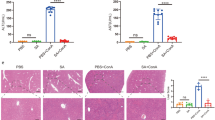
Abstract
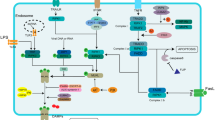
Sepsis-induced acute liver injury (ALI) is common in intensive care units. Angiotensin-converting enzyme 2 (ACE2) plays a vital role in hepatic fibrosis and steatosis; however, its role in sepsis-induced ALI remains unclear. This study found that hepatic ACE2 expression in cecal ligation and puncture (CLP)-treated mice significantly decreased 24 h after CLP. ACE2-transgenic (TG) mice exhibited a significant improvement in CLP-induced ALI, accompanied by the inhibition of hepatocyte apoptosis, oxidative stress, and inflammation, while ACE2-knockout mice demonstrated an opposite trend. During sepsis-induced ALI, ACE2-TG could also elevate the Ang-(1–7) and Mas receptor (MasR) levels in liver tissues. Interestingly, the MasR inhibitor A779 abrogated the favorable effects of ACE2 on CLP-induced ALI. In a bone marrow transplantation experiment, the ACE2-TG transplantation group showed significantly improved inflammation and liver dysfunction, less hepatocyte apoptosis, and reduced oxidative stress after CLP compared with the wild-type transplantation group. In contrast, the ACE2-knockout group showed poor inflammatory response and liver dysfunction, significantly more hepatocyte apoptosis, and elevated oxidative stress than the wild-type transplantation group after CLP. ACE2 protects against sepsis-induced ALI by inhibiting hepatocyte apoptosis, oxidative stress, and inflammation via the Ang-(1–7)–Mas receptor axis. Thus, targeting ACE2 may be a promising novel strategy for preventing and treating sepsis-induced ALI.







Similar content being viewed by others
Availability of Data and Materials
The data and materials used in this study are available from the corresponding authors upon reasonable request.
References
Singer, M., C.S. Deutschman, C.W. Seymour, M. Shankar-Hari, D. Annane, M. Bauer, R. Bellomo, G.R. Bernard, J.D. Chiche, C.M. Coopersmith, R.S. Hotchkiss, M.M. Levy, J.C. Marshall, G.S. Martin, S.M. Opal, G.D. Rubenfeld, T. van der Poll, J.L. Vincent, and D.C. Angus. 2016. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). Journal of the American Medical Association 315: 801–810. https://doi.org/10.1001/jama.2016.0287.
Woznica, E.A., M. Inglot, R.K. Woznica, and L. Lysenko. 2018. Liver dysfunction in sepsis. Advances in Clinical and Experimental Medicine 27: 547–551. https://doi.org/10.17219/acem/68363.
Nesseler, N., Y. Launey, C. Aninat, F. Morel, Y. Mallédant, and P. Seguin. 2012. Clinical review: The liver in sepsis. Critical Care 16: 235. https://doi.org/10.1186/cc11381.
Savio, L.E.B., P.D. Mello, V.R. Figliuolo, T.F.D. Almeida, P.T. Santana, S.D.S. Oliveira, C.L.M. Silva, L. Feldbrugge, E. Csizmadia, R.D. Minshall, M.S. Longhi, Y. Wu, S.C. Robson, and R. Coutinho-Silva. 2017. CD39 limits P2X7 receptor inflammatory signaling and attenuates sepsis-induced liver injury. Journal of Hepatology 67: 716–726. https://doi.org/10.1016/j.jhep.2017.05.021.
Qin, X., X. Jiang, X. Jiang, Y. Wang, Z. Miao, W. He, G. Yang, Z. Lv, Y. Yu, and Y. Zheng. 2016. Micheliolide inhibits LPS-induced inflammatory response and protects mice from LPS challenge. Scientific Reports 6: 23240. https://doi.org/10.1038/srep23240.
Sun, J., J.X. Zhang, X.F. Wang, F.X. Ji, C. Ronco, J.K. Tian, and Y.J. Yin. 2020. Gut-liver crosstalk in sepsis-induced liver injury. Critical Care 24: 614. https://doi.org/10.1186/s13054-020-03327-1.
Gleeson, P.J., I.A. Crippa, W. Mongkolpun, F.Z. Cavicchi, T. Van Meerhaeghe, S. Brimioulle, F.S. Taccone, J.-L. Vincent, and J. Creteur. 2019. Renin as a marker of tissue-perfusion and prognosis in critically ill patients. Critical Care Medicine 47: 152–158. https://doi.org/10.1097/CCM.0000000000003544.
