Abstract
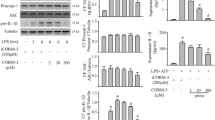
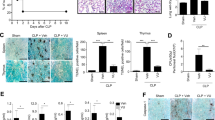
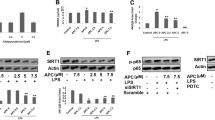
Sepsis is a disease that occurs as a result of systemic inflammatory response syndrome (SIRS) in response to an infection, contributing to multiple organ dysfunction and a high mortality rate. Interleukin-lβ (IL-1β) is a cytokine that plays critical roles in inflammation and cardiac dysfunction during severe sepsis. Intermedin1–53 (IMD1–53) has been recently discovered to possess potential endogenous anti-inflammatory and strong cardiovascular protective effects. To investigate whether IMD1–53 can inhibit the NLRP3/caspase-1/IL-1β pathway to alleviate cardiac injury and rescue heart function, sepsis was induced in vivo by caecal ligation and puncture (CLP) surgery, and lipopolysaccharides were used as septic stressors for cardiac fibroblasts (CFs) in vitro. The expressions of IMD1–53 receptors in sepsis rat heart were increased. After IMD1–53 treatment, inflammation caused by sepsis in vivo was greatly reduced, as shown by the downregulation of apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), nucleotide-binding domain and leucine-rich repeat containing family, pyrin containing 3 (NLRP3), pro-IL-1β, caspase 1, and nuclear translocation of nuclear factor-κB (NF-kB) protein levels. In addition, cardiac function was significantly improved and mean arterial blood pressure (MABP) increased by 34.8% (P < 0.05) which almost back to normal. Surprisingly, IMD1–53 inhibited cell apoptosis, as caspase 3 activity and Bax expression was significantly reduced in the heart upon IMD1–53 treatment. IMD1–53 abolished the upregulation of ASC, NLRP3, and caspase 1 protein levels in CFs induced by lipopolysaccharide (LPS). IMD1–53 increased cell survival rates and inhibited IL-1β production in the cell culture medium. IMD1–53 can protect against inflammation and heart injury during sepsis via attenuating the NLRP3/caspase-1/IL-1β pathway.




Similar content being viewed by others
References
Bell, D., and B.J. McDermott. 2008. Intermedin (adrenomedullin-2): a novel counter-regulatory peptide in the cardiovascular and renal systems. British Journal of Pharmacology 153 (Suppl 1): S247–S262. https://doi.org/10.1038/sj.bjp.0707494.
Bone, R.C., R.A. Balk, F.B. Cerra, R.P. Dellinger, A.M. Fein, W.A. Knaus, R. Schein, et al. 1992. American College of Chest Physicians/Society of Critical Care Medicine Consensus Conference: definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Critical Care Medicine 20 (6): 864–874.
Chang, J.R., X.H. Duan, B.H. Zhang, X. Teng, Y.B. Zhou, Y. Liu, Y.R. Yu, Y. Zhu, C.S. Tang, and Y.F. Qi. 2013. Intermedin1-53 attenuates vascular smooth muscle cell calcification by inhibiting endoplasmic reticulum stress via cyclic adenosine monophosphate/protein kinase A pathway. Experimental Biology and Medicine (Maywood, N.J.) 238 (10): 1136–1146. https://doi.org/10.1177/1535370213502619.
Fauvel, H., P. Marchetti, C. Chopin, P. Formstecher, and R. Neviere. 2001. Differential effects of caspase inhibitors on endotoxin-induced myocardial dysfunction and heart apoptosis. American Journal of Physiology. Heart and Circulatory Physiology 280 (4): H1608–H1614.
Fearon, D.T., and R.M. Locksley. 1996. The instructive role of innate immunity in the acquired immune response. Science 272 (5258): 50–53.
Fenton, K.E., and M.M. Parker. 2016. Cardiac function and dysfunction in sepsis. Clinics in Chest Medicine 37 (2): 289–298. https://doi.org/10.1016/j.ccm.2016.01.014.
