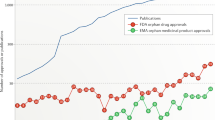
Abstract
Drug studies on Rett syndrome (RTT) have drastically increased over the past few decades. This review aims to provide master data on bench-to-bedside drug studies involving RTT. A comprehensive literature review was performed by searching in PUBMED, MEDLINE and Google Scholar, international, national and regional clinical trial registries and pharmaceutical companies using the keywords “Rett syndrome treatment and/or drug or compound or molecule”. Seventy drugs were investigated in non-clinical (N = 65 animal/cell line-based studies; N = 5 iPSC-based study) and clinical trials (N = 34) for ameliorating the symptoms of RTT. Though there is good progress in both clinical and non-clinical studies, none of these drugs entered phase III/IV for being launched as a therapeutic agent for RTT.

Similar content being viewed by others
Abbreviations
- RTT:
-
Rett syndrome
- MECP2 :
-
Human methyl CpG binding protein 2 gene
- Mecp2 :
-
Mouse methyl CpG binding protein 2
- MeCP2 :
-
Human methyl CpG binding protein 2
- CNS:
-
Central nervous system
- hiPSC:
-
Human induced pluripotent stem cell
- CDKL5 :
-
Cyclin-dependent kinase-like protein 5 gene
- HDAC :
-
Histone deacetylase
- QoL:
-
Quality of life
- CREB1 :
-
Cyclic adenine mono phosphate responsive element binding protein 1
- TrKB:
-
Tyrosine receptor kinase B
- DHF:
-
Dihydroxyflavone
- SNRI:
-
Selective norepinephrine reuptake inhibitor
- SSRI:
-
Selective serotonin reuptake inhibitor
- GABA:
-
Gamma-amino butyric acid
- PTP1B:
-
Protein tyrosine phosphate 1B
- CPT157633:
-
Difluoromethylphosphonic acid
- UA0713:
-
Ursolic acid derivative
- VPA:
-
Valproic acid
- NMDA:
-
N-methyl-d-aspartate
- KO:
-
Knock out
- BDNF:
-
Brain-derived neurotrophic factor
- 5-HT7R:
-
5-Hydroxytryptamine receptor 7
- BBB:
-
Blood brain barrier
- CNF-1:
-
Cytotoxic necrotizing factor 1
- IGF:
-
Insulin-like growth factor
- rhIGF:
-
Recombinant human insulin-like growth factor
- DCS:
-
d-Cycloserine
- F15599-3-Chloro-4:
-
Fluorophenyl-(4-fluoro-4-{[(5-methylpyrimidin-2-ylmethyl)-amino]-methyl}-piperidin-1-yl)-methanone
- 08-OH-DPAT :
-
8-Hydroxy-n,N-ipropylaminotetralin
- NO-711 :
-
1-[2-[[(diphenylmethylene)imino]oxy]ethyl]-1,2,5,6-Tetrahydro-3-pyridine-carboxylicacid-hydrochloride
References
Abdala, A. P., Dutschmann, M., Bissonnette, J. M., & Paton, J. F. (2010). Correction of respiratory disorders in a mouse model of Rett syndrome. Proceedings of the National Academy of Sciences of the United States of America. https://doi.org/10.1073/pnas.1012104107.
Abdala, A. P., Lioy, D. T., Garg, S. K., Knopp, S. J., Paton, J. F. R., & Bissonnette, J. M. (2014). Effect of Sarizotan, a 5-HT1a and D2-like receptor agonist, on respiration in three mouse models of Rett syndrome. American Journal of Respiratory Cell and Molecular Biology,50(6), 1031–1039. https://doi.org/10.1165/rcmb.2013-0372OC.
Arai, A., & Kessler, M. (2007). Pharmacology of ampakine modulators: From AMPA receptors to synapses and behavior. Current Drug Targets,8(5), 583–602.
Balachandar, V., Dhivya, V., Gomathi, M., Mohanadevi, S., Venkatesh, B., Geetha, B. (2016). A review of Rett syndrome (RTT) with induced pluripotent stem cells. Stem Cell Investigation, 3, 52. https://doi.org/10.21037/sci.2016.09.05
Ben-Zeev, B., Aharoni, R., Nissenkorn, A., & Arnon, R. (2011). Glatiramer acetate (GA, Copolymer-1) an hypothetical treatment option for Rett syndrome. Medical Hypotheses,76(2), 190–193.
Bharathi, G., Venugopal, A., & Vellingiri, B. (2019). Music therapy as a therapeutic tool in improving the social skills of autistic children. The Egyptian Journal of Neurology, Psychiatry and Neurosurgery,55(1), 44. https://doi.org/10.1186/s41983-019-0091-x.
Biohaven Pharmaceutical Holding Company Ltd. (n.d.). Biohaven Announces Initiation Of Clinical Development For BHV-5000, A Novel Low-Trapping NMDA Antagonist. PR Newswire. 2018. https://www.prnewswire.com/news-releases/biohaven-announces-initiation-of-clinical-development-for-bhv-5000-a-novel-low-trapping-nmda-antagonist-300584389.html (Accessed on Feb 6, 2019). NEW HAVEN, Conn. https://www.prnewswire.com/news-releases/biohaven-announces-initiation-of-clinical-development-for-bhv-5000-a-novel-low-trapping-nmda-antagonist-300584389.html. Accessed 6 Feb 2019
Bittolo, T., Raminelli, C. A., Deiana, C., Baj, G., Vaghi, V., Ferrazzo, S., et al. (2016). Pharmacological treatment with mirtazapine rescues cortical atrophy and respiratory deficits in MeCP2 null mice. Scientific Reports,6, 19796.
Brendel, C., Klahold, E., Gärtner, J., & Huppke, P. (2009). Suppression of nonsense mutations in Rett syndrome by aminoglycoside antibiotics. Pediatric Research,65(5), 520.
Brendel, C., Belakhov, V., Werner, H., Wegener, E., Gärtner, J., Nudelman, I., et al. (2011). Readthrough of nonsense mutations in Rett syndrome: Evaluation of novel aminoglycosides and generation of a new mouse model. Journal of Molecular Medicine,89(4), 389–398.
