Abstract
The moss Physcomitrella patens is becoming the model of choice for functional genomic studies at the cellular level. Studies report that Physcomitrella survives moderate osmotic and salt stress, and that desiccation tolerance can be induced by exogenous ABA. Our goal was to quantify the extent of dehydration tolerance in wild type moss and to examine the nature of cellular damage caused by desiccation. We exposed Physcomitrella to humidities that generate water potentials from −4 (97% RH) to −273 MPa (13% RH) and monitored water loss until equilibrium. Water contents were measured on a dry matter basis to determine the extent of dehydration because fresh weights (FW) were found to be variable and, therefore, unreliable. We measured electrolyte leakage from rehydrating moss, assessed overall regrowth, and imaged cells to evaluate their response to drying and rehydration. Physcomitrella did not routinely survive water potentials <−13 MPa. Upon rehydration, moss dried to water contents >0.4 g g dm−1 maintained levels of leakage similar to those of hydrated controls. Moss dried to lower water contents leaked extensively, suggesting that plasma membranes were damaged. Moss protonemal cells were shrunken and their walls twisted, even at −13 MPa. Moss cells rehydrated after drying to −273 MPa failed to re-expand completely, again indicating membrane damage. ABA treatment elicited tolerance of desiccation to at least −273 MPa and limited membrane damage. Results of this work will form the basis for ongoing studies on the functional genomics of desiccation tolerance at the cellular level.
Similar content being viewed by others
Introduction
The moss Physcomitrella patens is rapidly becoming the model of choice for functional genomic studies of plants at the cellular level (Cove et al. 2006; Quatrano et al. 2007; Reski 1998). The morphology of this moss is relatively simple; the dominant haploid stage comprises protonemal filaments that differentiate into photosynthetic chloronema and non-photosynthetic caulonema. The filamentous cells are large (c. 50 to >100 μm in length) and easily observed with a light microscope. The protonema can give rise to erect gametophores with leaf-like phyllidia whose lamellae are a single cell-layer in thickness. The simplicity of morphology allows direct access to individual plant cells without the complicating factor of tissue level organization and barriers: it is this aspect that makes Physcomitrella particularly attractive as a model. Moreover, Physcomitrella is amenable to genetic transformation, and the high frequency of homologous recombination events permits targeted gene replacement (Schaefer 2001). The recently sequenced genome (Rensing et al. 2008) provides a wealth of information and opportunity for researchers.
One area of active research benefitting from the Physcomitrella model has been the unraveling of genes and genetic pathways involved in cellular dehydration tolerance. Physcomitrella is considered a mesic bryophyte (Wood 2007) and does not, under normal circumstances, tolerate desiccation, defined as equilibrium with a low atmospheric relative humidity (RH) (Bewley 1979) or equilibrium to water potentials less than about −100 MPa (Alpert 2005; Oliver 2007). Several reports show, however, that Physcomitrella colonies can recover from severe dehydration, reported as the loss of 80–92% of their fresh weight, after air-drying (Cuming et al. 2007; Frank et al. 2005) or exposure to osmotic and salt-stress (Frank et al. 2005; Saavedra et al. 2006). These studies have revealed a large number of transcripts that respond to dehydration, 130 that are up-regulated and 75 that are down-regulated. Within this population of transcripts, 52 (49 up- and 3 down-regulated) also respond to exogenous ABA (Cuming et al. 2007). These data are testament to the complexity of the cellular response to dehydration and underscore the need for a precise understanding of the water relations and limits of tolerance for this moss.
A recent paper (Wang et al. 2009) claims that Physcomitrella is desiccation tolerant; the basis for this assertion is that the moss, grown and assessed on an agar medium, survived the loss of 90% of its fresh weight after lengthy exposure to air in a chamber containing silica gel. Defining Physcomitrella as desiccation tolerant contradicts previous reports that the moss cannot survive extensive water loss (Cuming et al. 2007; Frank et al. 2005; Saavedra et al. 2006), nor can it survive equilibration to 35% RH (Oldenhof et al. 2006). Desiccation tolerant cells survive the loss of all bulk water, plus the loss of interfacial water associated with macromolecular surfaces; this generally occurs at water contents less than about 0.1–0.3 g g dm−1 (dry matter) (Alpert 2005; Hoekstra et al. 2001), although the precise value depends upon the composition of the cells (Walters and Koster 2007). Given these standard definitions, the description of Physcomitrella as desiccation tolerant (Wang et al. 2009) merits closer investigation.
