Abstract
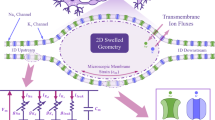
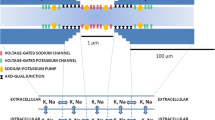
Focal axon swelling refers to localized swelling in axons that may occur because of trauma (e.g., traumatic brain injury) or neurodegenerative diseases (e.g., Alzheimer’s disease). Since the swelling region can be many times larger than its original axon size, many researchers hypothesize that the swelling can alter the action potential (AP) signal. This article discusses the results of a series of newly developed computational studies to elucidate the possible intervention or blockage of AP signals due to swelling in the brain. We argue that the spherical geometry of the swelling site with its enlarged conducting interior causes the entering electric currents to spread evenly over the entire swelled membrane. As such, when the swelled surface becomes larger than the threshold size, the electric current will spread too thin to trigger the AP to spike. In this study, we have used a hybrid membrane model to simulate AP propagation across axons of different radii and swelling radii. We used an integrated model where a cylindrical symmetric 2D model is used to examine the electric current inside a spherical swelling site. In addition, two 1D models are used to capture the current flows along the upstream and downstream stretch before and after the swelling site. The parameters for this model are obtained from literature dedicated to modeling the experimental outcomes of mammal neurons. We observed two factors, which simultaneously affect AP transmission across a swelled axon: a) the axon radius and b) the ratio of the swelled and unswelled axon radii. In general, a thicker axon needs a smaller swelling size and axon ratio to block AP transmission. On the other hand, a thinner axon will reach the threshold at a larger swelling size and axon ratio. When only swelling size is considered, then thinner axons will block AP transmission at a smaller swelling radius. The AP transmission delay inside the swelled region determines whether the AP transmits forward or not. Notably, the blockage is worse if the AP fires at a high frequency. An increase in the charging and reset time due to swelling appears to be the main reason for the variation in axonal response.







Similar content being viewed by others
References
Nikić, I., Merkler, D., Sorbara, C., Brinkoetter, M., Kreutzfeldt, M., Bareyre, F. M., Brück, W., Bishop, D., Misgeld, T., & Kerschensteiner, M. (2011). A reversible form of axon damage in experimental autoimmune encephalomyelitis and multiple sclerosis. Nature Medicine, 17, 495–499.
Tsai, J., Grutzendler, J., Duff, K., & Gan, W.-B. (2004). Fibrillar amyloid deposition leads to local synaptic abnormalities and breakage of neuronal branches. Nature Neuroscience, 7, 1181–1183.
Kolaric, K. V., Thomson, G., Edgar, J. M. & Brown, A. M. (2013) Focal axonal swellings and associated ultrastructural changes attenuate conduction velocity in central nervous system axons: a computer modeling study. Physiological reports 1.
Wang, J., Hamm, R. J., & Povlishock, J. T. (2011). Traumatic axonal injury in the optic nerve: Evidence for axonal swelling, disconnection, dieback, and reorganization. Journal of Neurotrauma, 28, 1185–1198.
MAXWELL, W. L., POVLISHOCK, J. T., & GRAHAM, D. L. (1997). A mechanistic analysis of nondisruptive axonal injury: A review. Journal of Neurotrauma, 14, 419–440.
Tang-Schomer, M. D., Johnson, V. E., Baas, P. W., Stewart, W., & Smith, D. H. (2012). Partial interruption of axonal transport due to microtubule breakage accounts for the formation of periodic varicosities after traumatic axonal injury. Experimental Neurology, 233, 364–372.
Tang-Schomer, M. D., Patel, A. R., Baas, P. W., & Smith, D. H. (2010). Mechanical breaking of microtubules in axons during dynamic stretch injury underlies delayed elasticity, microtubule disassembly, and axon degeneration. The FASEB Journal, 24, 1401–1410.
Smith, D. H., Wolf, J. A., Lusardi, T. A., Lee, V. M.-Y., & Meaney, D. F. (1999). High tolerance and delayed elastic response of cultured axons to dynamic stretch injury. Journal of Neuroscience, 19, 4263–4269.
Wu, Y.-T., & Adnan, A. (2018). Damage and failure of axonal microtubule under extreme high strain rate: An in-Silico molecular dynamics study. Scientific Reports, 8, 12260.
Wu, Y.-T., & Adnan, A. (2017). Effect of shock-induced cavitation bubble collapse on the damage in the simulated Perineuronal net of the brain. Scientific Reports, 7, 5323.