Buchovecky, C. M., Turley, S. D., Brown, H. M., Kyle, S. M., McDonald, J. G., Liu, B., et al. (2013). A suppressor screen in Mecp2 mutant mice implicates cholesterol metabolism in Rett syndrome. Nature Genetics,45(9), 1013–1020. https://doi.org/10.1038/ng.2714.
Castro, J., Garcia, R. I., Kwok, S., Banerjee, A., Petravicz, J., Woodson, J., et al. (2014). Functional recovery with recombinant human IGF1 treatment in a mouse model of Rett syndrome. Proceedings of the National Academy of Sciences,111(27), 9941.
Chahrour, M., Jung, S. Y., Shaw, C., Zhou, X., Wong, S. T., Qin, J., et al. (2008). MeCP2, a key contributor to neurological disease, activates and represses transcription. Science,320(5880), 1224–1229.
Chapleau, C. A., Lane, J., Pozzo-Miller, L., & Percy, A. K. (2013). Evaluation of current pharmacological treatment options in the management of Rett syndrome: From the present to future therapeutic alternatives. Current Clinical Pharmacology,8(4), 358–369.
Chin, E. W. M., Marcy, G., Yoon, S.-I., Ma, D., Rosales, F. J., Augustine, G. J., et al. (2016). Choline ameliorates disease phenotypes in human iPSC models of Rett syndrome. Neuromolecular Medicine,18(3), 364–377. https://doi.org/10.1007/s12017-016-8421-y.
Chin, E. W. M., Lim, W. M., Ma, D., Rosales, F. J., & Goh, E. L. K. (2019). Choline rescues behavioural deficits in a mouse model of Rett syndrome by modulating neuronal plasticity. Molecular Neurobiology,56(6), 3882–3896. https://doi.org/10.1007/s12035-018-1345-9.
De Filippis, B., Fabbri, A., Simone, D., Canese, R., Ricceri, L., Malchiodi-Albedi, F., et al. (2012). Modulation of RhoGTPases improves the behavioral phenotype and reverses astrocytic deficits in a mouse model of Rett syndrome. Neuropsychopharmacology,37(5), 1152–1163. https://doi.org/10.1038/npp.2011.301.
De Filippis, B., Ricceri, L., Fuso, A., & Laviola, G. (2013). Neonatal exposure to low dose corticosterone persistently modulates hippocampal mineralocorticoid receptor expression and improves locomotor/exploratory behaviour in a mouse model of Rett syndrome. Neuropharmacology,68, 174–183. https://doi.org/10.1016/j.neuropharm.2012.05.048.
De Filippis, B., Nativio, P., Fabbri, A., Ricceri, L., Adriani, W., Lacivita, E., et al. (2014). Pharmacological stimulation of the brain serotonin receptor 7 as a novel therapeutic approach for Rett syndrome. Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology, 39(11), 2506–2518.https://doi.org/10.1038/npp.2014.105
De Filippis, B., Valenti, D., Chiodi, V., Ferrante, A., de Bari, L., Fiorentini, C., et al. (2015a). Modulation of Rho GTPases rescues brain mitochondrial dysfunction, cognitive deficits and aberrant synaptic plasticity in female mice modeling Rett syndrome. European Neuropsychopharmacology: The Journal of the European College of Neuropsychopharmacology,25(6), 889–901. https://doi.org/10.1016/j.euroneuro.2015.03.012.
De Filippis, B., Valenti, D., de Bari, L., De Rasmo, D., Musto, M., Fabbri, A., et al. (2015b). Mitochondrial free radical overproduction due to respiratory chain impairment in the brain of a mouse model of Rett syndrome: Protective effect of CNF1. Free Radical Biology & Medicine,83, 167–177. https://doi.org/10.1016/j.freeradbiomed.2015.02.014.
Delépine, C., Meziane, H., Nectoux, J., Opitz, M., Smith, A. B., Ballatore, C., et al. (2016). Altered microtubule dynamics and vesicular transport in mouse and human MeCP2-deficient astrocytes. Human Molecular Genetics,25(1), 146–157. https://doi.org/10.1093/hmg/ddv464.
Deogracias, R., Yazdani, M., Dekkers, M. P., Guy, J., Ionescu, M. C. S., Vogt, K. E., et al. (2012). Fingolimod, a sphingosine-1 phosphate receptor modulator, increases BDNF levels and improves symptoms of a mouse model of Rett syndrome. Proceedings of the National Academy of Sciences,109(35), 14230–14235.
Djukic, A., Holtzer, R., Shinnar, S., Muzumdar, H., Rose, S. A., Mowrey, W., et al. (2016). Pharmacologic treatment of Rett syndrome with glatiramer acetate. Pediatric Neurology,61, 51–57. https://doi.org/10.1016/j.pediatrneurol.2016.05.010.
Edison Pharmaceuticals. (n.d.). Edison Pharmaceuticals Announces Phase 2 Positive Clinical Results for EPI-743 in Rett Syndrome. PR Newswire. 2014. https://www.prnewswire.com/news-releases/edison-pharmaceuticals-announces-phase-2-positive-clinical-results-for-epi-743-in-rett-syndrome-273691331.html (Accessed on Feb 6, 2019). MOUNTAIN VIEW, California, US. https://www.prnewswire.com/news-releases/edison-pharmaceuticals-announces-phase-2-positive-clinical-results-for-epi-743-in-rett-syndrome-273691331.html. Accessed 4 Jan 2019
Ehinger, Y., Matagne, V., Villard, L., Roux, J.-C. (2018). Rett syndrome from bench to bedside: Recent advances. F1000Research, 7, 398. https://doi.org/10.12688/f1000research.14056.1
El-Khoury, R., Panayotis, N., Matagne, V., Ghata, A., Villard, L., & Roux, J.-C. (2014). GABA and glutamate pathways are spatially and developmentally affected in the brain of Mecp2-deficient mice. PLoS ONE. https://doi.org/10.1371/journal.pone.0092169.
Ellaway, C., & Christodoulou, J. (2001). Rett syndrome: Clinical characteristics and recent genetic advances. Disability and Rehabilitation,23(3–4), 98–106.
