Abstract
Purpose
To study the role of Toll-like receptor 4 (TLR4) in human spermatozoa and to assess sperm parameters, oxidative stress markers, and acrosome reaction in response to the stimulation of TLR4 by its ligand, the lipopolysaccharide (LPS), as a major endotoxin of Gram-negative bacteria.
Methods
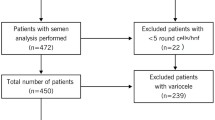
Our study was carried out in 73 sperm samples from patients undergoing semen analysis for couple infertility investigations. The studied patients were divided into three groups: normozoospermic fertile patients (n = 13), patients with abnormal and leukospermic semen (n = 13), and patients with abnormal and non-leukospermic semen (n = 47). TLR4 expression in human spermatozoa was initially analyzed by western blot. Sperm samples were incubated in the presence of LPS (200 ng/ml) for 18 h. Then, sperm motility and vitality were evaluated by microscopic observation and oxidative stress markers as malondialdehyde (MDA) and carbonyl groups (CG) were spectrophotometrically assessed in neat and selected sperm. A triple-stain technique was also performed to evaluate acrosome reaction in 15 sperm samples from infertile patients.
Results
TLR4 expression was confirmed in human spermatozoa with a molecular weight of 69 kDa. In the normozoospermic group, no significant differences in sperm parameters and oxidative stress markers were shown after incubation with LPS in neat and selected sperms. Regarding samples from the non-leukospermic group, LPS reduced spermatozoa motility and vitality rates in selected sperm (P = 0.003; P = 0.004, respectively). A significant increase of MDA and CG levels was also detected (P = 0.01; P = 0.02, respectively). However, only the MDA levels were significantly increased (P = 0.01) in neat LPS-stimulated sperm. The same results were shown within the leukospermic group. The comparison between the two groups, leukospermic and non-leukospermic, in selected sperms showed a more important LPS effect in the leukospermic group significantly on motility and MDA rates (P = 0.006; P = 0.009, respectively). Furthermore, a significant decrease in reacted spermatozoa rate was detected in response to LPS in selected sperm samples from infertile men (P = 0.03).
Conclusions
These findings indicate that human spermatozoa express TLR4 and respond to LPS stimulation with alterations in viability, motility, and the acrosome reaction implicating reactive oxygen species (ROS) production in sperm samples from infertile patients.






Similar content being viewed by others
References
Carlsen E, Andersson AM, Petersen JH, Skakkebaek NE. History of febrile illness and variation in semen quality. Hum Reprod. 2003;18:2089–2.
Nariculam J, Minhas S, Adeniyi A, Ralph DJ, Freeman A. Review of the efficacity of surgical treatment for and pathological changes in patients with chronic scrotal pain. BJU Int. 2007;99:1091–3.
Brecchia G, Cardinali R, Mourvaki E, Collodel G, Moretti E, Dal Bosco A, et al. Short- and long-term effects of lipopolysaccharide induced inflammation on rabbit sperm quality. Anim Reprod Sci. 2010;118:310–6.
Metukuri MR, Reddy CM, Reddy PR, Reddanna P. Bacterial LPS-mediated acute inflammation-induced spermatogenic failure in rats: role of stress response proteins and mitochondrial dysfunction. Inflammation. 2010;33:235–3.
Winnal WR, Hedger MP. Differential responses of epithelial Sertoli cells of the rat testis to toll-like receptor 2 and 4 ligands: implications for studies of testicular inflammation using bacterial lipopolysaccharides. J Innate Immun. 2011;17:123–36.
Aly HA, Lightfoot DA, El-Shemy HA. Bacterial lipopolysaccharide-induced oxidative stress in adult rat Sertoli cells in vitro. Toxicol in Vitro. 2010;24:1266–72.
Zhang M, Nii T, Isobe N, Yoshimura Y. Expression of Toll-like receptors and effects of lipopolysaccharide on the expression of proinflammatory cytokines and chemokine in the testis and epididymis of roosters. Poult Sci. 2012;91:1997–2003.
Henkel R, Kierspel E, Stalf T, Mehnert C, Menkveld R, Tinneberg HR, et al. Effect of reactive oxygen species produced by spermatozoa and leukocytes on sperm functions in non-leukocytospermic patients. Fertil Steril. 2005;83:635–42.
Tremellen K. Oxidative stress and male infertility—a clinical perspective. Hum Reprod Update. 2008;14:243–58.
Aitken RJ, Koppers AJ. Apoptosis and DNA damage in human spermatozoa. Asian J Androl. 2011;13:36–42.
Yun SB, Jee HL, Soo HC, Sunah K, Felicidad A, Joseph LW, et al. Macrophages generate reactive oxygen species in response to minimally oxidized LDL: TLR4- and Syk dependent activation of Nox2. Circ Res. 2009;104:210–8.
