Abstract
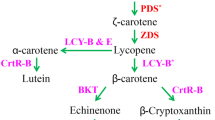
The green alga Haematococcus pluvialis is able to produce and accumulate large amounts of astaxanthin under stress conditions, but not every strain has such a capability. At present, there is little information on how strains differ for astaxanthin production. In this study, three Australian strains of H. pluvialis (New South Wales (NSW), South Australia (SA) and Queensland (QLD)) were cultured and exposed to 0.17 M NaCl for 10 days, to compare their molecular profiles for astaxanthin accumulation and carotenogenesis. After 10 days of salinity stress, the astaxanthin contents of strains NSW, SA and QLD increased up to 16.2, 5.6 and 17.7 mg g−1 dry weight (DW), respectively. Astaxanthin accumulation was more efficient in H. pluvialis QLD, followed by strain NSW and then SA. The transcript abundance of seven carotenogenesis genes (ipi-1, ipi-2, psy, lyc, crtR-B, bkt2 and crtO) was upregulated with different patterns and incremental expression levels amongst the three strains. The early upregulation of the rate-limiting genes, psy, lyc, bkt2, crtR-B and crtO, was more pronounced in the superior astaxanthin-accumulating QLD and NSW strains. Although the increased transcript level of carotenoid genes was not correlated to the different astaxanthin accumulation between the three strains, expression patterns of these genes likely are strain-specific. Overall, H. pluvialis strains QLD and NSW showed good potential to produce high astaxanthin contents, and the former strain displayed a higher productivity upon salinity stress.




Similar content being viewed by others
References
Boussiba S (2000) Carotenogenesis in the green alga Haematococcus pluvialis: cellular physiology and stress response. Physiol Plant 108:111–117
Borowitzka MA, Huisman JM, Osborn A (1991) Culture of the astaxanthin-producing green alga Haematococcus pluvialis 1. Effects of nutrients on growth and cell type. J Appl Phycol 3:295–304
Borowitzka MA (1992) Comparing carotenogenesis in Dunaliella and Haematococcus: implications for commercial production strategies. In: Villa TG, Abalde J (eds) Profiles on biotechnology. Universidade de Santiago de Compostela, Santiago de Compostela, pp 301–310
Ebrahimian A, Kariminia H-R, Vosoughi M (2014) Lipid production in mixotrophic cultivation of Chlorella vulgaris in a mixture of primary and secondary municipal wastewater. Renew Energ 71:502–508
Fanning KJ, Martin I, Wong L, Keating V, Pun S, O’Hare T (2010) Screening sweet corn for enhanced zeaxanthin concentration. J Sci Food Agr 90:91–96
Fu W, Magnúsdóttir M, Brynjólfson S, Palsson BO, Paglia G (2012) UPLC-UV-MSE analysis for quantification and identification of major carotenoid and chlorophyll species in algae. Anal Bioanal Chem 404:3145–3154
Gao Z, Meng C, Zhang X, Xu D, Miao X, Wang Y, Yang L, Lv H, Chen L, Ye N (2012) Induction of salicylic acid (SA) on transcriptional expression of eight carotenoid genes and astaxanthin accumulation in Haematococcus pluvialis. Enzyme Microb Tech 51:225–230
Grünewald K, Eckert M, Hirschberg J, Hagen C (2000) Phytoene desaturase is localized exclusively in the chloroplast and up-regulated at the mRNA level during accumulation of secondary carotenoids in Haematococcus pluvialis (Volvocales, Chlorophyceae). Plant Physiol 122:1261–1268
Han D, Li YT, Hu Q (2013) Astaxanthin in microalgae: pathways, functions and biotechnological implications. Algae 28:131–147
Harker M, Tsavalos AJ, Young AJ (1996) Factors responsible for astaxanthin formation in the chlorophyte Haematococcus pluvialis. Bioresource Technol 55:207–214
Huang J-C, Chen F, Sandmann G (2006) Stress-related differential expression of multiple β-carotene ketolase genes in the unicellular green alga Haematococcus pluvialis. J Biotech 122:176–185
Jin E, Lee CG, Polle JEW (2006) Secondary carotenoid accumulation in Haematococcus (Chlorophyceae): biosynthesis, regulation, and biotechnology. J Microbiol Biotechnol 16:821–831