Ellaway, C. J., Sholler, G., Leonard, H., & Christodoulou, J. (1999). Prolonged QT interval in Rett syndrome. Archives of Disease in Childhood,80(5), 470–472.
Freilinger, M., Dunkler, D., Lanator, I., Item, C. B., Mühl, A., Fowler, B., et al. (2011). Effects of creatine supplementation in Rett syndrome: A randomized, placebo-controlled trial. Journal of Developmental and Behavioral Pediatrics: JDBP,32(6), 454–460. https://doi.org/10.1097/DBP.0b013e31822177a8.
Gadalla, K. K., Bailey, M. E., Spike, R. C., Ross, P. D., Woodard, K. T., Kalburgi, S. N., et al. (2013). Improved survival and reduced phenotypic severity following AAV9/MECP2 gene transfer to neonatal and juvenile male Mecp2 knockout mice. Molecular Therapy,21(1), 18–30.
Garg, S. K., Lioy, D. T., Cheval, H., McGann, J. C., Bissonnette, J. M., Murtha, M. J., et al. (2013). Systemic delivery of MeCP2 rescues behavioral and cellular deficits in female mouse models of Rett syndrome. Journal of Neuroscience,33(34), 13612–13620.
Giampietro, P. F., Schowalter, D. B., Merchant, S., Campbell, L. R., Swink, T., & Roa, B. B. (2006). Widened clinical spectrum of the Q128P MECP2 mutation in Rett syndrome. Child’s Nervous System: ChNS: Official Journal of the International Society for Pediatric Neurosurgery,22(3), 320–324. https://doi.org/10.1007/s00381-005-1155-z.
Gibson, J. H., Slobedman, B., Kaipananickal, H., Williamson, S. L., Minchenko, D., El-Osta, A., et al. (2010). Downstream targets of methyl CpG binding protein 2 and their abnormal expression in the frontal cortex of the human Rett syndrome brain. BMC Neuroscience,11(1), 53. https://doi.org/10.1186/1471-2202-11-53.
Glaze, D. G., Neul, J. L., Percy, A., Feyma, T., Beisang, A., Yaroshinsky, A., et al. (2017). A double-blind, randomized, placebo-controlled clinical study of trofinetide in the treatment of Rett syndrome. Pediatric Neurology,76, 37–46. https://doi.org/10.1016/j.pediatrneurol.2017.07.002.
Glaze, D. G., Percy, A. K., Motil, K. J., Lane, J. B., Isaacs, J. S., Schultz, R. J., et al. (2009). A study of the treatment of Rett syndrome with folate and betaine. Journal of Child Neurology, 24(5), 551–556. https://doi.org/10.1177/0883073808327827.
Gogliotti, R. G., Senter, R. K., Rook, J. M., Ghoshal, A., Zamorano, R., Malosh, C., et al. (2016). mGlu5 positive allosteric modulation normalizes synaptic plasticity defects and motor phenotypes in a mouse model of Rett syndrome. Human Molecular Genetics,25(10), 1990–2004. https://doi.org/10.1093/hmg/ddw074.
Gogliotti, R. G., Senter, R. K., Fisher, N. M., Adams, J., Zamorano, R., Walker, A. G., et al. (2017). mGlu7 potentiation rescues cognitive, social, and respiratory phenotypes in a mouse model of Rett syndrome. Science Translational Medicine. https://doi.org/10.1126/scitranslmed.aai7459.
Gökben, S., Ardıç, U. A., & Serdaroğlu, G. (2012). Use of buspirone and fluoxetine for breathing problems in Rett syndrome. Pediatric Neurology,46(3), 192–194. https://doi.org/10.1016/j.pediatrneurol.2011.12.003.
Gold, W. A., & Christodoulou, J. (2015). The utility of next-generation sequencing in gene discovery for mutation-negative patients with Rett syndrome. Frontiers in Cellular Neuroscience. https://doi.org/10.3389/fncel.2015.00266.
Goldmann, T., Overlack, N., Möller, F., Belakhov, V., van Wyk, M., Baasov, T., et al. (2012). A comparative evaluation of NB30, NB54 and PTC124 in translational read-through efficacy for treatment of an USH1C nonsense mutation. EMBO Molecular Medicine,4(11), 1186–1199.
Gomathi, M., & Balachandar, V. (2017). Novel therapeutic approaches: Rett syndrome and human induced pluripotent stem cell technology. Stem Cell Investigation,4, 20.
Gonzales, M. L., & LaSalle, J. M. (2010). The role of MeCP2 in brain development and neurodevelopmental disorders. Current Psychiatry Reports,12(2), 127–134.
Goyal, M., O’Riordan, M. A., & Wiznitzer, M. (2004). Effect of topiramate on seizures and respiratory dysrhythmia in Rett syndrome. Journal of Child Neurology,19(8), 588–591. https://doi.org/10.1177/088307380401900804.
Guideri, F., & Acampa, M. (2005). Sudden death and cardiac arrhythmias in Rett syndrome. Pediatric Cardiology,26(1), 111. https://doi.org/10.1007/s00246-004-0701-x.
Guo, W., Tsujimura, K., Otsuka, I., & M., Irie, K., Igarashi, K., Nakashima, K., & Zhao, X., (2014). VPA alleviates neurological deficits and restores gene expression in a mouse model of Rett syndrome. PLoS ONE,9(6), e100215. https://doi.org/10.1371/journal.pone.0100215.
Halbach, N. S., Smeets, E. E., Bierau, J., Keularts, I. M., Plasqui, G., Julu, P. O., et al. (2012). Altered carbon dioxide metabolism and creatine abnormalities in rett syndrome. JIMD Rep. https://doi.org/10.1007/8904_2011_76.
Herrera, J. A., Ward, C. S., Pitcher, M. R., Percy, A. K., Skinner, S., Kaufmann, W. E., et al. (2015). Treatment of cardiac arrhythmias in a mouse model of Rett syndrome with Na+-channel-blocking antiepileptic drugs. Disease Models & Mechanisms,8(4), 363–371. https://doi.org/10.1242/dmm.020131.
