Abstract
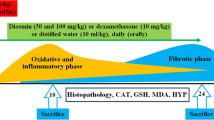
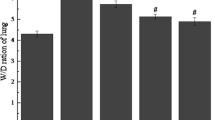
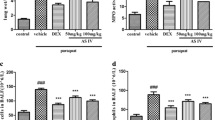
Methylsulfonylmethane (MSM) is a natural organosulfur compound that exhibits antioxidative and anti-inflammatory effects. This study was carried out to investigate the effect of MSM on paraquat (PQ)-induced acute lung and liver injury in mice. A single dose of PQ (50 mg/kg, i.p.) induced acute lung and liver toxicity. Mice were treated with MSM (500 mg/kg/day, i.p.) for 5 days. At the end of the experiment, animals were euthanized, and lung and liver tissues were collected for histological and biochemical analysis. Tissue samples were used to determine malondialdehyde (MDA), myeloperoxidase (MPO), catalase (CAT), superoxide dismutase (SOD), glutathione (GSH), and tumor necrosis factor-α (TNF-α) levels. Blood samples were used to measure plasma alanine transaminase (ALT), γ-glutamyl transferase (GGT), and alkaline phosphatase (ALP). Histological examination indicated that MSM decreased lung and liver damage caused by PQ. Biochemical results showed that MSM treatment significantly reduced tissue levels of MDA, MPO, and TNF-α, while increased the levels of SOD, CAT, and GSH compared with PQ group. MSM treatment also significantly reduced plasma levels of ALT, GGT, and ALP. These findings suggest that MSM as a natural product attenuates PQ-induced pulmonary and hepatic oxidative injury.







Similar content being viewed by others
References
Ishida, Y., T. Takayasu, A. Kimura, T. Hayashi, N. Kakimoto, T. Miyashita, and T. Kondo. 2006. Gene expression of cytokines and growth factors in the lungs after paraquat administration in mice. Legal Medicine 8: 102–109.
He, X., L. Wang, G. Szklarz, Y. Bi, and Q. Ma. 2012. Resveratrol inhibits paraquat-induced oxidative stress and fibrogenic response by activating the nuclear factor erythroid 2-related factor 2 pathway. Journal of Pharmacology and Experimental Therapeutics 342: 81–90.
Denicola, A., and R. Radi. 2005. Peroxynitrite and drug-dependent toxicity. Toxicology 208: 273–288.
Gawarammana, I.B., and N.A. Buckley. 2011. Medical management of paraquat ingestion. British Journal of Clinical Pharmacology 72: 745–757.
Parcell, S. 2002. Sulfur in human nutrition and applications in medicine. Alternative Medicine Review 7: 22–44.
Kim, L.S., L.J. Axelrod, P. Howard, N. Buratovich, and R.F. Waters. 2006. Efficacy of methylsulfonylmethane (MSM) in osteoarthritis pain of the knee: a pilot clinical trial. Osteoarthritis and Cartilage 14: 286–294.
Gregory, P.J., M. Sperry, and A.F. Wilson. 2008. Dietary supplements for osteoarthritis. American Family Physician 77: 177–184.
Childs, S.J. 1994. Dimethyl sulfone (DMSO2) in the treatment of interstitial cystitis. Urologic Clinics of North America 21: 85–88.
Marañón, G., B. Muñoz-Escassi, W. Manley, C. García, P. Cayado, M.S. de la Muela, B. Olábarri, R. León, and E. Vara. 2008. The effect of methyl sulphonyl methane supplementation on biomarkers of oxidative stress in sport horses following jumping exercise. Acta Veterinaria Scandinavica 50: 45.
Barmaki, S., S. Bohlooli, F. Khoshkhahesh, and B. Nakhostin-Roohi. 2012. Effect of methylsulfonylmethane supplementation on exercise-Induced muscle damage and total antioxidant capacity. The Journal of Sports Medicine and Physical Fitness 52: 170–174.
Caron, J.M., M. Bannon, L. Rosshirt, J. Luis, L. Monteagudo, and G.M. Sternstein. 2010. Methyl sulfone induces loss of metastatic properties and reemergence of normal phenotypes in a metastatic cloudman S-91 (M3) murine melanoma cell line. PLoS One 5: e11788.
Lim, E.J., D.Y. Hong, J.H. Park, Y.H. Joung, P. Darvin, S.Y. Kim, Y.M. Na, T.S. Hwang, S.K. Ye, and E.S. Moon. 2012. Methylsulfonylmethane suppresses breast cancer growth by down-regulating STAT3 and STAT5b pathways. PLoS One 7: e33361.
