Abstract
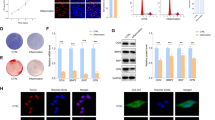
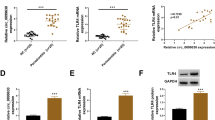
Collagen triple helix repeat containing 1 (CTHRC1), a secreted glycoprotein, is widely expressed in many tissues. It has been recently defined as a novel marker for rheumatoid arthritis (RA), a systemic inflammatory disorder. However, the precise role of CTHRC1 in other chronic inflammatory diseases, like periodontal disease, remains unclear. This research aimed to explore the presence of CTHRC1 in periodontal inflammation, determine the precise role in inflammatory response modulation in periodontal ligament cells (PDLCs), and explore its underlying mechanisms. In vivo gingival crevicular fluid (GCF) and gingivae were obtained from healthy people and chronic periodontitis patients. Maxillary tissues of mice with or without ligature-induced periodontitis were immunostained for CTHRC1. In vitro human PDLCs were treated with tumor necrosis factor alpha (TNF-α) to mimic the inflammatory environment. Small interfering RNA (siRNA) was used to silence CTHRC1. SB203580 was used to inhibit the p38 mitogen-activated protein kinase (MAPK) pathway. CTHRC1 was highly expressed in GCF and gingival tissues of periodontitis patients. Animal models also revealed the same tendency. CTHRC1 knockdown promoted inflammatory cytokine production and activated the p38 MAPK signaling pathway in PDLCs. Inhibiting the p38 MAPK signaling pathway partially attenuated the inflammatory responses. This study revealed that CTHRC1 was highly expressed in periodontitis and suggested that CTHRC1 might play an important role in modulating periodontal inflammation.





Similar content being viewed by others
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Kinane, D.F., P.G. Stathopoulou, and P.N. Papapanou. 2017. Periodontal diseases. Nature Reviews. Disease Primers 3: 17038.
Hajishengallis, G. 2015. Periodontitis: from microbial immune subversion to systemic inflammation. Nature Reviews. Immunology 15: 30–44.
Lekic, P., and C.A. McCulloch. 1996. Periodontal ligament cell population: the central role of fibroblasts in creating a unique tissue. The Anatomical Record 245 (2): 327–341.
Jönsson, D., D. Nebel, G. Bratthall, and B.O. Nilsson. 2011. The human periodontal ligament cell: a fibroblast-like cell acting as an immune cell. Journal of Periodontal Research 46 (2): 153–157.
El-Awady, A.R., R.L. Messer, A.Y. Gamal, M.M. Sharawy, K.H. Wenger, and C.A. Lapp. 2010. Periodontal ligament fibroblasts sustain destructive immune modulators of chronic periodontitis. Journal of Periodontology 81 (9): 1324–1335.
Liu, J., X. Tang, C. Li, C. Pan, Q. Li, F. Geng, and Y. Pan. 2015. Porphyromonas gingivalis promotes the cell cycle and inflammatory cytokine production in periodontal ligament fibroblasts. Archives of Oral Biology 60 (8): 1153–1161.
Abidi, A.H., C.S. Presley, M. Dabbous, D.A. Tipton, S.M. Mustafa, and B.M. Moore 2nd. 2018. Anti-inflammatory activity of cannabinoid receptor 2 ligands in primary hPDL fibroblasts. Archives of Oral Biology 87: 79–85.
Zhang, Y., and X. Li. 2015. Lipopolysaccharide-regulated production of bone sialoprotein and interleukin-8 in human periodontal ligament fibroblasts: the role of toll-like receptors 2 and 4 and the MAPK pathway. Journal of Periodontal Research 50 (2): 141–151.
Pyagay, P., M. Heroult, Q. Wang, W. Lehnert, J. Belden, L. Liaw, R.E. Friesel, and V. Lindner. 2005. Collagen triple helix repeat containing 1, a novel secreted protein in injured and diseased arteries, inhibits collagen expression and promotes cell migration. Circulation Research 96 (2): 261–268.
Stohn, J.P., Q. Wang, M.E. Siviski, K. Kennedy, Y.R. Jin, D. Kacer, V. DeMambro, L. Liaw, C.P. Vary, C.J. Rosen, I. Prudovsky, and V. Lindner. 2015. Cthrc1 controls adipose tissue formation, body composition, and physical activity. Obesity (Silver Spring) 23 (8): 1633–1642.
Jin, Y.R., J.P. Stohn, Q. Wang, K. Nagano, R. Baron, M.L. Bouxsein, C.J. Rosen, V.A. Adarichev, and V. Lindner. 2017. Inhibition of osteoclast differentiation and collagen antibody-induced arthritis by CTHRC1. Bone. 97: 153–167.
Wang, Y., and D.N. Tatakis. 2017. Human gingiva transcriptome during wound healing. Journal of Clinical Periodontology 44 (4): 394–402.