Hirofuji, S., Hirofuji, Y., Kato, H., Masuda, K., Yamaza, H., Sato, H., et al. (2018). Mitochondrial dysfunction in dopaminergic neurons differentiated from exfoliated deciduous tooth-derived pulp stem cells of a child with Rett syndrome. Biochemical and Biophysical Research Communications,498(4), 898–904.
Hung, S. S. C., Khan, S., Lo, C. Y., Hewitt, A. W., & Wong, R. C. B. (2017). Drug discovery using induced pluripotent stem cell models of neurodegenerative and ocular diseases. Pharmacology & Therapeutics,177, 32–43. https://doi.org/10.1016/j.pharmthera.2017.02.026.
Janc, O. A., & Müller, M. (2014). The free radical scavenger Trolox dampens neuronal hyperexcitability, reinstates synaptic plasticity, and improves hypoxia tolerance in a mouse model of Rett syndrome. Frontiers in Cellular Neuroscience,8, 56. https://doi.org/10.3389/fncel.2014.00056.
Janc, O. A., Hüser, M. A., Dietrich, K., Kempkes, B., Menzfeld, C., Hülsmann, S., et al. (2016). Systemic radical scavenger treatment of a mouse model of Rett syndrome: Merits and limitations of the vitamin E derivative trolox. Frontiers in Cellular Neuroscience,10, 266. https://doi.org/10.3389/fncel.2016.00266.
Jefferson, A., Leonard, H., Siafarikas, A., Woodhead, H., Fyfe, S., Ward, L. M., et al. (2016). Clinical guidelines for management of bone health in Rett syndrome based on expert consensus and available evidence. PLoS ONE,11(2), e0146824. https://doi.org/10.1371/journal.pone.0146824.
Jin, X., Zhong, W., & Jiang, C. (2013). Time-dependent modulation of GABA(A)-ergic synaptic transmission by allopregnanolone in locus coeruleus neurons of Mecp2-null mice. American Journal of Physiology; Cell Physiology, 305(11), C1151–1160. https://doi.org/10.1152/ajpcell.00195.2013
Johnson, R. A., Lam, M., Punzo, A. M., Li, H., Lin, B. R., Ye, K., et al. (2011). 7, 8-dihydroxyflavone exhibits therapeutic efficacy in a mouse model of Rett syndrome. Journal of Applied Physiology,112(5), 704–710.
Katz, D. M., Berger-Sweeney, J. E., Eubanks, J. H., Justice, M. J., Neul, J. L., Pozzo-Miller, L., et al. (2012). Preclinical research in Rett syndrome: Setting the foundation for translational success. Disease Models & Mechanisms,5(6), 733–745. https://doi.org/10.1242/dmm.011007.
Katz, D. M., Bird, A., Coenraads, M., Gray, S. J., Menon, D. U., Philpot, B. D., et al. (2016). Rett syndrome: Crossing the threshold to clinical translation. Trends in Neurosciences,39(2), 100–113. https://doi.org/10.1016/j.tins.2015.12.008.
Khwaja, O. S., Ho, E., Barnes, K. V., O’Leary, H. M., Pereira, L. M., Finkelstein, Y., et al. (2014). Safety, pharmacokinetics, and preliminary assessment of efficacy of mecasermin (recombinant human IGF-1) for the treatment of Rett syndrome. Proceedings of the National Academy of Sciences of the United States of America,111(12), 4596–4601. https://doi.org/10.1073/pnas.1311141111.
Kolb-Kokocinski, A., Mehrle, A., Bechtel, S., Simpson, J. C., Kioschis, P., Wiemann, S., et al. (2006). The systematic functional characterisation of Xq28 genes prioritises candidate disease genes. BMC Genomics,7(1), 29. https://doi.org/10.1186/1471-2164-7-29.
Kozikowski, A. P., Shen, S., Pardo, M., Tavares, M. T., Szarics, D., Benoy, V., et al. (2019). Brain penetrable histone deacetylase 6 inhibitor SW-100 ameliorates memory and learning impairments in a mouse model of fragile X syndrome. ACS Chemical Neuroscience, 10(3), 1679–1695. https://doi.org/10.1021/acschemneuro.8b00600.
Krishnan, N., Krishnan, K., Connors, C. R., Choy, M. S., Page, R., Peti, W., et al. (2015). PTP1B inhibition suggests a therapeutic strategy for Rett syndrome. The Journal of Clinical Investigation,125(8), 3163–3177. https://doi.org/10.1172/JCI80323.
Kron, M., Howell, C. J., Adams, I. T., Ransbottom, M., Christian, D., Ogier, M., & Katz, D. M. (2012). Brain activity mapping in Mecp2 mutant mice reveals functional deficits in forebrain circuits, including key nodes in the default mode network, that are reversed with ketamine treatment. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 32(40), 13860–13872. https://doi.org/10.1523/JNEUROSCI.2159-12.2012.
Kron, M., Lang, M., Adams, I. T., Sceniak, M., Longo, F., & Katz, D. M. (2014). A BDNF loop-domain mimetic acutely reverses spontaneous apneas and respiratory abnormalities during behavioral arousal in a mouse model of Rett syndrome. Disease Models & Mechanisms,7(9), 1047–1055.
Kumandas, S., Caksen, H., Ciftçi, A., Oztürk, M., & Per, H. (2001). Lamotrigine in two cases of Rett syndrome. Brain & Development,23(4), 240–242.
Lallar, M., Rai, A., Srivastava, P., Mandal, K., Gupta, N., Kabra, M., et al. (2018). Molecular testing of MECP2 gene in Rett syndrome phenotypes in Indian girls. Indian Pediatrics,55(6), 474–477. https://doi.org/10.1007/s13312-018-1336-y.
Landucci, E., Brindisi, M., Bianciardi, L., Catania, L. M., Daga, S., Croci, S., et al. (2018). iPSC-derived neurons profiling reveals GABAergic circuit disruption and acetylated α-tubulin defect which improves after iHDAC6 treatment in Rett syndrome. Experimental Cell Research,368(2), 225–235. https://doi.org/10.1016/j.yexcr.2018.05.001.