Park HS, Jung HY, Park EY, Kim J, Lee WJ, Bae YS. Cutting edge: direct interaction of TLR4 with NADPH oxidase 4 isozyme is essential for lipopolysaccharide-induced production of reactive oxygen species and activation of NF-kappa B. J Immunol. 2004;173:3589–93.
Mark P. Toll-like receptors and signaling in spermatogenesis and responses to inflammation-aperspective. J Reprod Immunol. 2011;88:130–41.
Shoulong D, Kun Y, Baolu Z, Yuchang Y, Zhixian W, Jinlong Z, et al. Toll-like receptor 4 promotes NO synthesis by upregulating GCHI expression under oxidative stress conditions in sheep monocytes/macrophages. Oxidative Med Cell Longev. 2015;2015:359315.
Beutler BA. TLRs and innate immunity. Blood. 2009;113:1399–407.
Chang ZL. Important aspects of Toll-like receptors, ligands and their signaling pathways. Inflamm Res. 2010;59:791–808.
Nishimura M, Naito S. Tissue-specific Mrna expression profiles of human toll-like receptors and related genes. Biol Pharm Bull. 2005;28:886–92.
Saeidi S, Shapouri F, Amirchaghmaghi E, Hoseinifar H, Sabbaghian M, SadighiGilani MA, et al. Sperm protection in the male reproductive tract by Toll-like. Recept Androl. 2014;46:784–90.
Palladino MA, Johnson TA, Gupta R, Chapman JL, Ojha P. Members of the Toll-like receptor family of innate immunity pattern-recognition receptors are abundant in the male rat reproductive tract. Biol Reprod. 2007;76:958–64.
Palladino MA, Savarese MA, Chapman JL, Dughi MK, Plaska D. Localization of Toll-like receptors on epididymal epithelial cells and spermatozoa. Am J Reprod Immunol. 2008;60:541–55.
Bhushan S, Tchatalbachev S, Klug J, Fijak M, Pineau C, Chakraborty T, et al. Uropathogenic Escherichia coli block MyD88- dependent and activate MyD88 independent signaling pathways in rat testicular cells. J Immunol. 2008;180:5537–47.
Fujita Y, Mihara T, Okazaki T, Shitanaka M, Kushino R, Ikeda C, et al. Toll-like receptors (TLR) 2 and 4 on human sperm recognize bacterial endotoxins and mediate apoptosis. Hum Reprod. 2011;26:2799–806.
Hagan S, Khurana N, Chandra S, Abdel-Mageed AB, Mondal D, Hellstrom WJG, et al. Differential expression of novel biomarkers (TLR-2, TLR-4, COX-2, and Nrf-2) of inflammation and oxidative stress in semen of leukocytospermia patients. Andrology. 2015;3:848–55.
Aitken RJ, Fisher HM, Fulton N, Gomez E, Knox W, Lewis B, et al. Reactive oxygen species generation by human spermatozoa is induced by exogenous NADPH and inhibited by the flavoprotein inhibitors diphenyleneiodonium and quinacrine. Mol Reprod Dev. 1997;47:468–82.
Bánfi B, Molnár G, Maturana A, Steger K, Hegedûs B, Demaurex N, et al. A Ca (2+)-activated NADPH oxidase in testis, spleen, and lymph nodes. J Biol Chem. 2001;276:37594–601.
Musset B, Clark RA, DeCoursey TE, Petheo GL, Geiszt M, Chen Y, et al. NOX5 in human spermatozoa: expression, function, and regulation. J Biol Chem. 2012;287:9376–88.
Gomez E, Buckingham D, Brindle J, Lanzafame F, Irvine DS, Aitken RJ. Development of an image analysis system to monitor the retention of residual cytoplasm by human spermatozoa: correlation with biochemical markers of the cytoplasmic space, oxidative stress and sperm function. J Androl. 1996;17:276–87.
Lewis SE, Donnelly ET, Sterling ES, Kennedy MS, Thompson W, et al. Nitric oxide synthase and nitrite production in human spermatozoa: evidence that endogenous nitric oxide is beneficial to sperm motility. Mol Hum Reprod. 1996;2:873–8.
World Health Organization. Laboratory manual for the examination and processing of human semen. 5th ed. Geneva: WHO; 2010.
Chandrasekhar A, Laloraya M, Kumar PG. Modulation of nicotinamide adenine dinucleotide phosphate oxidase activity through sequential post translational modifications of p22 phagocytic oxidase during capacitation and acrosome reaction in goat spermatozoa. J Anim Sci. 2011;89:2995–3007.
Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54.
Buege J, Aust S. Microsomal lipid peroxidation. Methods Enzymol. 1978;52:302–10.
Reznick AZ, Packer L. Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Methods Enzymol. 1994;233:357–63.
Kitiyanant Y, Chaisalee B, Pavasuthipaisit K. Evaluation of the acrosome reaction and viability in buffalo spermatozoa using two staining methods: the effects of heparin and calcium ionophore A23187. Int J Androl. 2002;25:215–22.