Qin, S., J.H. Zheng, Z.H. Xia, J. Qian, C.L. Deng, and S.L. Yang. 2019. CTHRC1 promotes wound repair by increasing M2 macrophages via regulating the TGF-β and notch pathways. Biomedicine & Pharmacotherapy 113: 108594.
Wang, C., Z. Li, F. Shao, X. Yang, X. Feng, S. Shi, Y. Gao, and J. He. 2017. High expression of Collagen Triple Helix Repeat Containing 1 (CTHRC1) facilitates progression of oesophageal squamous cell carcinoma through MAPK/MEK/ERK/FRA-1 activation. Journal of Experimental & Clinical Cancer Research 36 (1): 84.
Wang, C., W. Gu, B. Sun, Y. Zhang, Y. Ji, X. Xu, and Y. Wen. 2017. CTHRC1 promotes osteogenic differentiation of periodontal ligament stem cells by regulating TAZ. Journal of Molecular Histology 48 (4): 311–319.
Wu, Q., Q. Yang, and H. Sun. 2018. Collagen triple helix repeat containing-1: a novel biomarker associated with disease activity in Systemic lupus erythematosus. Lupus. 27 (13): 2076–2085.
Duarte CW, Stohn JP, Wang Q, Emery IF, Prueser A, and Lindner V. 2014. Elevated plasma levels of the pituitary hormone Cthrc1 in individuals with red hair but not in patients with solid tumors. PLoS One. 19;9(6): e100449.
Shekhani, M.T., T.S. Forde, A. Adilbayeva, M. Ramez, A. Myngbay, Y. Bexeitov, V. Lindner, and V.A. Adarichev. 2016. Collagen triple helix repeat containing 1 is a new promigratory marker of arthritic pannus. Arthritis Research & Therapy 18: 171.
Li, Y.K., Y.M. Li, Y. Li, Y.R. Wei, J. Zhang, B. Li, Z.R. You, Y. Chen, B.Y. Huang, Q. Miao, Q.X. Wang, Y.S. Peng, M.E. Gershwin, R.Q. Tang, Z.L. Bian, and X. Ma. 2019. CTHRC1 expression in primary biliary cholangitis. Journal of Digestive Diseases 20 (7): 371–376.
Chamberlain, C.S., S.H. Brounts, D.G. Sterken, K.I. Rolnick, G.S. Baer, and R. Vanderby. 2011. Gene profiling of the rat medial collateral ligament during early healing using microarray analysis. Journal of Applied Physiology (Bethesda, MD: 1985) 111 (2): 552–565.
Patil, C.S., and K.L. Kirkwood. 2007. p38 MAPK signaling in oral-related diseases. Journal of Dental Research 86 (9): 812–825.
Lee, H.J., J.W. Cho, S.C. Kim, K.H. Kang, S.K. Lee, S.H. Pi, S.K. Lee, and E.C. Kim. 2006. Roles of p38 and ERK MAP kinases in IL-8 expression in TNF-alpha- and dexamethasone-stimulated human periodontal ligament cells. Cytokine. 35 (1-2): 67–76.
Xu, S., C. Jiang, H. Liu, H. Zhang, H. Liao, X. Wang, S. Yao, L. Ma, Y. Guo, and Z. Cao. 2020. Integrin-α9 and its corresponding ligands play regulatory roles in chronic periodontitis. Inflammation. 43 (4): 1488–1497.
Schmittgen, T.D., and K.J. Livak. 2008. Analyzing real-time PCR data by the comparative C(T) method. Nature Protocols 3 (6): 1101–1108.
Yli-Karjanmaa, M., K.S. Larsen, C.D. Fenger, L.K. Kristensen, N.A. Martin, P.T. Jensen, A. Breton, L. Nathanson, P.V. Nielsen, M.C. Lund, S.L. Carlsen, J.B. Gramsbergen, B. Finsen, J. Stubbe, L.H. Frich, H. Stolp, R. Brambilla, D.C. Anthony, M. Meyer, and K.L. Lambertsen. 2019. TNF deficiency causes alterations in the spatial organization of neurogenic zones and alters the number of microglia and neurons in the cerebral cortex. Brain, Behavior, and Immunity 82: 279–297.
Kudryavtseva, E., T.S. Forde, A.D. Pucker, and V.A. Adarichev. 2012. Wnt signaling genes of murine chromosome 15 are involved in sex-affected pathways of inflammatory arthritis. Arthritis and Rheumatism 64 (4): 1057–1068.
Myngbay, A., Y. Bexeitov, A. Adilbayeva, Z. Assylbekov, B.P. Yevstratenko, R.M. Aitzhanova, B. Matkarimov, V.A. Adarichev, and J. Kunz. 2019. CTHRC1: a new candidate biomarker for improved rheumatoid arthritis diagnosis. Frontiers in Immunology 10: 1353.
Buduneli, N., and D.F. Kinane. 2011. Host-derived diagnostic markers related to soft tissue destruction and bone degradation in periodontitis. Journal of Clinical Periodontology 8 (Suppl 11): 85–105.