Lane, J. B., Salter, A. R., Jones, N. E., Cutter, G., Horrigan, J., Skinner, S. A., et al. (2017). Assessment of caregiver inventory for Rett syndrome. Journal of Autism and Developmental Disorders,47(4), 1102–1112. https://doi.org/10.1007/s10803-017-3034-3.
Levitt, E. S., Hunnicutt, B. J., Knopp, S. J., Williams, J. T., & Bissonnette, J. M. (2013). A selective 5-HT 1a receptor agonist improves respiration in a mouse model of Rett syndrome. Journal of Applied Physiology,115(11), 1626–1633. https://doi.org/10.1152/japplphysiol.00889.2013.
Li, W., Bellot-Saez, A., Phillips, M. L., Yang, T., Longo, F. M., & Pozzo-Miller, L. (2017). A small-molecule TrkB ligand restores hippocampal synaptic plasticity and object location memory in Rett syndrome mice. Disease Models & Mechanisms,10(7), 837–845.
Liebhaber, G. M., Riemann, E., & Baumeister, F. A. M. (2003). Ketogenic diet in Rett syndrome. Journal of Child Neurology,18(1), 74–75. https://doi.org/10.1177/08830738030180011801.
Liyanage, V. R. B., & Rastegar, M. (2014). Rett syndrome and MeCP2. Neuromolecular Medicine,16(2), 231–264. https://doi.org/10.1007/s12017-014-8295-9.
Lyst, M. J., & Bird, A. (2015). Rett syndrome: A complex disorder with simple roots. Nature Reviews Genetics,16(5), 261.
Mancini, J., Dubus, J.-C., Jouve, E., Roux, J.-C., Franco, P., Lagrue, E., et al. (2018). Effect of desipramine on patients with breathing disorders in RETT syndrome. Annals of Clinical and Translational Neurology,5(2), 118–127. https://doi.org/10.1002/acn3.468.
Marchetto, M. C. N., Carromeu, C., Acab, A., Yu, D., Yeo, G., & Mu, Y. (2010). A model for neural development and treatment of Rett syndrome using human induced pluripotent stem cells. Cell. https://doi.org/10.1016/j.cell.2010.10.016.
Marschik, P. B., Einspieler, C., Oberle, A., Laccone, F., & Prechtl, H. F. R. (2009). Case report: Retracing atypical development: A preserved speech variant of Rett syndrome. Journal of Autism and Developmental Disorders,39(6), 958–961. https://doi.org/10.1007/s10803-009-0703-x.
Martínez, A. R., Turon, M., Callejón-Póo, L., Sole, E., Armstrong, J., & Pineda, M. (2013). Treatment response in behaviour disorders in Rett syndrome. Journal of Behavioral and Brain Science,03(02), 217–224. https://doi.org/10.4236/jbbs.2013.32023.
Mellios, N., Woodson, J., Garcia, R. I., Crawford, B., Sharma, J., Sheridan, S. D., et al. (2014). β2-Adrenergic receptor agonist ameliorates phenotypes and corrects microRNA-mediated IGF1 deficits in a mouse model of Rett syndrome. Proceedings of the National Academy of Sciences of the United States of America,111(27), 9947–9952. https://doi.org/10.1073/pnas.1309426111.
Mendhekar, D. N., & Duggal, H. S. (2007). Acquired variant of Rett’s disorder and response to lamotrigine. The Journal of Neuropsychiatry and Clinical Neurosciences,19(4), 474–475. https://doi.org/10.1176/jnp.2007.19.4.474.
Mierau, S. B., Patrizi, A., Hensch, T. K., & Fagiolini, M. (2016). Cell-specific regulation of N-methyl-D-aspartate receptor maturation by Mecp2 in cortical circuits. Biological Psychiatry,79(9), 746–754. https://doi.org/10.1016/j.biopsych.2015.05.018.
Miranda-Lourenço, C., Duarte, S. T., Palminha, C., Colino-Oliveira, M., Jéssica, R., Gomes, R., et al. (2019). Reestablishment of adenosine levels: A possible strategy for Rett Syndrome. Frontiers in Cellular Neuroscience, Conference Abstract: XVI Meeting of the Portuguese Society for Neuroscience (SPN2019). https://doi.org/10.3389/conf.fncel.2019.01.00046
Na, E. S., De Jesús-Cortés, H., Martinez-Rivera, A., Kabir, Z. D., Wang, J., Ramesh, V., et al. (2017). D-Cycloserine improves synaptic transmission in an animal model of Rett syndrome. PLoS ONE,12(8), e0183026. https://doi.org/10.1371/journal.pone.0183026.
Naegelin, Y., Barde, Y. A., Weber, P., Plecko, B., Datta, A. N., & Kappos, L. (2015). OP24—2321: FINGORETT—An ongoing phase I clinical study to assess safety and efficacy of oral fingolimod (FTY720) in children with Rett syndrome. European Journal of Paediatric Neurology,19, S8. https://doi.org/10.1016/S1090-3798(15)30025-8.
Nag, N., & Berger-Sweeney, J. E. (2007). Postnatal dietary choline supplementation alters behavior in a mouse model of Rett syndrome. Neurobiology of Disease,26(2), 473–480. https://doi.org/10.1016/j.nbd.2007.02.003.
Nag, N., Mellott, T. J., & Berger-Sweeney, J. E. (2008). Effects of postnatal dietary choline supplementation on motor regional brain volume and growth factor expression in a mouse model of Rett syndrome. Brain Research,1237, 101–109. https://doi.org/10.1016/j.brainres.2008.08.042.
Nance, E., Kambhampati, S. P., Smith, E. S., Zhang, Z., Zhang, F., Singh, S., et al. (2017). Dendrimer-mediated delivery of N-acetyl cysteine to microglia in a mouse model of Rett syndrome. Journal of Neuroinflammation,14(1), 252. https://doi.org/10.1186/s12974-017-1004-5.
Nelson, E. D., Kavalali, E. T., & Monteggia, L. M. (2006). MeCP2-dependent transcriptional repression regulates excitatory neurotransmission. Current Biology,16(7), 710–716. https://doi.org/10.1016/j.cub.2006.02.062.