Nguyen, M., D. Denimal, A. Dargent, P.G. Guinot, L. Duvillard, J.P. Quenot, and B. Bouhemad. 2019. Plasma renin concentration is associated with hemodynamic deficiency and adverse renal outcome in septic shock. Shock (Augusta, Ga.) 52: e22–e30. https://doi.org/10.1097/SHK.0000000000001285.
Bellomo, R., R.G. Wunderink, H. Szerlip, S.W. English, L.W. Busse, A.M. Deane, A.K. Khanna, M.T. McCurdy, M. Ostermann, P.J. Young, D.R. Handisides, L.S. Chawla, G.F. Tidmarsh, and T.E. Albertson. 2020. Angiotensin I and angiotensin II concentrations and their ratio in catecholamine-resistant vasodilatory shock. Critical Care (London, England) 24: 43. https://doi.org/10.1186/s13054-020-2733-x.
Grace, J.A., C.B. Herath, K.Y. Mak, L.M. Burrell, and P.W. Angus. 2012. Update on new aspects of the renin-angiotensin system in liver disease: Clinical implications and new therapeutic options. Clinical Science (London, England) 123: 225–239. https://doi.org/10.1042/cs20120030.
Rodrigues Prestes, T.R., N.P. Rocha, A.S. Miranda, A.L. Teixeira, and A.C. Simoes-E-Silva. 2017. The anti-inflammatory potential of ACE2/angiotensin-(1–7)/Mas receptor axis: Evidence from basic and clinical research. Current Drug Targets 18: 1301–1313. https://doi.org/10.2174/1389450117666160727142401.
Mak, K.Y., R. Chin, S.C. Cunningham, M.R. Habib, J. Torresi, A.F. Sharland, I.E. Alexander, P.W. Angus, and C.B. Herath. 2015. ACE2 therapy using adeno-associated viral vector inhibits liver fibrosis in mice. Molecular Therapy 23: 1434–1443. https://doi.org/10.1038/mt.2015.92.
Wu, H.T., Y.W. Chuang, C.P. Huang, and M.H. Chang. 2018. Loss of angiotensin converting enzyme II (ACE2) accelerates the development of liver injury induced by thioacetamide. Experimental Animals 67: 41–49. https://doi.org/10.1538/expanim.17-0053.
Li, Y., Y. Cao, Z. Zeng, M. Liang, Y. Xue, C. Xi, M. Zhou, and W. Jiang. 2015. Angiotensin-converting enzyme 2/angiotensin-(1–7)/Mas axis prevents lipopolysaccharide-induced apoptosis of pulmonary microvascular endothelial cells by inhibiting JNK/NF-κB pathways. Scientific Reports 5: 8209. https://doi.org/10.1038/srep08209.
Tao, L., Y. Qiu, X. Fu, R. Lin, C. Lei, J. Wang, and B. Lei. 2016. Angiotensin-converting enzyme 2 activator diminazene aceturate prevents lipopolysaccharide-induced inflammation by inhibiting MAPK and NF-kappaB pathways in human retinal pigment epithelium. Journal of Neuroinflammation 13: 35. https://doi.org/10.1186/s12974-016-0489-7.
Rittirsch, D., M.S. Huber-Lang, M.A. Flierl, and P.A. Ward. 2009. Immunodesign of experimental sepsis by cecal ligation and puncture. Nature Protocols 4: 31–36. https://doi.org/10.1038/nprot.2008.214.
Abdel-Hakeem, E.A., S.M.N. Abdel Hafez, B.A. Kamel, and H.A. Abdel-Hamid. 2022. Angiotensin 1–7 mitigates rhabdomyolysis induced renal injury in rats via modulation of TLR-4/NF-kB/iNOS and Nrf-2/heme-oxygenase-1 signaling pathways. Life Sciences 303: 120678. https://doi.org/10.1016/j.lfs.2022.120678.
Guo, X., J. Qiu, and Y. Qian. 2021. 6-Shogaol Mitigates sepsis-associated hepatic injury through transcriptional regulation. Nutrients 13: 3427. https://doi.org/10.3390/nu13103427.