Although most previous studies have shown that Physcomitrella is sensitive to desiccation, one study (Oldenhof et al. 2006) demonstrated that application of exogenous abscisic acid (ABA) for 1 week at a concentration of 100 μM can induce desiccation tolerance, including survival of equilibration at 35% RH (−140 MPa). At this RH, the protonema were reported to lose 99.8% of their fresh weight, corresponding to a water content of 0.02 g g dm−1 (Oldenhof et al. 2006). Moreover, Oldenhof and colleagues reported that ABA treatment altered membrane phase properties in the moss, supporting the idea that membrane protection is one result of ABA-induced gene expression. In other moss species, ABA treatment has been linked with improved membrane stability during desiccation and with the accumulation of specific proteins (Beckett 1999; Bopp and Werner 1993; Guschina et al. 2002; Werner et al. 1991). The hypothesis that an exogenous ABA treatment elicits a cellular protection mechanism has been confirmed and expanded upon in two recent publications (Khandelwal et al. 2010; Pressel and Duckett 2010).
Given the importance of Physcomitrella as a new model for cellular level studies of stress responses, our goals were to quantify the extent of dehydration tolerance in this species and to examine the nature of cellular damage caused by desiccation. We show that Physcomitrella does not survive equilibration to atmospheric water potentials lower than about −13 MPa and, therefore, cannot be considered desiccation tolerant. Plasma membrane damage results from lethal desiccation, as indicated by the extensive leakage of electrolytes from the rehydrated moss and failure of the plasma membrane and plastids to return to their previous positions within the cell. Exposure to exogenous ABA prior to drying induces both desiccation tolerance, measured as survival of equilibration to 13% RH (−273 MPa), and membrane protective mechanisms. Results of this work will form the basis for ongoing studies on the functional genomics of desiccation tolerance at the cellular level.
Materials and methods
Physcomitrella patens ssp. patens (Hedwig) ‘Gransden’ was kindly provided by Ralph Quatrano and David Cove and grown on cellophane discs atop solid BCDA medium as described by Cove et al. (2009). Moss cultures were maintained at 24°C with a 16 h photoperiod at 100 μmol m−2 s−1 and were used between 4 and 6 weeks after subculture. At this time, the moss cultures comprised both protonemal filaments and gametophores. For ABA treatments prior to drying, moss on cellophane was transferred to fresh BCDA medium containing 50 μM cis–trans ABA. To minimize tissue damage during handling, the moss was left on its cellophane substratum during all assays other than dry weight determination.
Dehydration
Excess water was wicked away from moss-covered cellophane using tissue paper; the moss-covered cellophane then was cut into sections approximately 2 cm in diameter using sterile scissors. These sections were transferred into pre-weighed plastic petri dishes (3.5 cm diameter) and quickly weighed to obtain the fresh weight of the moss plus cellophane. The petri dishes were sealed in chambers containing atmospheres of known RH, generated by saturated salt solutions, and these were placed in an incubator at 17°C with a 16 h light, 8 h dark cycle. The RH used, corresponding water potential values at 17°C, and the salt solutions used to obtain them were: 97% (−4 MPa), K2SO4; 95% (−7 MPa), KNO3; 91% (−13 MPa), MgSO4; 86% (−20 MPa), KCl; 75% (−39 MPa), NaCl; 68% (−52 MPa), NH4NO3; 33% (−149 MPa), MgCl2; and 13% (−273 MPa), LiCl (Rockland 1960; Sun 2002; Winston and Bates 1960). Petri dishes containing moss samples were weighed daily until there was no further change in mass, indicating that the moss had equilibrated to the RH within the chamber. Samples were then tested for survival, electrolyte leakage, and water content, using triplicate samples for each.