Nissenkorn, A., Kidon, M., & Ben-Zeev, B. (2017). A potential life-threatening reaction to glatiramer acetate in Rett syndrome. Pediatric Neurology,68, 40–43. https://doi.org/10.1016/j.pediatrneurol.2016.11.006.
O’Leary, H. M., Kaufmann, W. E., Barnes, K. V., Rakesh, K., Kapur, K., Tarquinio, D. C., et al. (2018). Placebo-controlled crossover assessment of mecasermin for the treatment of Rett syndrome. Annals of Clinical and Translational Neurology,5(3), 323–332. https://doi.org/10.1002/acn3.533.
Ogier, M., Wang, H., Hong, E., Wang, Q., Greenberg, M. E., & Katz, D. M. (2007). Brain-derived neurotrophic factor expression and respiratory function improve after ampakine treatment in a mouse model of Rett syndrome. Journal of Neuroscience,27(40), 10912–10917.
Ohya, T., Yamashita, Y., & Matsuishi, T. (2005). [Sudden death in Rett syndrome]. Nihon Rinsho. Japanese Journal of Clinical Medicine, 63(7), 1178–1182.
Panighini, A., Duranti, E., Santini, F., Maffei, M., Pizzorusso, T., Funel, N., et al. (2013). Vascular dysfunction in a mouse model of Rett syndrome and effects of curcumin treatment. PLoS ONE,8(5), e64863. https://doi.org/10.1371/journal.pone.0064863.
Park, M. J., Aja, S., Li, Q., Degano, A. L., Penati, J., Zhuo, J., et al. (2014). Anaplerotic triheptanoin diet enhances mitochondrial substrate use to remodel the metabolome and improve lifespan, motor function, and sociability in MeCP2-null mice. PLoS ONE,9(10), e109527. https://doi.org/10.1371/journal.pone.0109527.
Patrizi, A., Picard, N., Simon, A. J., Gunner, G., Centofante, E., Andrews, N. A., et al. (2016). Chronic administration of the N-methyl-D-aspartate receptor antagonist ketamine improves Rett syndrome phenotype. Biological Psychiatry,79(9), 755–764. https://doi.org/10.1016/j.biopsych.2015.08.018.
Pecorelli, A., Cervellati, C., Hayek, J., & Valacchi, G. (2017). Modulation of oxidative stress response in neurodevelopment disorders. The case of the Rett syndrome variants: MECP2 and CDKL5. Free Radical Biology and Medicine,108, S43.
Percy, A., Glaze, D. G., Neul, J. L., Kaufmann, W. E., Berry-Kravis, E., Condon, S., et al. (2017). 1.49 Trofinetide, a novel IGF-1 related treatment for neurodevelopmental disorders, demonstrates efficacy for children and adolescents with Rett syndrome. Journal of the American Academy of Child & Adolescent Psychiatry, 56(10), S168–S169. https://doi.org/10.1016/j.jaac.2017.09.063
Percy, A. K., Glaze, D. G., Schultz, R. J., Zoghbi, H. Y., Williamson, D., Frost, J. D., et al. (1994). Rett syndrome: Controlled study of an oral opiate antagonist, naltrexone. Annals of Neurology,35(4), 464–470. https://doi.org/10.1002/ana.410350415.
Pini, G., Congiu, L., Benincasa, A., DiMarco, P., Bigoni, S., Dyer, A. H., et al. (2016). Illness severity, social and cognitive ability, and EEG analysis of ten patients with Rett syndrome treated with mecasermin (recombinant human IGF-1). Autism Research and Treatment,2016, 1–9. https://doi.org/10.1155/2016/5073078.
Pini, G., Scusa, M. F., Benincasa, A., Bottiglioni, I., Congiu, L., Vadhatpour, C., et al. (2014). Repeated insulin-like growth factor 1 treatment in a patient with rett syndrome: A single case study. Frontiers in Pediatrics,2, 52. https://doi.org/10.3389/fped.2014.00052.
Pini, G., Scusa, M. F., Congiu, L., Benincasa, A., Morescalchi, P., Bottiglioni, I., et al. (2012). IGF1 as a potential treatment for Rett syndrome: Safety assessment in six Rett patients. Autism Research and Treatment,2012, 1–14. https://doi.org/10.1155/2012/679801.
Pitcher, M. R., Herrera, J. A., Buffington, S. A., Kochukov, M. Y., Merritt, J. K., Fisher, A. R., et al. (2015). Rett syndrome like phenotypes in the R255X Mecp2 mutant mouse are rescued by MECP2 transgene. Human Molecular Genetics,24(9), 2662–2672.
Pitcher, M. R., Ward, C. S., Arvide, E. M., Chapleau, C. A., Pozzo-Miller, L., Hoeflich, A., et al. (2013). Insulinotropic treatments exacerbate metabolic syndrome in mice lacking MeCP2 function. Human Molecular Genetics,22(13), 2626–2633.
Popescu, A. C., Sidorova, E., Zhang, G., & Eubanks, J. H. (2010). Aminoglycoside-mediated partial suppression of MECP2 nonsense mutations responsible for Rett syndrome in vitro. Journal of Neuroscience Research,88(11), 2316–2324.
Reichow, B., George-Puskar, A., Lutz, T., Smith, I. C., & Volkmar, F. R. (2015). Brief report: Systematic review of Rett syndrome in males. Journal of Autism and Developmental Disorders,45(10), 3377–3383. https://doi.org/10.1007/s10803-015-2519-1.
Rett, A. (1966). [On a unusual brain atrophy syndrome in hyperammonemia in childhood]. Wiener Medizinische Wochenschrift (1946), 116(37), 723–726.
Ricceri, L., De Filippis, B., Fuso, A., & Laviola, G. (2011). Cholinergic hypofunction in MeCP2-308 mice: Beneficial neurobehavioural effects of neonatal choline supplementation. Behavioural Brain Research,221(2), 623–629. https://doi.org/10.1016/j.bbr.2011.03.051.