Fan, J., M. He, C.-J. Wang, and M. Zhang. 2021. Gadolinium chloride inhibits the production of liver interleukin-27 and mitigates liver injury in the CLP mouse model. Mediators of Inflammation 2021: 2605973. https://doi.org/10.1155/2021/2605973.
Donoghue, M., F. Hsieh, E. Baronas, K. Godbout, M. Gosselin, N. Stagliano, M. Donovan, B. Woolf, K. Robison, R. Jeyaseelan, R.E. Breitbart, and S. Acton. 2000. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circulation Research 87: E1–9. https://doi.org/10.1161/01.res.87.5.e1.
Clausen, T.M., D.R. Sandoval, C.B. Spliid, J. Pihl, H.R. Perrett, C.D. Painter, A. Narayanan, S.A. Majowicz, E.M. Kwong, R.N. McVicar, B.E. Thacker, C.A. Glass, Z. Yang, J.L. Torres, G.J. Golden, P.L. Bartels, R.N. Porell, A.F. Garretson, L. Laubach, J. Feldman, X. Yin, Y. Pu, B.M. Hauser, T.M. Caradonna, B.P. Kellman, C. Martino, P.L.S.M. Gordts, S.K. Chanda, A.G. Schmidt, K. Godula, S.L. Leibel, J. Jose, K.D. Corbett, A.B. Ward, A.F. Carlin, and J.D. Esko. 2020. SARS-CoV-2 infection depends on cellular heparan sulfate and ACE2. Cell 183: 1043-1057.e15. https://doi.org/10.1016/j.cell.2020.09.033.
Zou, X., K. Chen, J. Zou, P. Han, J. Hao, and Z. Han. 2020. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Frontiers of Medicine 14: 185–192. https://doi.org/10.1007/s11684-020-0754-0.
Baig, A.M., A. Khaleeq, U. Ali, and H. Syeda. 2020. Evidence of the COVID-19 virus targeting the CNS: Tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chemical Neuroscience 11: 995–998. https://doi.org/10.1021/acschemneuro.0c00122.
Garabelli, P.J., J.G. Modrall, J.M. Penninger, C.M. Ferrario, and M.C. Chappell. 2008. Distinct roles for angiotensin-converting enzyme 2 and carboxypeptidase A in the processing of angiotensins within the murine heart. Experimental Physiology 93: 613–621. https://doi.org/10.1113/expphysiol.2007.040246.
Tipnis, S.R., N.M. Hooper, R. Hyde, E. Karran, G. Christie, and A.J. Turner. 2000. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. Journal of Biological Chemistry 275: 33238–33243. https://doi.org/10.1074/jbc.M002615200.
Zhao, Y.X., H.Q. Yin, Q.T. Yu, Y. Qiao, H.Y. Dai, M.X. Zhang, L. Zhang, Y.F. Liu, L.C. Wang, D.S. Liu, B.P. Deng, Y.H. Zhang, C.M. Pan, H.D. Song, X. Qu, H. Jiang, C.X. Liu, X.T. Lu, B. Liu, F. Gao, and B. Dong. 2010. ACE2 overexpression ameliorates left ventricular remodeling and dysfunction in a rat model of myocardial infarction. Human Gene Therapy 21: 1545–1554. https://doi.org/10.1089/hum.2009.160.
Badae, N.M., A.S. El Naggar, and S.M. El Sayed. 2019. Is the cardioprotective effect of the ACE2 activator diminazene aceturate more potent than the ACE inhibitor enalapril on acute myocardial infarction in rats? Canadian Journal of Physiology and Pharmacology 97: 638–646. https://doi.org/10.1139/cjpp-2019-0078.
Silva de Almeida, T.C., K. Lanza, R. da Silva Filha, and LMC, C. C., Fonseca, E. G., Chagas, M. W., Rocha, N. P., de Sá, M. A., Vieira, M. A. R., Caliari, M. V., Kangussu, L. M., Ferreira, A. J., Simões, E. S. A. C. 2020. ACE2 activator diminazene aceturate exerts renoprotective effects in gentamicin-induced acute renal injury in rats. Clinical Science (London, England) 134: 3093–3106. https://doi.org/10.1042/cs20201022.
Wang, T., F. Xie, Y.H. Li, and B. Liang. 2021. Downregulation of ACE2 is associated with advanced pathological features and poor prognosis in clear cell renal cell carcinoma. Future Oncology 17: 5033–5044. https://doi.org/10.2217/fon-2020-1164.