Talbot P, Chacon RS. A triple-stain technique for evaluating normal acrosome reactions of human sperm. J Exp Zool. 1981;215:201–8.
Iwami KI, Matsuguchi T, Masuda A, Kikuchi T, Musikacharoen T, Yoshikai Y. Cutting edge: naturally occurring soluble form of mouse toll-like receptor 4 inhibits lipopolysaccharide signaling. J Immunol. 2000;165:6682–6.
Janardhan KS, McIsaac M, Fowlie J, Shrivastav A, Caldwell S, Sharma RK, et al. Toll like receptor-4 expression in lipopolysaccharide induced lung inflammation. Histol Histopathol. 2006;21:687–96.
Hosseinzadeh S, Pacey AA, Eley A. Chlamydia trachomatis-induced death of human spermatozoa is caused primarily by lipopolysaccharide. J Med Microbiol. 2003;52:193–200.
Zhongyuan L, Dahu Z, Yuanqiao H, Zhiyong D, Fei M, Tao L, et al. Lipopolysaccharide compromises human sperm function by reducing intracellular cAMP. Tohoku J Exp Med. 2016;238:105–12.
Gow RM, O’Bryan MK, Canny BJ, Ooi GT, Hedger MP. Differential effects of dexamethasone treatment on lipopolysaccharide induced testicular inflammation and reproductive hormone inhibition in adult rats. J Endocrinol. 2001;168:193–201.
Ochsendorf FR, Buhl R, Bastlein A, Beschmann H. Glutathione in spermatozoa and seminal plasma of infertile men. Hum Reprod. 1998;13:353–9.
Jones R, Mann T, Sherins R. Peroxidative breakdown of phospholipids in human spermatozoa, spermicidal properties of fatty acid peroxides, and protective action of seminal plasma. Fertil Steril. 1979;31:531–7.
Fraga CG, Motchnik PA, Shigenaga MK, Helbock HJ, Jacob RA, Ames BN. Ascorbic acid protects against endogenous oxidative DNA damage in human sperm. Proc Natl Acad Sci U S A. 1991;88:11003–6.
Urata K, Narahara H, Tanaka Y, Egashira T, Takayama F, Miyakawa. Effect of endotoxin-induced reactive oxygen species on sperm motility. Fertil Steril. 2001;76:163–6.
Iadecola C, Anrather J. The immunology of stroke: from mechanisms to translation. Nat Med. 2011;17:796–808.
Christopher A, Anthony R, Tricia A, Scott H, Joseph E N. LPS-stimulated RAW 264.7 macrophage inducible nitric oxide synthase (iNOS) and nitric oxide production is decreased by an omega-3 fatty acid lipid emulsion. J Surg Res. 2008;149:296–302.
Sa T, Cheryl LS, Elebeoba EM. Investigating the role of TNF-α and IFN-γ activation on the dynamics of iNOS gene expression in LPS stimulated macrophages. PLoS One. 2016;11(6):e0153289.
West AP, Brodsky IE, Rahner C, Woo DK, Erdjument-Bromage H, Tempst P, et al. TLR signaling augments macrophage bactericidal activity through mitochondrial ROS. Nature. 2011;472:476–80.
Cloonan SM, Choi AM. Mitochondria: sensors and mediators of innate immune receptor signaling. Curr Opin Microbiol. 2013;16:327–38.
De Lamirande E, Lamothe G, Villemure M. Control of superoxide and nitric oxide formation during human sperm capacitation. Free Radic Biol Med. 2009;46:1420–7.
Griveau JF, Renard P, Le Lannou D. An in vitro promoting role for hydrogen peroxide in human sperm capacitation. Int J Androl. 1994;17:300–7.
Revelli A, Costamagna C, Moffa F, Aldieri E, Ochetti S, Bosia A, et al. Signaling pathway of nitric oxide-induced acrosome reaction in human spermatozoa. Biol Reprod. 2001;64:1708–12.
Rosselli M, Dubey RK, Imthurn B, Macas E, Keller PJ. Effects of nitric oxide on human spermatozoa: evidence that nitric oxide decreases sperm motility and induces sperm toxicity. Hum Reprod. 1995;10:1786–90.
Acknowledgements
The authors thank all the members of the Higher Institute of Biotechnology, Sfax, and Mrs. Olfa Abida, a member of Autoimmunity and Immunogenetics Research Unit, Habib Bourguiba Hospital, Sfax, who generously contributed to this study. We thank also Mr. Mourad Ben Hmida for his helpful review of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the Institutional Review Board (IRB) of Medicine Faculty of Sfax, Tunisia. Informed written consent was obtained from all patients for being included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Sahnoun, S., Sellami, A., Chakroun, N. et al. Human sperm Toll-like receptor 4 (TLR4) mediates acrosome reaction, oxidative stress markers, and sperm parameters in response to bacterial lipopolysaccharide in infertile men. J Assist Reprod Genet 34, 1067–1077 (2017). https://doi.org/10.1007/s10815-017-0957-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-017-0957-8