Amirshahrokhi, K., S. Bohlooli, and M.M. Chinifroush. 2011. The effect of methylsulfonylmethane on the experimental colitis in the rat. Toxicology and Applied Pharmacology 253: 197–202.
Nakhostin-Roohi, B., S. Barmaki, F. Khoshkhahesh, and S. Bohlooli. 2011. Effect of chronic supplementation with methylsulfonylmethane on oxidative stress following acute exercise in untrained healthy men. Journal of Pharmacy and Pharmacology 63: 1290–1294.
Mateos, R., E. Lecumberri, S. Ramos, L. Goya, and L. Bravo. 2005. Determination of malondialdehyde (MDA) by high-performance liquid chromatography in serum and liver as a biomarker for oxidative stress. Application to a rat model for hypercholesterolemia and evaluation of the effect of diets rich in phenolic antioxidants from fruits. Journal of Chromatography B Analytical Technologies in the Biomedical and Life Sciences 827: 76–82.
Dinis-Oliveira, R.J., F. Remião, J.A. Duarte, R. Ferreira, A. Sánchez Navarro, M.L. Bastos, and F. Carvalho. 2006. P-glycoprotein induction: an antidotal pathway for paraquat-induced lung toxicity. Free Radical Biology & Medicine 41: 1213–1224.
Giustarini, D., I. Dalle-Donne, R. Colombo, A. Milzani, and R. Rossi. 2003. An improved HPLC measurement for GSH and GSSG in human blood. Free Radical Biology & Medicine 35: 1365–1372.
Morán, J.M., M.A. Ortiz-Ortiz, L.M. Ruiz-Mesa, and J.M. Fuentes. 2010. Nitric oxide in paraquat-mediated toxicity: a review. Journal of Biochemical and Molecular Toxicology 24: 402–409.
Dragin, N., M. Smani, S. Arnaud-Dabernat, C. Dubost, I. Moranvillier, P. Costet, J.Y. Daniel, and E. Peuchant. 2006. Acute oxidative stress is associated with cell proliferation in the mouse liver. FEBS Letters 580: 3845–3852.
Zhi, Q., H. Sun, X. Qian, and L. Yang. 2011. Edaravone, a novel antidote against lung injury and pulmonary fibrosis induced by paraquat? International Immunopharmacology 11: 96–102.
Bus, J.S., S.D. Aust, and J.E. Gibson. 1976. Paraquat toxicity: proposed mechanism of action involving lipid peroxidation. Environmental Health Perspectives 16: 139–146.
Melchiorri, D., R.J. Reiter, E. Sewerynek, M. Hara, L. Chen, and G. Nisticò. 1996. Paraquat toxicity and oxidative damage. Reduction by melatonin. Biochemical Pharmacology 51: 1095–1099.
Dinis-Oliveira, R.J., J.A. Duarte, F. Remião, A. Sánchez-Navarro, M.L. Bastos, and F. Carvalho. 2006. Single high dose dexamethasone treatment decreases the pathological score and increases the survival rate of paraquat-intoxicated rats. Toxicology 227: 73–85.
Kim, Y.H., D.H. Kim, H. Lim, D.Y. Baek, H.K. Shin, and J.K. Kim. 2009. The anti-inflammatory effects of methylsulfonylmethane on lipopolysaccharide-induced inflammatory responses in murine macrophages. Biological & Pharmaceutical Bulletin 32: 651–656.
Novaes, R.D., R.V. Gonçalves, M.C. Cupertino, D.C. Marques, D.D. Rosa, C. Peluzio Mdo, C.A. Neves, and J.P. Leite. 2012. Bark extract of Bathysa cuspidata attenuates extra-pulmonary acute lung injury induced by paraquat and reduces mortality in rats. International Journal of Experimental Pathology 93: 225–233.
Novaes, R.D., R.V. Gonçalves, D.C. Marques, C. Cupertino Mdo, C. Peluzio Mdo, J.P. Leite, and I.R. Maldonado. 2012. Effect of bark extract of Bathysa cuspidata on hepatic oxidative damage and blood glucose kinetics in rats exposed to paraquat. Toxicologic Pathology 40: 62–70.
Suntres, Z.E. 2002. Role of antioxidants in paraquat toxicity. Toxicology 180: 65–77.
Acknowledgments
This study was supported by a grant from Vice Chancellor of Research of Ardabil University of Medical Sciences. We also wish to thank Dr. AR Khalili for technical assistance in histological analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amirshahrokhi, K., Bohlooli, S. Effect of Methylsulfonylmethane on Paraquat-Induced Acute Lung and Liver Injury in Mice. Inflammation 36, 1111–1121 (2013). https://doi.org/10.1007/s10753-013-9645-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-013-9645-8