Fisher, C.J., Jr., J.F. Dhainaut, S.M. Opal, J.P. Pribble, R.A. Balk, G.J. Slotman, T.J. Iberti, et al. 1994. Recombinant human interleukin 1 receptor antagonist in the treatment of patients with sepsis syndrome. Results from a randomized, double-blind, placebo-controlled trial. Phase III rhIL-1ra Sepsis Syndrome Study Group. JAMA 271 (23): 1836–1843.
Fisher, C.J., Jr., G.J. Slotman, S.M. Opal, J.P. Pribble, R.C. Bone, G. Emmanuel, D. Ng, D.C. Bloedow, M.A. Catalano, and Il-Ra Sepsis Syndrome Study Group. 1994. Initial evaluation of human recombinant interleukin-1 receptor antagonist in the treatment of sepsis syndrome: a randomized, open-label, placebo-controlled multicenter trial. Critical Care Medicine 22 (1): 12–21.
Furian, T., C. Aguiar, K. Prado, R.V. Ribeiro, L. Becker, N. Martinelli, N. Clausell, L.E. Rohde, and A. Biolo. 2012. Ventricular dysfunction and dilation in severe sepsis and septic shock: relation to endothelial function and mortality. Journal of Critical Care 27 (3): 319 e319–319 e315. https://doi.org/10.1016/j.jcrc.2011.06.017.
Hagiwara, M., G. Bledsoe, Z.R. Yang, R.S. Smith Jr., L. Chao, and J. Chao. 2008. Intermedin ameliorates vascular and renal injury by inhibition of oxidative stress. American Journal of Physiology. Renal Physiology 295 (6): F1735–F1743. https://doi.org/10.1152/ajprenal.90427.2008.
He, L., X. Peng, J. Zhu, X. Chen, H. Liu, C. Tang, Z. Dong, F. Liu, and Y. Peng. 2014. Mangiferin attenuate sepsis-induced acute kidney injury via antioxidant and anti-inflammatory effects. American Journal of Nephrology 40 (5): 441–450. https://doi.org/10.1159/000369220.
Hesse, D.G., K.J. Tracey, Y. Fong, K.R. Manogue, M.A. Palladino Jr., A. Cerami, G.T. Shires, and S.F. Lowry. 1988. Cytokine appearance in human endotoxemia and primate bacteremia. Surgery, Gynecology & Obstetrics 166 (2): 147–153.
Hong, Y., D.L. Hay, R. Quirion, and D.R. Poyner. 2012. The pharmacology of adrenomedullin 2/intermedin. British Journal of Pharmacology 166 (1): 110–120. https://doi.org/10.1111/j.1476-5381.2011.01530.x.
Hubbard, W.J., M. Choudhry, M.G. Schwacha, J.D. Kerby, L.W. Rue 3rd, K.I. Bland, and I.H. Chaudry. 2005. Cecal ligation and puncture. Shock 24 (Suppl 1): 52–57.
Kakkar, R., and R.T. Lee. 2010. Intramyocardial fibroblast myocyte communication. Circulation Research 106 (1): 47–57. https://doi.org/10.1161/CIRCRESAHA.109.207456.
Khan, M.M., W.L. Yang, and P. Wang. 2015. Endoplasmic reticulum stress in sepsis. Shock 44 (4): 294–304. https://doi.org/10.1097/SHK.0000000000000425.
Kinoshita, T., R. Imamura, H. Kushiyama, and T. Suda. 2015. NLRP3 mediates NF-kappaB activation and cytokine induction in microbially induced and sterile inflammation. PLoS One 10 (3): e0119179. https://doi.org/10.1371/journal.pone.0119179.
Li, H., Y. Bian, N. Zhang, J. Guo, C. Wang, W.B. Lau, and C. Xiao. 2013. Intermedin protects against myocardial ischemia-reperfusion injury in diabetic rats. Cardiovascular Diabetology 12: 91. https://doi.org/10.1186/1475-2840-12-91.
Luo, Y.P., L. Jiang, K. Kang, D.S. Fei, X.L. Meng, C.C. Nan, S.H. Pan, M.R. Zhao, and M.Y. Zhao. 2014. Hemin inhibits NLRP3 inflammasome activation in sepsis-induced acute lung injury, involving heme oxygenase-1. International Immunopharmacology 20 (1): 24–32. https://doi.org/10.1016/j.intimp.2014.02.017.