Survival
Dehydrated moss on cellophane was placed on fresh BCDA medium and held in darkness for 24 h at 24°C as the moss absorbed water from the medium. The rehydrated moss was then exposed to light and monitored for 7 days. Survival was scored as the maintenance of green colour by the moss tissues and the beginning of further protonemal growth. In some instances, survival of the colonies was patchy; when about 30–70% of the colony on the cellophane became chlorotic, samples were scored as 50% survival. If less than about 30% of the sample remained green, it was scored as dead, even if the remaining green patches could eventually regenerate new moss. We used this scoring system because our interest is in studying mechanisms of dehydration tolerance at the cellular, rather than the organismal level.
Electrolyte leakage
Petri dishes containing dehydrated moss on cellophane were placed in a chamber lined with wet paper towels to permit absorption of water vapor for 2 h prior to the leakage assay and so to limit possible damage caused by the rapid influx of water into the completely dry cells. Four mL of deionized water were then added gently to each sample, and the cellophane inverted to submerge the moss. Samples were swirled at low speed (40 rpm) on an orbital shaker for 15 min, then the bathing solution was transferred to a test tube and conductivity measured using a Mettler Toledo Seven Easy conductivity meter. Preliminary experiments showed that 15 min was sufficient to bypass the initial rapid increase in conductivity, believed to arise primarily from extracellular electrolytes; thus, leakage at 15 min fell within the linear phase of leakage that reflects membrane permeability. Total electrolyte content for each sample was determined by adding the moss back to its bathing solution and freezing the tissue at −80°C to disrupt the cellular membranes. Samples were warmed to room temperature and agitated vigourously with a vortex mixer, and the conductivity measured again. Leakage is expressed as the percent of total electrolytes that had leaked into the bathing solution during the initial 15 min exposure.
Water content
After achieving a constant weight at a given RH, dehydrated moss tissue was quickly scraped from its cellophane substrate and transferred into a pre-weighed aluminum dish. Samples were weighed to obtain the fresh weight of the dehydrated tissue, and oven dry weight was measured after drying 48 h at 70°C. Water content, expressed on a dry matter (dm) basis, was calculated as the difference between the fresh weight and the oven dry weight, divided by the oven dry weight.
Microscopy
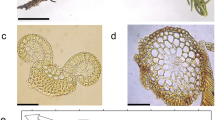
Physcomitrella was observed prior to drying (fresh controls), after dehydrating at 91% and 13% RH, and after rehydration. Dehydrated samples were removed carefully from the cellophane and placed on a microscope slide for viewing. Images were collected quickly to limit the exposure of the sample to the ambient RH of the room. Samples were rehydrated by wicking liquid water on the slide to the edge of the sample, then allowing water to move by capillary action into the sample. Materials were observed in situ using an Olympus (Center Valley, PA USA) IX71 inverted light microscope with epifluorescence and D.I.C. capability and imaged digitally. Micrographs of entire gametophores were imaged at 10X using a Leica MZFLIII (Heerbrugg, Switzerland) stereomicroscope (Fig. 4a, brightfield). Protonemal images were captured at 100X using either brightfield (Fig. 4b) or D.I.C. (Fig. 5a–e).
