N, Wong E, Cooper D, Chao H. A case of new-onset cardiomyopathy and ventricular tachycardia in a patient receiving ibrutinib for relapsed mantle cell lymphoma. Clin Case Rep. 2016;4(12):1120–1.
Tomcsányi J, Nényei Z, Mátrai Z, Bózsik B. Ibrutinib, an approved tyrosine kinase inhibitor as a potential cause of recurrent polymorphic ventricular tachycardia. JACC: Clin Electrophysiol. 2016;2(7):847–9.
Prashar A, Ilsar R, Roncolato F, Hopkins A. Recurrent ventricular fibrillation with different tyrosine kinase inhibitors for chronic myeloid leukemia. HeartRhythm Case Rep. 2020;6(10):770–3.
Spechbach H, Morel P, IngLorenzini K, Besson M, Gétaz L, Sunthorn H, et al. Reversible ventricular arrythmia induced by dasatinib. Clin Case Rep. 2013;1(1):20–5.
Madgula AS, Singh M, Almnajam M, Pickett CC, Kim AS. Ventricular tachycardia storm in a patient treated with ibrutinib for Waldenstrom macroglobulinemia. Cardio Oncol. 2020;2(3):523–6.
Abu Rmilah AA, Lin G, Begna KH, Friedman PA, Herrmann J. Risk of QTc prolongation among cancer patients treated with tyrosine kinase inhibitors. Int J Cancer. 2020;147(11):3160–7.
Du B, Chakraborty P, Azam MA, Massé S, Lai PFH, Niri A, et al. Acute effects of ibrutinib on ventricular arrhythmia in spontaneously hypertensive rats. JACC: Cardio Oncol. 2020;2(4):614–29.
Tuomi JM, Xenocostas A, Jones DL. Increased susceptibility for atrial and ventricular cardiac arrhythmias in mice treated with a single high dose of ibrutinib. Can J Cardiol. 2018;34(3):337–41.
Scheers E, Leclercq L, de Jong J, Bode N, Bockx M, Laenen A, et al. Absorption, metabolism, and excretion of oral 14c radiolabeled ibrutinib: an open-label, phase I, single-dose study in healthy men. Drug Metab Dispos. 2015;43(2):289–97.
Priori SG, Blomström-Lundqvist C, Mazzanti A, Blom N, Borggrefe M, Camm J, et al. ESC guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: the task force for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death of the European Society of Cardiology (ESC). Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC). Eur Heart J. 2015;36(41):2793–867.
Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Circulation. 2018;138(13):e272-391.
Sapp JL, Wells GA, Parkash R, Stevenson WG, Blier L, Sarrazin JF, et al. Ventricular Tachycardia Ablation versus Escalation of Antiarrhythmic Drugs. N Engl J Med. 2016;375(2):111–21.
Madonna R, Pieragostino D, Cufaro MC, Del Boccio P, Pucci A, Mattii L, et al. Sex-related differential susceptibility to ponatinib cardiotoxicity and differential modulation of the Notch1 signalling pathway in a murine model. J Cell Mol Med. 2022;26(5):1380–91.
Funding
This work is supported by National Institutes of Health grants K08 HL148540 (J.W.R), K08 HL153798 (P.C.) and K23 HL145017 (T.B.), R01 HL149134 and HL83359 (S.M.N.) and American Heart Association Career Development Awards (J.W.R. and P.C.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Consent to participate
This retrospective article of case reports is not subject to Informed Consent by the participants.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fazal, M., Wei, C., Chuy, K.L. et al. Tyrosine kinase inhibitor–associated ventricular arrhythmias: a case series and review of literature. J Interv Card Electrophysiol 66, 1165–1175 (2023). https://doi.org/10.1007/s10840-022-01400-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-022-01400-z