Cao, X., F. Yang, T. Shi, M. Yuan, Z. Xin, R. Xie, S. Li, H. Li, and J.K. Yang. 2016. Angiotensin-converting enzyme 2/angiotensin-(1–7)/Mas axis activates Akt signaling to ameliorate hepatic steatosis. Science and Reports 6: 21592. https://doi.org/10.1038/srep21592.
Nehme, A., F.A. Zouein, Z.D. Zayeri, and K. Zibara. 2019. An update on the tissue renin angiotensin system and its role in physiology and pathology. Journal of Cardiovascular Development and Disease 6: 14. https://doi.org/10.3390/jcdd6020014.
Ambrocio-Ortiz, E., G. Pérez-Rubio, A.D. Del Ángel-Pablo, I. Buendía-Roldán, L. Chávez-Galán, R.J. Hernández-Zenteno, A. Ramírez-Venegas, J. Rojas-Serrano, M. Mejía, R. Pérez-Padilla, C. Guadarrama-Pérez, and R. Falfán-Valencia. 2021. Angiotensin-converting enzyme 2 (ACE2) in the context of respiratory diseases and its importance in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Pharmaceuticals (Basel) 14: 805. https://doi.org/10.3390/ph14080805.
Santos, R.A.S., W.O. Sampaio, A.C. Alzamora, D. Motta-Santos, N. Alenina, M. Bader, and M.J. Campagnole-Santos. 2018. The ACE2/angiotensin-(1–7)/MAS axis of the renin-angiotensin system: Focus on angiotensin-(1–7). Physiological Reviews 98: 505–553. https://doi.org/10.1152/physrev.00023.2016.
Wang, L., T. Jiang, Y. Yang, J. Mao, Q. Wang, R. Yu, B. Wang, and J. Yin. 2022. Angiotensin-(1–7) alleviates acute lung injury by activating the Mas receptor in neutrophil. Annals of Translational Medicine's 10: 1395. https://doi.org/10.21037/atm-22-6193.
Pan, H., W. Huang, Z. Wang, F. Ren, L. Luo, J. Zhou, M. Tian, and L. Tang. 2021. The ACE2-Ang-(1–7)-Mas axis modulates M1/M2 macrophage polarization to relieve CLP-induced inflammation via TLR4-mediated NF-кb and MAPK pathways. Journal of Inflammation Research 14: 2045–2060. https://doi.org/10.2147/jir.S307801.
Strnad, P., F. Tacke, A. Koch, and C. Trautwein. 2017. Liver - guardian, modifier and target of sepsis. Nature Reviews Gastroenterology & Hepatology 14: 55–66. https://doi.org/10.1038/nrgastro.2016.168.
Wang, Y., B. Liu, P. Wu, Y. Chu, S. Gui, Y. Zheng, and X. Chen. 2022. Dietary selenium alleviated mouse liver oxidative stress and NAFLD induced by obesity by regulating the KEAP1/NRF2 pathway. Antioxidants (Basel) 11: 349. https://doi.org/10.3390/antiox11020349.
Lim, J.-O., K.H. Song, I.S. Lee, S.-J. Lee, W.-I. Kim, S.-W. Pak, I.-S. Shin, and T. Kim. 2021. Cimicifugae Rhizoma extract attenuates oxidative stress and airway inflammation via the upregulation of Nrf2/HO-1/NQO1 and downregulation of NF-κB phosphorylation in ovalbumin-induced asthma. Antioxidants (Basel, Switzerland) 10: 1626. https://doi.org/10.3390/antiox10101626.
Farzanegi, P., A. Dana, Z. Ebrahimpoor, M. Asadi, and M.A. Azarbayjani. 2019. Mechanisms of beneficial effects of exercise training on non-alcoholic fatty liver disease (NAFLD): Roles of oxidative stress and inflammation. European Journal of Sport Science 19: 994–1003. https://doi.org/10.1080/17461391.2019.1571114.
Klaunig, J.E. 2018. Oxidative Stress and Cancer. Current Pharmaceutical Design 24: 4771–4778. https://doi.org/10.2174/1381612825666190215121712.
Zamudio-Cuevas, Y., K. Martínez-Flores, G.A. Martínez-Nava, D. Clavijo-Cornejo, J. Fernández-Torres, and R. Sánchez-Sánchez. 2022. Rheumatoid arthritis and oxidative stress. Cellular and Molecular Biology (Noisy-le-Grand, France) 68: 174–184. https://doi.org/10.14715/cmb/2022.68.6.28.