Results
Physcomitrella samples lost water when exposed to atmospheric RH 97% and lower. The dehydration rate was rapid for moss exposed to the lowest RH; samples at 13% RH (−273 MPa) equilibrated within 24 h, while moss held at RH above 86% (−20 MPa) took more than 4 d to equilibrate (Fig. 1a). Water loss was monitored by recording the fresh weights of the samples until no further change was noted; for samples used in experiments assessing viability and leakage, final weights ranged from ca. 14–30% of the initial weight (Fig. 1a). These weights include the cellophane substrate, kept to minimize handling damage to the moss, and so cannot be compared directly to other published reports in which water loss from Physcomitrella is reported as a percentage of the initial FW. In some experiments, replicate samples were dried without their cellophane substrate to determine whether the cellophane changed the drying rate. As seen in Fig. 1b, the presence of the cellophane did not significantly affect the drying rate for samples at the same RH; the lines more or less parallel one another. Moss samples without cellophane that equilibrated to atmospheres with water potentials from −4 to −52 MPa lost between about 85–95% of their fresh weight, corresponding to water contents between 1.9 and 0.23 g g dm−1 (Fig. 1c). The relationship between the extent of water loss on a fresh weight basis and the water content on a dry matter basis differed significantly between experiments (data not shown), reflecting differences in water contents of fully hydrated moss. These occurred because of variation in the amount of surface water coating the protonemal filaments and gametophores. When excess water was wicked carefully away from the cellophane with blotting paper, the water contents of the moss (taken after removal from the cellophane) ranged from 11 to 23 g g dm−1 and depended on the density of the protonema. Therefore, using the loss of fresh weight to describe the extent of cellular dehydration was deemed misleading, and thus uninformative, so water contents on a dry matter basis are used in subsequent experiments.
Water loss from Physcomitrella exposed to atmospheres of controlled RH. Water contents during dehydration are expressed as a percentage of the initial fresh weight of each sample. Moss samples were routinely dried on cellophane (a). Drying rates were compared for replicate samples dried either with or without cellophane (b). Water contents on fresh weight and dry matter bases were compared for moss dried without cellophane (c). Data shown are means ± SD of three to six replicate samples. Error bars are not shown where they are smaller than the symbol
Physcomitrella routinely survived mild dehydration to RH of 95% or greater (water potentials >−7 MPa), as indicated by maintenance of green color and resumption of growth when returned to moist BCDA medium (Fig. 2a). Water contents of samples at 95% RH averaged 0.64 g g dm−1 (Fig. 2b). When equilibrated at 91% RH (−13 MPa), colonies exhibited more variable response: half were able to survive, with at least 50% of the sample retaining green colour throughout the 7 day recovery period; while others became chlorotic upon rehydration. The mean water content of non-ABA-treated controls at 91% RH was 0.32 g g dm−1 and did not vary significantly between samples that survived and those that died (Fig. 2b). Physcomitrella equilibrated to RH less than 91% became almost completely chlorotic upon rehydration, indicating that this moss is not tolerant of extensive desiccation. However, when pre-treated with 50 μM ABA prior to drying, Physcomitrella survived equilibration to 13% RH (−273 MPa), the lowest value tested in these experiments (Fig. 2a).
Dehydration tolerance of Physcomitrella dried by equilibration with RH from 13 to 97%. Overall survival at each RH was scored as the percentage of samples that had ≥50% survival when tested in seven independent experiments (a). Water contents attained by Physcomitrella at each RH were calculated on a dry matter basis (mean ± SEM) from mean water contents measured in seven independent experiments (b). Water contents were grouped and averaged according to sample survival. Individual samples were judged as having survived (>70% remained green, diamond), about 50% survival (circle), or died (<30% remained green, down triangle). ABA-treated samples (square) survived at all RH tested. Error bars are not shown where they are smaller than the symbol
Plasma membrane damage, measured as electrolyte leakage from rehydrating samples, occurred in Physcomitrella that did not survive dehydration (Fig. 3). Replicates of moss samples that lost more than 65% of their total electrolytes upon rehydration failed to maintain their green color and resume growth; this was observed in moss samples dried to water contents less than approximately 0.4 g g dm−1 (Fig. 3). Fresh moss samples leached between 21 and 27% of their total electrolyte content, even without drying (data not shown); these salts are presumed to originate primarily from the extracellular solution coating the protonema. Pre-treatment with ABA significantly decreased the leakage from dried moss during rehydration, even from moss dried to water contents as low as 0.05 g g dm−1 (Fig. 3). ABA pre-treatment also enabled Physcomitrella equilibrated to −52 MPa (68% RH) or higher to retain more water than untreated controls during drying (P < 0.01, Fig. 2b). At −273 MPa (13% RH), differences in water content between ABA-treated and control moss were not apparent; all samples reached water contents less than 0.1 g g dm−1 (Fig. 2b).