Krstic, D., & Knuesel, I. (2013). Deciphering the mechanism underlying late-onset Alzheimer disease. Nature Reviews Neurology, 9, 25–34.
Trapp, B. D., Peterson, J., Ransohoff, R. M., Rudick, R., Mörk, S., & Bö, L. (1998). Axonal transection in the lesions of multiple sclerosis. New England Journal of Medicine, 338, 278–285.
Magdesian, M. H., Sanchez, F. S., Lopez, M., Thostrup, P., Durisic, N., Belkaid, W., Liazoghli, D., Grütter, P., & Colman, D. R. (2012). Atomic force microscopy reveals important differences in axonal resistance to injury. Biophysical Journal, 103, 405–414.
Maia, P. D., & Kutz, J. N. (2014). Identifying critical regions for spike propagation in axon segments. Journal of Computational Neuroscience, 36, 141–155.
Goldstein, S. S., & Rall, W. (1974). Changes of action potential shape and velocity for changing core conductor geometry. Biophysical Journal, 14, 731–757.
Ofer, N., & Shefi, O. (2016). Axonal geometry as a tool for modulating firing patterns. Applied Mathematical Modelling, 40, 3175–3184.
Rall, W. (1995) The theoretical foundation of dendritic function: selected papers of Wilfrid Rall with commentaries. (MIT press).
Khodorov, B., & Timin, E. (1976). Nerve impulse propagation along nonuniform fibres:(investigations using mathematical models). Progress in Biophysics and Molecular Biology, 30, 145–184.
Tekieh, T., Shahzadi, S., Rafii-Tabar, H., & Sasanpour, P. (2016). Are deformed neurons electrophysiologically altered? A simulation study. Current Applied Physics, 16, 1413–1417.
Pods, J., Schönke, J., & Bastian, P. (2013). Electrodiffusion models of neurons and extracellular space using the Poisson-Nernst-Planck equations—Numerical simulation of the intra-and extracellular potential for an axon model. Biophysical Journal, 105, 242–254.
Pospischil, M., Toledo-Rodriguez, M., Monier, C., Piwkowska, Z., Bal, T., Frégnac, Y., Markram, H., & Destexhe, A. (2008). Minimal Hodgkin–Huxley type models for different classes of cortical and thalamic neurons. Biological Cybernetics, 99, 427–441.
Tahayori, B., et al. (2014). Modelling extracellular electrical stimulation: IV. Effect of the cellular composition of neural tissue on its spatio-temporal filtering properties. Journal of neural engineering, 11, 065005.
Elia, S., Lamberti, P. & Tucci, V. (n.d.) in COMSOL Europe Conference. 1–7.
Xylouris, K., Queisser, G., & Wittum, G. (2010). A three-dimensional mathematical model of active signal processing in axons. Computing and Visualization in Science, 13, 409–418.
Izhikevich, E. M. (2004). Which model to use for cortical spiking neurons? IEEE Transactions on Neural Networks, 15, 1063–1070.
Ermentrout, G. B., & Rinzel, J. (1996). Reflected waves in an inhomogeneous excitable medium. SIAM Journal on Applied Mathematics, 56, 1107–1128.
Wei, Y., Ullah, G., & Schiff, S. J. (2014). Unification of neuronal spikes, seizures, and spreading depression. Journal of Neuroscience, 34, 11733–11743.
Dijkstra, K., Hofmeijer, J., van Gils, S. A., & van Putten, M. J. (2016). A biophysical model for cytotoxic cell swelling. Journal of Neuroscience, 36, 11881–11890.
Acknowledgments
This work has been funded by the Computational Cellular Biology of Blast (C2B2) program through the Office of Naval Research (ONR) (Award # N00014-18-1-2082- Dr. Timothy Bentley, Program Manager). The authors gratefully acknowledge the Texas Advanced Computing Center (TACC) at The University of Texas at Austin for providing HPC resources that contributed to the research results reported in this paper. URL: http://www.tacc.utexas.edu.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The Authors have no Conflict of Interest in this work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 688 kb)
Rights and permissions
About this article
Cite this article
Wu, YT., Gilpin, K. & Adnan, A. Effects of Focal Axonal Swelling Level on the Action Potential Signal Transmission. J Comput Neurosci 48, 253–263 (2020). https://doi.org/10.1007/s10827-020-00750-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-020-00750-9