Abstract
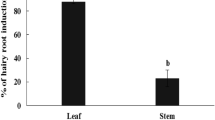
The flowers of Pyrethrum (Chrysanthemum cinerariaefolium) are known to contain Pyrethrins that are naturally occurring potential insecticide. Hairy roots were induced from leaves of C. cinerariaefolium using Agrobacterium rhizogenes strain A4. The root clones were characterized in to four groups i.e. thick, unbranched (D2 and D5), thin, highly branched (D3), thick, branched (B2) and thick, highly branched (D1, D6). Six established hairy root clones showed the presence of pyrethrin and were selected for elicitation studies. Growth kinetics studies revealed highest growth index in hairy root clone D1 (592.0) followed by D6 and D3 on dry weight basis after 40 days of culture. The maximum pyrethrin content was found in the clone D3 (7.2 mg/g dw) which is comparable to the flowers obtained from the variety “Avadh”. Hairy root clone D2 (5.2 mg/g dw) and D6 (1.3 mg/g dw) contained pyrethrin but in less amount as compared to clone D3. The PCR analysis showed the presence of rol B and rol C genes in all the six hairy root clones while rol A was detected only in D2 clone. The methanolic extract of D3 clone showed antifungal activities against phytopathogenic fungal strains which were found maximum against Curvuleria andropogonis followed by Colletotrichum acutatum and Rhizoctonia solani. Hairy root clones D2, D3 and D6 were elicited with culture filtrate of endophytic fungus (Fusarium oxysporum) and bacteria (Bacillus subtilis). The culture filtrate (4.0 %v/v) of both the fungal and bacterial origin was found to be effective in enhancing the pyrethrin content in all the tested hairy root clones. Clone D3 showed maximum pyrethrin content on elicitation with F. oxysporum (9.7 mg/g dw) and B. subtilis (9.7 mg/g dw) culture filtrate, which is 32 % higher than the non elicited D3 hairy roots (7.2 mg/g dw). F. oxysporum also enhanced the hairy root growth resulting into the higher biomass yield of D3 (50 %) and D2 (76 %) in comparison to control non elicited hairy root clones of D3 and D2, respectively leading to higher pyrethrin yield.






Similar content being viewed by others
References
Abhyankar G, Rao KV, Reddy VD (2013) Genomic and metabolomic fingerprinting of Phyllanthus amarus (Schumm & Thonn) hairy root clones. Ann Phytomed 2:74–88
Ahn JC, Chong WS, Kim YS, Hwang B (2006) Optimization of the sucrose and ion concentration for Saikosaponin production in hairy root cultures of Bupleurum falcatum. Biotechnol Bioproc E 11:121–126
Bisht C, Badoni A, Vashishtha RK, Nautiyal MC (2009) Photoperiodic effect on seed germination in Pyrethrum(Chrysanthemum cinerariaefolium vis.) under the influence of some growth regulators. J Am Sci 5:147–150
Bulgakov VP (2008) Functions of rol genes in plant secondary metabolism. Biotechnol Adv 26:318–324
Crawford DL, Lunch JM, Whipps JM, Ousley MA (1993) Isolation and characterization of actinomycetes antagonists of fungal root pathogen. Appl Environ Microbiol 59:3889
Dessalgne FH, Mekonnen SA, Idris BA (2011) Variability of Pyrethrum (Chrysanthemum cinerariaefolim) clone for chemical traits grown at Bekoji and Meraro of south eastern Ethiopia. Int J Med Arom Plants 1:166–174
Edwards A, Johnstone C, Thompson C (1991) A simple and rapid method for the preparation of plant genomic DNA for PCR analysis. Nucl Acid Res 19:1349
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soyabean root cells. Exp Cell Res 50:151–158
Gao Y, Zhao Y (2013) Epigenetic suppression of T-DNA insertion mutants in Arabidopsis. Mol Plant 6:539–545
Gennarelli MC, Hagiwara JC, Tosto D, Alvarez MA, Borja MA, Escandon AS (2009) Genetic transformation of Calibrachoa excellens via Agrobacterium rhizogenes: Changing morphological traits. J Hort Sci Biotechnol 84:305–311
Guillon S, Tremouillaux-Guiller J, Pati PK, Rideau M, Gantet P (2006) Hairy root research: recent scenario and exciting prospects. Curr Opin Plant Biol 9:341–346
Guillon S, Tremouillaux-Guiller J, Pati PK, Rideau M, Gantet P (2007) Harnessing the potential of hairy roots: dawn of a new era. Trends Biotechnol 24:403–409
Hedayat M, Abdi G, Khosh-Khui M (2009) Regeneration via direct organogenesis from leaf and petiole segments of Pyrethrum [Tanacetum cinerariifolium (Trevir.) Schultz-Bip.]. Am Eurasian J Agric Environ Sci 6:81–87
Hitmi A, Coudret A, Barthomeuf C (2000) The Production of Pyrethrins by plant cell and tissue cultures of Chrysanthemum cinerariaefolium and Tagetes species. Crit Rev Biochem Mol Biol 35:317–337