Electrolyte leakage from Physcomitrella upon rehydration. Leakage is expressed as a percentage of the total conductivity of the sample. Each data point represents the leakage (mean ± SD) from at least three replicate samples in a single experiment; data from five independent experiments are shown. Water contents were determined for three replicate samples from the same experiment (mean ± SD). Error bars are not shown where they are smaller than the symbol
Microscopic examination revealed that Physcomitrella colonies 4–6 weeks after subculture contained both gametophores and filamentous protonemata (Fig. 4). Prior to drying, protonemal cells appeared turgid, with numerous green plastids around the periphery of the cell (Fig. 4b). When Physcomitrella samples were dehydrated at either 13% (Fig. 5a) or 91% RH (Fig. 5b) there were similar gross morphological alterations in cell morphology and organelle displacement. Protonemal cells were flattened and highly twisted. The cytoplasm was concentrated, sometimes near the end walls and sometimes nearer to the midpoints. In many cases, thin strands of cytoplasm and plastids also could be discerned along the peripheral edges of the flattened cells. When material dried at 13% RH (−273 MPa) was rehydrated, protonemal filaments quickly filled with fluid and the cell walls returned to a cylindrical orientation. However, the plasma membrane in many cases failed to realign with the cell walls and had a plasmolyzed appearance (Fig. 5c). Additionally, plastids were indistinct and the cytoplasm appeared dense (Fig. 5c). Plant material rehydrated after drying at 91% RH (−13 MPa) revealed differential cell morphology and degree of organelle reconstitution. Many filaments rapidly rehydrated and appeared indistinguishable from fresh controls, with plastids located around the cell periphery, suggesting that a central vacuole was present (Fig. 5d). However, the appearance of protonemata was variable. After rehydration, cells at and near the tip of chloronemal filaments often appeared indistinguishable from fresh controls, while cells distal from the tips often revealed a disorganized internal structure and a dense cytoplasm (Fig. 5e).
Discussion
Bryophytes in general are tolerant of a certain amount of cellular dehydration. The morphology of their leaves and filaments does not allow for much in the way of water retention and thus they require efficient capillary driven transport of water and liquid water on their surfaces to maintain growth and efficient photosynthesis. Once liquid water is removed from the surfaces, cells quickly lose water and equilibrate to the water potential of the air. Since this is a common occurrence in many bryophyte habitats, it is clear that most species have developed some means of surviving periods, however short, of dehydration (reviewed by Oliver 2009). Many grow in mesic habitats and can only survive mild dehydration; others grow in more challenging habitats and can survive prolonged periods of dehydration to levels where all bulk water is lost from their tissues. The latter are considered desiccation tolerant. Many species, whether desiccation sensitive or tolerant, have evolved a clump growth strategy as a means of retaining liquid water for as long as possible in drying atmospheres (Proctor 1980; Rice et al. 2001).
Desiccation sensitivity in bryophytes has been loosely defined as the ability to survive equilibration with air no drier than between 80 and 70% RH, which equates to maximum limits of dehydration to water potentials of −30 to −48 MPa (Wood 2007). Although this is considered desiccation sensitive for a bryophyte, the level of dehydration tolerance in these mosses is far beyond that reported for most angiosperms, which can rarely survive −4 to −6 MPa (Oliver 2007). Desiccation tolerant bryophytes (as well as their angiosperm counterparts) can survive equilibration to dry atmospheres, 50% RH and below, that generate water potentials less than −100 MPa (Alpert and Oliver 2002; Wood 2007). Given the uncertainty as to where Physcomitrella fits into this classification, it was important to establish its level of dehydration tolerance in order to make sense of the growing physiological, biochemical, and molecular data relating to the response of this moss to water loss.