Makara, M.A., K.V. Hoang, L.P. Ganesan, E.D. Crouser, J.S. Gunn, J. Turner, L.S. Schlesinger, P.J. Mohler, and M.V. Rajaram. 2016. Cardiac electrical and structural changes during bacterial infection: an instructive model to study cardiac dysfunction in sepsis. Journal of the American Heart Association 5 (9). https://doi.org/10.1161/JAHA.116.003820.
Mao, K., S. Chen, M. Chen, Y. Ma, Y. Wang, B. Huang, Z. He, et al. 2013. Nitric oxide suppresses NLRP3 inflammasome activation and protects against LPS-induced septic shock. Cell Research 23 (2): 201–212. https://doi.org/10.1038/cr.2013.6.
McDonald, T.E., M.N. Grinman, C.M. Carthy, and K.R. Walley. 2000. Endotoxin infusion in rats induces apoptotic and survival pathways in hearts. American Journal of Physiology. Heart and Circulatory Physiology 279 (5): H2053–H2061.
Merx, M.W., and C. Weber. 2007. Sepsis and the heart. Circulation 116 (7): 793–802. https://doi.org/10.1161/CIRCULATIONAHA.106.678359.
Morelli, A., S. De Castro, J.L. Teboul, M. Singer, M. Rocco, G. Conti, L. De Luca, et al. 2005. Effects of levosimendan on systemic and regional hemodynamics in septic myocardial depression. Intensive Care Medicine 31 (5): 638–644. https://doi.org/10.1007/s00134-005-2619-z.
Ni, X., J. Zhang, C. Tang, and Y. Qi. 2014. Intermedin/adrenomedullin2: an autocrine/paracrine factor in vascular homeostasis and disease. Science China Life Sciences 57 (8): 781–789. https://doi.org/10.1007/s11427-014-4701-7.
Parrillo, J.E., M.M. Parker, C. Natanson, A.F. Suffredini, R.L. Danner, R.E. Cunnion, and F.P. Ognibene. 1990. Septic shock in humans. Advances in the understanding of pathogenesis, cardiovascular dysfunction, and therapy. Annals of Internal Medicine 113 (3): 227–242.
Potz, B.A., F.W. Sellke, and M.R. Abid. 2016. Endothelial ROS and impaired myocardial oxygen consumption in sepsis-induced cardiac dysfunction. Journal of Intensive Critical Care 2 (1).
Rattarasarn, C. 1997. Hypoglycemia in sepsis: risk factors and clinical characteristics. Journal of the Medical Association of Thailand 80 (12): 760–766.
Rudiger, A., and M. Singer. 2007. Mechanisms of sepsis-induced cardiac dysfunction. Critical Care Medicine 35 (6): 1599–1608. https://doi.org/10.1097/01.CCM.0000266683.64081.02.
Sarkar, A., M. Duncan, J. Hart, E. Hertlein, D.C. Guttridge, and M.D. Wewers. 2006. ASC directs NF-kappaB activation by regulating receptor interacting protein-2 (RIP2) caspase-1 interactions. Journal of Immunology 176 (8): 4979–4986.
Schroder, K., and J. Tschopp. 2010. The inflammasomes. Cell 140 (6): 821–832. https://doi.org/10.1016/j.cell.2010.01.040.
Teng, X., J. Song, G. Zhang, Y. Cai, F. Yuan, J. Du, C. Tang, and Y. Qi. 2011. Inhibition of endoplasmic reticulum stress by intermedin(1-53) protects against myocardial injury through a PI3 kinase-Akt signaling pathway. Journal of Molecular Medicine (Berlin, Germany) 89 (12): 1195–1205. https://doi.org/10.1007/s00109-011-0808-5.