Abdel-Fattah, M.M., A.N.A.M. Elgendy, and W.R. Mohamed. 2021. Xanthenone, ACE2 activator, counteracted gentamicin-induced nephrotoxicity in rats: Impact on oxidative stress and ACE2/Ang-(1–7) signaling. Life Sciences 275: 119387. https://doi.org/10.1016/j.lfs.2021.119387.
Zhao, S., Sun, W., Jiang, P. 2021. Role of the ACE2/Ang-(1–7)/Mas axis in glucose metabolism. Reviews in Cardiovascular Medicine 22: 769–777. https://doi.org/10.31083/j.rcm2203083.
Song, X., W. Hu, H. Yu, L. Zhao, Y. Zhao, X. Zhao, H.-H. Xue, and Y. Zhao. 2023. Little to no expression of angiotensin-converting enzyme-2 on most human peripheral blood immune cells but highly expressed on tissue macrophages. Cytometry. Part A : The Journal of the International Society For Analytical Cytology 103: 136–145. https://doi.org/10.1002/cyto.a.24285.
Nuovo, G.J., D. Suster, H. Awad, J.-J. Michaille, and E. Tili. 2022. The histologic and molecular correlates of liver disease in fatal COVID-19 including with alcohol use disorder. Annals of Diagnostic Pathology 57: 151881. https://doi.org/10.1016/j.anndiagpath.2021.151881.
Stevens, J.P., V.L. Kolachala, G.N. Joshi, S. Nagpal, G. Gibson, and N.A. Gupta. 2022. Angiotensin-converting enzyme-2 (ACE2) expression in pediatric liver disease. Applied Immunohistochemistry & Molecular Morphology : AIMM 30: 647–653. https://doi.org/10.1097/PAI.0000000000001068.
Liu, A., W. Wang, H. Fang, Y. Yang, X. Jiang, S. Liu, J. Hu, Q. Hu, U. Dahmen, and O. Dirsch. 2015. Baicalein protects against polymicrobial sepsis-induced liver injury via inhibition of inflammation and apoptosis in mice. European Journal of Pharmacology 748: 45–53. https://doi.org/10.1016/j.ejphar.2014.12.014.
Tian, L.L., J. Zhang, Z.Z. Wang, S.C. Chen, X.B. Zou, Z.K. Yu, and C.C. Kang. 2020. KLF15 reduces the level of apoptosis in mouse liver induced by sepsis by inhibiting p38MAPK/ERK1/2 signaling pathway. European Review for Medical and Pharmacological Sciences 24: 10819–10828. https://doi.org/10.26355/eurrev_202010_23444.
Chazaud, B. 2020. Inflammation and skeletal muscle regeneration: Leave it to the macrophages! Trends in Immunology 41: 481–492. https://doi.org/10.1016/j.it.2020.04.006.
Liu, E.H., Z.N. Zheng, C.X. Xiao, X. Liu, and X.Q. Lin. 2020. IL-22 relieves sepsis-induced liver injury via activating JAK/STAT3 signaling pathway. Journal of Biological Regulators and Homeostatic Agents 34: 1719–1727. https://doi.org/10.23812/20-326-A.
Funding
This work was supported by the National Natural Science Foundation of China (82172123).
Author information
Authors and Affiliations
Contributions
Methodology: LL and YL; formal analysis: LL; validation: LL, YL, JL, and XX; software: LL; investigation: LL, YL, JL, XX, and TW; data curation: LL; visualization: LL; conceptualization: HL and SG; writing—original draft preparation: LL; writing-review and editing: HL and SG; supervision: SG; funding acquisition: SG. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethics Approval and Consent to Participate
Animal experiments were performed in accordance with the National Animal Protection and Use Guidelines and approved by the Local Ethical Committee of the Beijing Chaoyang Hospital of Capital Medicine University (No: 2021-animal-35).
Consent for Publication
All authors have approved the manuscript for publication.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, L., Li, Y., Li, JX. et al. ACE2 Expressed on Myeloid Cells Alleviates Sepsis-Induced Acute Liver Injury via the Ang-(1–7)–Mas Receptor Axis. Inflammation (2024). https://doi.org/10.1007/s10753-023-01949-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10753-023-01949-5