Studies intended to elucidate cellular mechanisms of dehydration tolerance depend upon reproducible methods of dehydration and accurate definitions of the hydration status of the tissues. One of our goals was to establish a baseline data set defining the limits of dehydration tolerance by Physcomitrella. Most previous studies of this species have found that colonies can survive some dehydration brought about by exposure to ambient air of unknown RH (Frank et al. 2005) or to air at 75% RH (Cuming et al. 2007), although the moss did not survive equilibrium to either atmosphere. Therefore, we set out to quantify the extent of tolerance in terms of the equilibrium RH, or atmospheric water potential, survived by cells of Physcomitrella. Direct measurement of the water potential of the plant is difficult, and below −10 MPa almost impossible with current instrumentation. However, the equilibrium atmospheric water potential by definition is a surrogate measurement. As shown in Fig. 2a, Physcomitrella was able to survive equilibrium with 95% RH (−7 MPa) and, in many instances, with 91% RH (−13 MPa). The moss was routinely killed by equilibrium to RH 86% (−20 MPa) and lower (Fig. 2). The colonies used in these experiments had grown 4–6 weeks after subculturing and contained both protonemata and gametophores. In some experiments with older colonies (7–10 weeks after subculturing), patchy survival was observed after equilibration to RH as low as 68% (−52 MPa), although survival was never great at RH below 86%. This suggests that the age and associated density of the colony contributed to tolerance of lengthy exposure to low RH. These data demonstrate that at least the lab strain of Physcomitrella, under normal conditions, is not capable of surviving dehydration that results in sustained water potentials of less than ca. −13 MPa, which clearly places it amongst the desiccation-sensitive bryophytes. It is possible and probable that Physcomitrella growing in its natural habitat is more dehydration tolerant, perhaps reaching the levels seen in other desiccation sensitive bryophytes (−30 to −48 MPa).
The patchy survival of some colonies at RH less than 95%, or water potentials below −7 MPa, also illustrates the need to distinguish between cellular and organismal tolerance of dehydration. From an ecological perspective, an organism, organ, or tissue may be judged desiccation tolerant if it can regenerate new cells upon rehydration; this despite the fact that the majority of cells in the tissue may be irreparably damaged by the desiccation stress. Partial survival may result from differences in desiccation sensitivity among tissues within the same organism or from differences in the actual extent of water loss among cells or tissues. Physcomitrella, like many bryophytes, can regenerate new cells from damaged tissue, allowing stressed colonies to recover by replacing the damaged cells (e.g. Frank et al. 2005; Saavedra et al. 2006). Moreover, when ageing, stressed or treated with ABA, Physcomitrella forms dehydration tolerant reproductive cells at the tips of protonemal filaments; these brood cells or brachycytes can regenerate new colonies (Goode et al. 1993; Pressel and Duckett, 2010; Rowntree et al. 2007). Our observations confirm that the tip cells of Physcomitrella protonemata generally appeared to regain an organized internal structure upon rehydration, even when many or most of the other cells in the same sample appeared disorganized (Fig. 5e). Although definitions of dehydration tolerance at the organismal level that include the ability to regenerate from specialized cells or structures are important for ecological inferences, they should not be confused with definitions of desiccation tolerance that are directed at understanding how plant vegetative cells tolerate dehydration.
The relationship between survival and water content can be seen in Fig. 2, which shows that moss that dried to water contents less than ca. 0.3 g g dm−1 did not routinely survive unless pre-treated with ABA. Water contents in this study are expressed on a dry matter basis, which reflects how much water remains to solvate and separate cellular contents, rather than the extent of water lost from the original hydrated tissue. In contrast, most previous studies of the dehydration tolerance of Physcomitrella have reported water loss in terms of fresh weight (Cuming et al. 2007; Frank et al. 2005; Pressel and Duckett 2010; Saavedra et al. 2006; Wang et al. 2009), which is common for measures of drought tolerance, but which may be less useful for studies of moss, whose fresh weight may include significant, but variable, amounts of surface water. In our studies, the initial water content of the colonies varied extensively; therefore, water losses corresponding to 90% of the fresh weight could leave from 0.2 to 2 g g dm−1 water remaining in the sample (e.g. Fig. 1c).