Tissier, S., S. Lancel, X. Marechal, S. Mordon, F. Depontieu, A. Scherpereel, C. Chopin, and R. Neviere. 2004. Calpain inhibitors improve myocardial dysfunction and inflammation induced by endotoxin in rats. Shock 21 (4): 352–357.
van Nieuwenhoven, F.A., and N.A. Turner. 2013. The role of cardiac fibroblasts in the transition from inflammation to fibrosis following myocardial infarction. Vascular Pharmacology 58 (3): 182–188. https://doi.org/10.1016/j.vph.2012.07.003.
Vieillard-Baron, A., V. Caille, C. Charron, G. Belliard, B. Page, and F. Jardin. 2008. Actual incidence of global left ventricular hypokinesia in adult septic shock. Critical Care Medicine 36 (6): 1701–1706. https://doi.org/10.1097/CCM.0b013e318174db05.
Wang, Y., J. Tian, H. Guo, Y. Mi, R. Zhang, and R. Li. 2016. Intermedin ameliorates IgA nephropathy by inhibition of oxidative stress and inflammation. Clinical and Experimental Medicine 16 (2): 183–192. https://doi.org/10.1007/s10238-015-0351-8.
Wu, R., S. Tang, M. Wang, X. Xu, C. Yao, and S. Wang. 2016. MicroRNA-497 induces apoptosis and suppresses proliferation via the Bcl-2/Bax-caspase9-caspase3 pathway and cyclin D2 protein in HUVECs. PLoS One 11 (12): e0167052. https://doi.org/10.1371/journal.pone.0167052.
Wu, Y., J. Ren, B. Zhou, C. Ding, J. Chen, G. Wang, G. Gu, et al. 2015. Gene silencing of non-obese diabetic receptor family (NLRP3) protects against the sepsis-induced hyper-bile acidaemia in a rat model. Clinical and Experimental Immunology 179 (2): 277–293. https://doi.org/10.1111/cei.12457.
Yang, S.M., J. Liu, and C.X. Li. 2014. Intermedin protects against myocardial ischemia-reperfusion injury in hyperlipidemia rats. Genetics and Molecular Research 13 (4): 8309–8319. https://doi.org/10.4238/2014.October.20.7.
Zhang, B., Y. Liu, Y.B. Sui, H.Q. Cai, W.X. Liu, M. Zhu, and X.H. Yin. 2015. Cortistatin inhibits NLRP3 Inflammasome activation of cardiac fibroblasts during sepsis. Journal of Cardiac Failure 21 (5): 426–433. https://doi.org/10.1016/j.cardfail.2015.01.002.
Zhang, B., Y. Liu, J.S. Zhang, X.H. Zhang, W.J. Chen, X.H. Yin, and Y.F. Qi. 2015. Cortistatin protects myocardium from endoplasmic reticulum stress induced apoptosis during sepsis. Molecular and Cellular Endocrinology 406: 40–48. https://doi.org/10.1016/j.mce.2015.02.016.
Zhang, J.S., Y.L. Hou, W.W. Lu, X.Q. Ni, F. Lin, Y.R. Yu, C.S. Tang, and Y.F. Qi. 2016. Intermedin1-53 protects against myocardial fibrosis by inhibiting endoplasmic reticulum stress and inflammation induced by homocysteine in apolipoprotein E-deficient mice. Journal of Atherosclerosis and Thrombosis 23 (11): 1294–1306. https://doi.org/10.5551/jat.34082.
Zhang, W., X. Xu, R. Kao, T. Mele, P. Kvietys, C.M. Martin, and T. Rui. 2014. Cardiac fibroblasts contribute to myocardial dysfunction in mice with sepsis: the role of NLRP3 inflammasome activation. PLoS One 9 (9): e107639. https://doi.org/10.1371/journal.pone.0107639.
Acknowledgments
This study is supported by the Research Foundation of the Aerospace Central Hospital (NO. YN201316 to Bin Wang) and the National Natural Sciences Foundation of China (NO. 81670434 to Yongfen Qi).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Wu, D., Shi, L., Li, P. et al. Intermedin1–53 Protects Cardiac Fibroblasts by Inhibiting NLRP3 Inflammasome Activation During Sepsis. Inflammation 41, 505–514 (2018). https://doi.org/10.1007/s10753-017-0706-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-017-0706-2