Kamath BS, Srikanta BM, Dharmesh SM, Sarada R, Ravishankar GA (2008) Ulcer preventive and antioxidative properties of astaxanthin from Haematococcus pluvialis. Eur J Pharmacol 590:387–395
Kobayashi M, Kurimura Y, Kakizono T, Nishio N, Tsuji Y (1997) Morphological changes in the life cycle of the green alga Haematococcus pluvialis. J Ferment Bioeng 84:94–97
Li Y, Sommerfeld M, Chen F, Hu Q (2008) Consumption of oxygen by astaxanthin biosynthesis: a protective mechanism against oxidative stress in Haematococcus pluvialis (Chlorophyceae). J Plant Physiol 165:1783–1797
Li Y, Sommerfeld M, Chen F, Hu Q (2010) Effect of photon flux densities on regulation of carotenogenesis and cell viability of Haematococcus pluvialis (Chlorophyceae). J Appl Phycol 22:253–263
Lotan T, Hirschberg J (1995) Cloning and expression in Escherichia coli of the gene encoding β-C-4-oxygenase, that converts β-carotene to the ketocarotenoid canthaxanthin in Haematococcus pluvialis. FEBS Lett 364:125–128
Lu QY, Zhang Y, Wang Y, Wang D, Lee RP, Gao K, Byns R, Heber D (2009) California Hass avocado: profiling of carotenoids, tocopherol, fatty acid, and fat content during maturation and from different growing areas. J Agric Food Chem 57:10408–10413
Sarada R, Bhattacharya S, Bhattacharya S, Ravishankar GA (2002a) A response surface approach for the production of natural pigment astaxanthin from green alga, Haematococcus pluvialis: effect of sodium acetate, culture age, and sodium chloride. Food Biotechnol 16:107–120
Sarada R, Tripathi U, Ravishankar GA (2002b) Influence of stress on astaxanthin production in Haematococcus pluvialis grown under different culture conditions. Process Biochem 37:623–627
Steinbrenner J, Linden H (2001) Regulation of two carotenoid biosynthesis genes coding for phytoene synthase and carotenoid hydroxylase during stress-induced astaxanthin formation in the green alga Haematococcus pluvialis. Plant Physiol 125:810–817
Sun Z, Cunningham FX, Gantt E (1998) Differential expression of two isopentenyl pyrophosphate isomerases and enhanced carotenoid accumulation in a unicellular chlorophyte. Proc Natl Acad Sci U S A 95:11482–11488
Vidhyavathi R, Venkatachalam L, Sarada R, Ravishankar GA (2008) Regulation of carotenoid biosynthetic genes expression and carotenoid accumulation in the green alga Haematococcus pluvialis under nutrient stress conditions. J Exp Bot 59:1409–1418
Vidhyavathi R, Sarada R, Ravishankar GA (2009) Expression of carotenogenic genes and carotenoid production in Haematococcus pluvialis under the influence of carotenoid and fatty acid synthesis inhibitors. Enzyme Microb Tech 45:88–93
Acknowledgments
Dr. Zhengquan Gao and Dr. Chunxiao Meng were visiting scholars at The University of Queensland supported by the National Natural Science Foundation of China (41106124, 31170279), the National Natural Science Foundation of Shandong Province (ZR2011DM006, ZR2011CQ010), the open funds of State Key Laboratory of Agricultural Microbiology (AMLKF201003) and the supporting project for young teachers in Shandong University of Technology. The authors are grateful to the James Cook University/MBD Microalgae Research & Development Facility for providing the experimental microalgae strains. This work was financially supported by the James Cook University Miscellaneous Research Fund (20730) and Advanced Manufacturing Cooperative Research Centre (AMCRC) grant (no. 2.3.2).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhengquan Gao and Chunxiao Meng contributed equally to this work
Rights and permissions
About this article
Cite this article
Gao, Z., Meng, C., Chen, Y.C. et al. Comparison of astaxanthin accumulation and biosynthesis gene expression of three Haematococcus pluvialis strains upon salinity stress. J Appl Phycol 27, 1853–1860 (2015). https://doi.org/10.1007/s10811-014-0491-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-014-0491-3