High rates of electrolyte leakage were observed from colonies that did not survive drying (Fig. 3), indicating that plasma membrane damage is a component of dehydration damage. Similarly high leakage rates have been reported for other bryophytes damaged by drying (e.g. Beckett 1999, 2001; Brown and Buck 1979; Oliver et al. 1993; Schonbeck and Bewley 1981) and were reported for Physcomitrella patens damaged by freezing (Minami et al. 2003; Nagao et al. 2006). In those studies, researchers observed that the extensive leakage from lethally frozen cells was associated with ultrastructural changes in plasma membranes, including the formation of aparticulate domains, fracture jump lesions, and regions of non-bilayer hexagonal II phase (Nagao et al. 2005). These types of membrane damage arise when membranes are brought into close apposition during cellular dehydration (Koster and Bryant 2006) and are often seen in cells damaged by freezing, when extracellular ice draws water out of cells (Steponkus et al. 1993; Uemura and Steponkus 2003). When plasma membrane integrity is compromised in this way, cells typically become unresponsive to osmotic changes (Steponkus et al. 1993; Uemura and Steponkus 2003). The failure of Physcomitrella protoplasts to re-expand with their walls when rehydrated after drying at −273 MPa (Fig. 5c) further suggests that plasma membrane semipermeability was compromised by lethal desiccation. In contrast, protonemal cells rehydrated after drying at −13 MPa (91% RH) appeared to have osmotically functional plasma membranes (Figs. 5d, e), although some cells exhibited dense cytoplasm, suggesting that metabolic damage, and perhaps precipitation of some cytosolic constituents, had occurred. These observations support the hypothesis that membranes are an important site of dehydration damage in Physcomitrella, as they are in most organisms (e.g. Hoekstra et al. 2001; Koster and Bryant 2006; Walters et al. 2002).
Membranes may be lethally damaged by desiccation, but they are not the only vulnerable structures within cells. Pressel and Duckett (2010) recently published a detailed study on the effects of dehydration on the anatomy of protonemata from several mosses. They show that controlled cytoskeletal disassembly, changes in organellar morphology, and reorganization of endomembranes are key to survival of dehydration. These changes can be brought about by slow drying (2–3 days) in some species, or by the application of exogenous ABA (Pressel and Duckett 2010). Pressel and Duckett (2010) confirm that Physcomitrella does not survive slow drying, but does survive dehydration to 10% of its initial water content after ABA-treatment. They also noted the twisting of flattened protonemata that we observed in Physcomitrella dehydrated to water potentials ca. −13 MPa and lower.
ABA-treated Physcomitrella in our study was tolerant of desiccation at −273 MPa (13% RH) (Fig. 2a), which led to water contents of 0.075 g g dm−1 (Fig. 2b). Similar observations have been documented for other desiccation-sensitive mosses such as Atrichum androgynum (Beckett 1999, 2001) and Funaria hygrometrica (Werner et al. 1991). ABA-treatment prior to dehydration led to significantly decreased leakage from rehydrated cells, indicating that membrane-protective mechanisms had been induced in the moss by ABA. A similar effect was measured in the moss A. androgynum after ABA treatment (Beckett 2001). Previous studies with Physcomitrella found that ABA treatments affected both the thermodynamic and ultrastructural properties of membranes. ABA treatment conferred desiccation tolerance and prevented increases in the fluid-gel phase transition temperature of dried Physcomitrella membranes; untreated controls had elevated transition temperatures and failed to survive desiccation (Oldenhof et al. 2006). Moreover, plasma membranes of ABA-treated and, therefore, freezing-tolerant cells did not have any of the ultrastructural lesions that had been observed in freeze-damaged cells (Nagao et al. 2005). These studies indicate that ABA treatments confer protection to membranes via mechanisms that prevent dehydrative damage. One protective mechanism, observed in both studies, is the accumulation of sucrose and other non-reducing sugars, which can act as osmotic spacers between membranes and other cellular structures that are drawn together as water exits cells. By limiting the close approach of these structures, sugars can minimize hydration forces that otherwise cause physical damage to membranes, including membrane phase changes and fusions that can cause leakage of the internal solutes from the cell (Bryant et al. 2001; Koster and Bryant 2006; Koster et al. 2000).