Stadler, A.F., P.D. Angst, R.M. Arce, S.C. Gomes, R.V. Oppermann, and C. Susin. 2016. Gingival crevicular fluid levels of cytokines/chemokines in chronic periodontitis: a meta-analysis. Journal of Clinical Periodontology 43 (9): 727–745.
Han, P., T. Lloyd, Z. Chen, and Y. Xiao. 2016. Proinflammatory cytokines regulate cementogenic differentiation of periodontal ligament cells by Wnt/Ca (2+) signaling pathway. Journal of Interferon & Cytokine Research 36 (5): 328–337.
Baker, P.J., M. Dixon, R.T. Evans, L. Dufour, E. Johnson, and D.C. Roopenian. 1999. CD4(+) T cells and the proinflammatory cytokines gamma interferon and interleukin-6 contribute to alveolar bone loss in mice. Infection and Immunity 67 (6): 2804–2809.
Silva, T.A., G.P. Garlet, S.Y. Fukada, J.S. Silva, and F.Q. Cunha. 2007. Chemokines in oral inflammatory diseases: apical periodontitis and periodontal disease. Journal of Dental Research 86 (4): 306–319.
Pan, W., Q. Wang, and Q. Chen. 2019. The cytokine network involved in the host immune response to periodontitis. International Journal of Oral Science 11 (3): 30.
Mao, C.Y., Y.G. Wang, X. Zhang, X.Y. Zheng, T.T. Tang, and E.Y. Lu. 2016. Double-edged-sword effect of IL-1β on the osteogenesis of periodontal ligament stem cells via crosstalk between the NF-κB, MAPK and BMP/Smad signaling pathways. Cell Death & Disease 7 (7): e2296.
Mei, D., Y. Zhu, L. Zhang, and W. Wei. 2020. The role of CTHRC1 in regulation of multiple signaling and tumor progression and metastasis. Mediators of Inflammation 2020: 9578701.
Kumar, S., J. Boehm, and J.C. Lee. 2003. p38 MAP kinases: key signalling molecules as therapeutic targets for inflammatory diseases. Nature Reviews. Drug Discovery 2 (9): 717–726.
Guan, S.M., M. Zhang, J.J. He, and J.Z. Wu. 2009. Mitogen-activated protein kinases and phosphatidylinositol 3-kinase are involved in Prevotella intermedia-induced proinflammatory cytokines expression in human periodontal ligament cells. Biochemical and Biophysical Research Communications 386 (3): 471–476.
Huang, W., Y. Zhan, Y. Zheng, Y. Han, W. Hu, and J. Hou. Up-regulated ferritin in periodontitis promotes inflammatory cytokine expression in human periodontal ligament cells through transferrin receptor via ERK/P38 MAPK pathways. Clinical Science (London, England) 133 (1): 135–148.
Tang, L., X. Li, Y. Bai, P. Wang, and Y. Zhao. 2019. MicroRNA-146a negatively regulates the inflammatory response to Porphyromonas gingivalis in human periodontal ligament fibroblasts via TRAF6/p38 pathway. Journal of Periodontology 90 (4): 391–399.
Francis, M., G. Gopinathan, A. Salapatas, S. Nares, M. Gonzalez, T.G.H. Diekwisch, and X. Luan. 2020. SETD1 and NF-κB regulate periodontal inflammation through H3K4 trimethylation. Journal of Dental Research 99 (13): 1486–1493.
Du, L., Y. Li, and W. Liu. 2018. Maresin 1 regulates autophagy and inflammation in human periodontal ligament cells through glycogen synthase kinase-3β/β-catenin pathway under inflammatory conditions. Archives of Oral Biology 87: 242–247.
Song, H.K., E.M. Noh, J.M. Kim, Y.O. You, K.B. Kwon, and Y.R. Lee. 2019. Reversine inhibits MMP-3, IL-6 and IL-8 expression through suppression of ROS and JNK/AP-1 activation in interleukin-1β-stimulated human gingival fibroblasts. Archives of Oral Biology 108: 104530.
Funding
The study was supported by grants from the National Natural Science Foundation of China to Zhengguo Cao (81870776 and 81570946).
Author information
Authors and Affiliations
Contributions
YG contributed to conception, design, data acquisition, analysis, and interpretation, drafted and critically revised the manuscript; CJ, SY, LM contributed to design, data acquisition, and analysis, critically revised the manuscript; HZ, XW, SX contributed to data acquisition and analysis, critically revised the manuscript; ZC contributed to conception, design, and interpretation, drafted and critically revised the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The animal study was performed in accordance with the care and use of the laboratory animal manual by the US National Institutes of Health. This clinical study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the Wuhan University School of Stomatology (authorized c-14/2014). Informed consent was obtained from all individual participants included in the study.
Consent for publication
Patients signed informed consent regarding publishing their data.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, Y., Jiang, C., Yao, S. et al. CTHRC1 Knockdown Promotes Inflammatory Responses Partially by p38 MAPK Activation in Human Periodontal Ligament Cells. Inflammation 44, 1831–1842 (2021). https://doi.org/10.1007/s10753-021-01461-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-021-01461-8