Robinson, L., Guy, J., McKay, L., Brockett, E., Spike, R. C., Selfridge, J., et al. (2012). Morphological and functional reversal of phenotypes in a mouse model of Rett syndrome. Brain: A Journal of Neurology, 135(Pt 9), 2699–2710. https://doi.org/10.1093/brain/aws096
Roux, J.-C., Dura, E., Moncla, A., Mancini, J., & Villard, L. (2007). Treatment with desipramine improves breathing and survival in a mouse model for Rett syndrome. The European Journal of Neuroscience,25(7), 1915–1922. https://doi.org/10.1111/j.1460-9568.2007.05466.x.
Roux, J.-C., Zala, D., Panayotis, N., Borges-Correia, A., Saudou, F., & Villard, L. (2012). Modification of Mecp2 dosage alters axonal transport through the Huntingtin/Hap1 pathway. Neurobiology of Disease,45(2), 786–795.
Schaevitz, L. R., Nicolai, R., Lopez, C. M., D’Iddio, S., Iannoni, E., & Berger-Sweeney, J. E. (2012). Acetyl-L-carnitine improves behavior and dendritic morphology in a mouse model of Rett syndrome. PLoS ONE,7(12), e51586. https://doi.org/10.1371/journal.pone.0051586.
Schmid, D. A., Yang, T., Ogier, M., Adams, I., Mirakhur, Y., Wang, Q., et al. (2012). A TrkB small molecule partial agonist rescues TrkB phosphorylation deficits and improves respiratory function in a mouse model of Rett syndrome. Journal of Neuroscience,32(5), 1803–1810.
Segawa, M. (2001). Pathophysiology of Rett syndrome from the stand point of clinical characteristics. Brain and Development,23, S94–S98.
Shapiro, J. R., Boskey, A. L., Doty, S. B., Lukashova, L., & Blue, M. E. (2017). Zoledronic acid improves bone histomorphometry in a murine model of Rett syndrome. Bone,99, 1–7. https://doi.org/10.1016/j.bone.2017.03.040.
Sheikh, A. (2016). Pre-clinical study of NLX-101 for the treatment of apnoeas in two mouse models of Rett syndrome. Presented at the Pharmacology, London, UK. https://www.pa2online.org/abstracts/vol16issue1abst027p.pdf. Accessed 3 Jan 2019
Sheikh, A. (2017). NLX-101 ameliorates responses to hypercapnia in a mouse model of Rett syndrome. Presented at the Pharmacology 2017, London, UK. https://www.pa2online.org/abstracts/vol18issue1abst139p.pdf. Accessed 3 Jan 2019
Shulyakova, N. O. (2016). Studies of mitochondrial dysfunction in models of Rett syndrome (PhD Thesis).
Simacek, J., Dimian, A. F., & McComas, J. J. (2017). Communication intervention for young children with severe neurodevelopmental disabilities via telehealth. Journal of Autism and Developmental Disorders,47(3), 744–767. https://doi.org/10.1007/s10803-016-3006-z.
Smeets, E. E. J., Pelc, K., & Dan, B. (2011). Rett syndrome Molecular Syndromology,2(3–5), 113–127. https://doi.org/10.1159/000337637.
Smith-Hicks, C. L., Gupta, S., Ewen, J. B., Hong, M., Kratz, L., Kelley, R., et al. (2017). Randomized open-label trial of dextromethorphan in Rett syndrome. Neurology,89(16), 1684–1690. https://doi.org/10.1212/WNL.0000000000004515.
Specchio, N., Balestri, M., Striano, P., Cilio, M. R., Nardello, R., Patanè, S., et al. (2010). Efficacy of levetiracetam in the treatment of drug-resistant Rett syndrome. Epilepsy Research,88(2–3), 112–117. https://doi.org/10.1016/j.eplepsyres.2009.10.005.
Stenbom, Y., Tonnby, B., & Hagberg, B. (1998). Lamotrigine in Rett syndrome: treatment experience from a pilot study. European Child & Adolescent Psychiatry,7(1), 49–52.
Strebl, M. G., Campbell, A. J., Zhao, W.-N., Schroeder, F. A., Riley, M. M., Chindavong, P. S., et al. (2017). HDAC6 brain mapping with [18F]Bavarostat enabled by a Ru-mediated deoxyfluorination. ACS Central Science,3(9), 1006–1014. https://doi.org/10.1021/acscentsci.7b00274.
Szczesna, K., de la Caridad, O., Petazzi, P., Soler, M., Roa, L., Saez, M. A., et al. (2014). Improvement of the Rett syndrome phenotype in a MeCP2 mouse model upon treatment with levodopa and a dopa-decarboxylase inhibitor. Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology, 39(12), 2846–2856. https://doi.org/10.1038/npp.2014.136
Tao, J., Wu, H., Coronado, A. A., de Laittre, E., Osterweil, E. K., Zhang, Y., et al. (2016). Negative allosteric modulation of mGluR5 partially corrects pathophysiology in a mouse model of Rett syndrome. The Journal of Neuroscience,36(47), 11946–11958. https://doi.org/10.1523/JNEUROSCI.0672-16.2016.
Titlestad, K. B., & Eldevik, S. (2019). Brief report: Modest but clinically meaningful effects of early behavioral intervention in twins with Rett syndrome—A case study. Journal of Autism and Developmental Disorders. https://doi.org/10.1007/s10803-019-04185-9.
Toward, M. A., Abdala, A. P., Knopp, S. J., Paton, J. F. R., & Bissonnette, J. M. (2013). Increasing brain serotonin corrects CO2 chemosensitivity in methyl-CpG-binding protein 2 (Mecp2)-deficient mice. Experimental Physiology,98(3), 842–849. https://doi.org/10.1113/expphysiol.2012.069872.
Tropea, D., Giacometti, E., Wilson, N. R., Beard, C., McCurry, C., Fu, D. D., et al. (2009). Partial reversal of Rett Syndrome-like symptoms in MeCP2 mutant mice. Proceedings of the National Academy of Sciences,106(6), 2029–2034.
Uldall, P., Hansen, F. J., & Tonnby, B. (1993). Lamotrigine in Rett syndrome. Neuropediatrics,24(6), 339–340. https://doi.org/10.1055/s-2008-1071568.