Hooykaas PJJ, Klapwijk PM, Nuti MP, Schilperoort RA, Rorsch A (1977) Transfer of the Agrobacterium tumefaciens Ti-plasmid to avirulent Agrobacteria and to Rhizobium explanta. J Gen Microbiol 98:477–482
Hu Z-B, Du M (2006) Hairy root and its application in plant genetic engineering. J Integ Plant Biol 48:121–127
Komarovska H, Kosuth J, Giovannini A, Smelcerovic A, Zuehlkec S, Cellarova E (2010) Effect of the number of rol genes integrations on phenotypic variation in hairy root-derived Hypericum perforatum L. plants. Z Naturforsch 65:701–712
Lebeda A, Svabova L (2010) In vitro screening methods for assessment of plant disease resistance. In: Spencer MM, Lebeda A. (eds) Mass screening techniques for selecting crops resistant to disease. IAEA, Vienna, pp 5–45
Matsuda K (2012) Pyrethrin Biosynthesis and Its Regulation in Chrysanthemum cinerariaefolium. Top Curr Chem 314:73–82
Metcalf RL, Metcalf RA (1993) Destructive and useful insects: their habits and control. McGraw-Hill, New York
Murashige T, Skoog F (1962) A revised medium of rapid growth and assays within Tobacco tissue cultures. Physiol Plant 15:473–497
Panda H (2005) Herbs cultivation and medicinal uses. Nat Ins Ind Res, Delhi, pp 1–9
Park SU, Lee SY (2009) Anthraquinone production by hairy root culture of Rubia akane Nakai: influence of media and auxin treatment. Sci Res Essay 4:690–693
Park NI, Li X, Uddin MR, Park SU (2011) Phenolic compound production by different morphological phenotypes in hairy root cultures of Fagopyrum tataricum Gaertn. Arch Biol Sci Belgrade 63:193–198
Picman AK (1983) Anti-fungal activity of helenin and isohelenin. Biochem Syst Ecol 11: 183–186
Ramirez AM, Saillard N, Yang T, Franssen MCR, Bouwmeester HJ, Jongsma MA (2013) Biosynthesis of Sesquiterpene Lactones in Pyrethrum (Tanacetum cinerariifolium). Plos One 8(5): e65030. doi:10.1371/journal.pone.0065030
Rudrappa T, Neelwarne B, Lakshmanan V, Venkataramareddy SR, Aswathanarayana RG (2006) Elicitation of peroxidase activity in genetically transformed root cultures of Beta vulgaris L. Electron. J Biotechnol 9:512–521
Savitha BC, Thimmaraju R, Bhagyalakshmi N, Ravishankar GA (2006) Different biotic and abiotic elicitorsinfluence betalain production in hairy root cultures of Beta vulgaris in shake-flask and bioreactor. Process Biochem 41:50–60
Shkryl YN, Veremeichik GN, Bulgakov VP, Tchernoded GK, Mischenko NP, Fedoreyev SA, Zhuravlev YN (2008) Individual and combined effects of the rol A, B, and C genes on anthraquinone production in Rubia cordifolia transformed calli. Biotechnol Bioeng 100:118–125
Slightom JL, Durand-Tardif M, Jouanin L, Tepfer D (1986) Nucleotide sequence analysis of TL-DNA of Agrobacterium rhizogenes agropine-type plasmid: identification of open reading frames. J Biol Chem 261:108–121
Svabova L, Lebeda A, Kitner M, Sedlarova M, Petrivalsky M, Dostalova R, Ondrej M, Horacek J, Smykalov I, Griga M (2011) Comparison of the effects of Fusarium solani filtrates in vitro and in vivo on the morphological characteristics and peroxidase activity in pea cultivars with different susceptibility. J Plant Pathol 93:19–30
Vasconsuelo A, Boland R (2007) Molecular aspects of the early stages of elicitation of secondary metabolites in plants. Plant Sci 172:861–875
Verma P, Mathur AK, Shanker K (2012) Growth, alkaloid production, rol genes integration, bioreactor up-scaling and plant regeneration studies in hairy root lines of Catharanthus roseus. Plant Biosyst 146:27–40
Verma P, Khan SA, Mathur AK, Ghosh S, Shanker K, Kalra A (2014) Fungal endophytes enhanced the growth and production kinetics of Vinca minor hairy roots and cell suspensions grown in bioreactor. Plant Cell Tissue Organ Cult 118:257–268
Wongwicha W, Tanaka H, Shoyama Y, Putalun W (2011) Methyl jasmonate elicitation enhances glycyrrhizin production in Glycyrrhiza inflate hairy roots cultures. Z Naturforsch 66:423–424
Zhao JT, Davisb LC, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23:283–333
Acknowledgments
We gratefully acknowledge the financial support received from the Council of Scientific & Industrial Research as CSIR SRF, Government of India, and New Delhi to carry out this work and Director CSIR-CIMAP for providing all the necessary facilities.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Khan, S.A., Verma, P., Banerjee, S. et al. Pyrethrin accumulation in elicited hairy root cultures of Chrysanthemum cinerariaefolium . Plant Growth Regul 81, 365–376 (2017). https://doi.org/10.1007/s10725-016-0213-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-016-0213-8