Valacchi, G., Virgili, F., Cervellati, C., & Pecorelli, A. (2018). OxInflammation: From subclinical condition to pathological biomarker. Frontiers in Physiology. https://doi.org/10.3389/fphys.2018.00858.
Valenti, D., de Bari, L., Vigli, D., Lacivita, E., Leopoldo, M., Laviola, G., et al. (2017). Stimulation of the brain serotonin receptor 7 rescues mitochondrial dysfunction in female mice from two models of Rett syndrome. Neuropharmacology,121, 79–88. https://doi.org/10.1016/j.neuropharm.2017.04.024.
Vashi, N., & Justice, M. J. (2019). Treating Rett syndrome: from mouse models to human therapies. Mammalian Genome,30(5), 90–110. https://doi.org/10.1007/s00335-019-09793-5.
Vecsler, M., Zeev, B. B., Nudelman, I., Anikster, Y., Simon, A. J., Amariglio, N., et al. (2011). Ex vivo treatment with a novel synthetic aminoglycoside NB54 in primary fibroblasts from Rett syndrome patients suppresses MECP2 nonsense mutations. PLoS ONE,6(6), e20733.
Venugopal, A., Chandran, M., Eruppakotte, N., Kizhakkillach, S., Breezevilla, S. C., & Vellingiri, B. (2018). Monogenic diseases in India. Mutation Research,776, 23–31. https://doi.org/10.1016/j.mrrev.2018.03.003.
Vigli, D., Cosentino, L., Raggi, C., Laviola, G., Woolley-Roberts, M., & De Filippis, B. (2018). Chronic treatment with the phytocannabinoid Cannabidivarin (CBDV) rescues behavioural alterations and brain atrophy in a mouse model of Rett syndrome. Neuropharmacology,140, 121–129. https://doi.org/10.1016/j.neuropharm.2018.07.029.
Vignoli, A., Savini, M. N., Nowbut, M. S., Peron, A., Turner, K., La Briola, F., et al. (2017). Effectiveness and tolerability of antiepileptic drugs in 104 girls with Rett syndrome. Epilepsy & Behavior: E&B,66, 27–33. https://doi.org/10.1016/j.yebeh.2016.10.006.
Villani, C., Sacchetti, G., Bagnati, R., Passoni, A., Fusco, F., Carli, M., & Invernizzi, R. W. (2016). Lovastatin fails to improve motor performance and survival in methyl-CpG-binding protein2-null mice. eLife. https://doi.org/10.7554/eLife.22409
Wang, D., Belakhov, V., Kandasamy, J., Baasov, T., Li, S.-C., Li, Y.-T., et al. (2012). The designer aminoglycoside NB84 significantly reduces glycosaminoglycan accumulation associated with MPS IH in the Idua-W392X mouse. Molecular genetics and metabolism,105(1), 116–125.
Ward, B. C., Agarwal, S., Wang, K., Berger-Sweeney, J., & Kolodny, N. H. (2008). Longitudinal brain MRI study in a mouse model of Rett syndrome and the effects of choline. Neurobiology of Disease,31(1), 110–119. https://doi.org/10.1016/j.nbd.2008.03.009.
Wiedemann, A., Renard, E., Hernandez, M., Dousset, B., Brezin, F., Lambert, L., et al. (2018). Annual injection of zoledronic acid improves bone status in children with cerebral palsy and Rett syndrome. Calcified Tissue International. https://doi.org/10.1007/s00223-018-0505-2.
Xu, X., Kozikowski, A. P., & Pozzo-Miller, L. (2014). A selective histone deacetylase-6 inhibitor improves BDNF trafficking in hippocampal neurons from Mecp2 knockout mice: implications for Rett syndrome. Frontiers in Cellular Neuroscience,8, 68. https://doi.org/10.3389/fncel.2014.00068.
Yu, D., Sakurai, F., & Corey, D. R. (2011). Clonal Rett Syndrome cell lines to test compounds for activation of wild-type MeCP2 expression. Bioorganic & Medicinal Chemistry Letters,21(18), 5202–5205. https://doi.org/10.1016/j.bmcl.2011.07.053.
Zanella, S., Mebarek, S., Lajard, A.-M., Picard, N., Dutschmann, M., & Hilaire, G. (2008). Oral treatment with desipramine improves breathing and life span in Rett syndrome mouse model. Respiratory Physiology & Neurobiology, 160(1), 116–121. https://doi.org/10.1016/j.resp.2007.08.009.
Zhang, S., & Zhang, Z.-Y. (2007). PTP1B as a drug target: recent developments in PTP1B inhibitor discovery. Drug Discovery Today,12(9–10), 373–381. https://doi.org/10.1016/j.drudis.2007.03.011.
Zhong, W., Johnson, C. M., Wu, Y., Cui, N., Xing, H., Zhang, S., et al. (2016). Effects of early-life exposure to THIP on phenotype development in a mouse model of Rett syndrome. Journal of Neurodevelopmental Disorders,8, 37. https://doi.org/10.1186/s11689-016-9169-2.
Acknowledgment
This work was supported by University Grants Commission—National Fellowship for Scheduled Caste Candidates Mrs. Gomathi, UGC—RGNF JRF (Award number and date: F1-17.1/2017-18/RGNF-2017-18-SC-TAM-35724 /(SA-III/Website) 02/08/2017) and also this study was supported by grants from the Science and Engineering Research Board (SERB) Early Career Research (ECR) Award funded by the Government of India, New Delhi (Grant No. ECR/2016/ 001688). We acknowledge the department of Human Genetics and Molecular Biology, Bharathiar University for providing necessary infrastructure facilities and technical assistance to conduct this article.
Author information
Authors and Affiliations
Contributions
MG conceived of the study, participated in its design and coordination and drafted the manuscript; SP participated in the design and interpretation of the data; VB participated in the design and coordination of the study and helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gomathi, M., Padmapriya, S. & Balachandar, V. Drug Studies on Rett Syndrome: From Bench to Bedside. J Autism Dev Disord 50, 2740–2764 (2020). https://doi.org/10.1007/s10803-020-04381-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10803-020-04381-y