Another potentially protective feature induced by exogenous ABA is the synthesis of various proteins that may strengthen cellular glasses that form as the viscosity of intracellular solutions rises during dehydration (Oldenhof et al. 2006). ABA treatment triggers the expression of numerous genes in Physcomitrella, including those of several homologs to stress proteins that accumulate in other plants as part of the dehydration response (Cuming et al. 2007; Machuka et al. 1999). Included among these are two dehydrin-like proteins, one of which has been demonstrated to play a role in osmotic and salinity tolerance in Physcomitrella (Saavedra et al. 2006). Recently, Khandelwal et al. (2010) demonstrated that the Physcomitrella homolog of the Arabidopsis gene Abscisic acid Insensitive 3 (ABI3) is required for desiccation tolerance to be induced by the application of ABA. As in wild-type Physcomitrella, exogenous ABA induced the expression of 22 ABA up-regulated genes during desiccation of moss in which the ABI3 homolog had been removed by gene targeting. However, the same genes were dramatically down-regulated during the rehydration phase compared to ABA-treated wild-type levels. This result reiterates the importance of the rehydration phase of a wet-dry-wet cycle in the overall mechanism of dehydration tolerance in bryophytes (Oliver 2009).
It must be pointed out here that Physcomitrella patens is desiccation sensitive in its natural habitat and completes its life cycle as an “annual” during relatively wet periods (Mishler, personal communication). It does survive moderate dehydration, but when desiccated it relies on spore regeneration to recolonize its habitat. Although it is clear that ABA is important in the dehydration tolerance of this moss, it is also empirically clear that the response of the moss to an increase in endogenous ABA is insufficient to render the moss desiccation tolerant. That the moss becomes desiccation tolerant when exogenous ABA is added in relatively large amounts indicates that during evolution, the sensitivity of Physcomitrella to ABA has diminished, if one ascribes to the hypothesis that primitive plants were desiccation tolerant when they colonized the land.
References
Alpert P (2005) The limits and frontiers of desiccation-tolerant life. Integr Comp Biol 45:685–6951
Alpert P, Oliver MJ (2002) Drying without dying. In: Black M, Pritchard H (eds) Desiccation and survival in plants: drying without dying. CAB International Press, Wallingford, pp 3–46
Beckett RP (1999) Partial dehydration and ABA induce tolerance to desiccation-induced ion leakage in the moss Atrichum androgynum. S Afr J Bot 65:1–6
Beckett RP (2001) ABA-induced tolerance to ion leakage during rehydration following desiccation in the moss Atrichum androgynum. J Plant Growth Reg 35:131–135
Bewley JD (1979) Physiological aspects of desiccation-tolerance. Ann Rev Plant Physiol 30:195–238
Bopp M, Werner O (1993) Abscisic acid and desiccation tolerance in mosses. Bot Acta 106:103–106
Brown DH, Buck GW (1979) Desiccation effects and cation distribution in bryophytes. New Phytol 82:115–125
Bryant G, Koster KL, Wolfe J (2001) Membrane behaviour in seeds and other systems at low water content: the various effects of solutes. Seed Sci Res 11:17–25
Cove DJ, Bezanilla M, Harries P, Quatrano RS (2006) Mosses as model systems for the study of metabolism and development. Ann Rev Plant Biol 57:497–520
Cove DJ, Perroud PF, Charron AJ, McDaniel SF, Khandelwal A, Quatrano RS (2009) Culturing the moss Physcomitrella patens. Cold Spring Harb Protoc. doi: 10.1101/pdb.prot5136
Cuming AC, Cho SH, Kamisugi Y, Graham H, Quatrano RS (2007) Microarray analysis of transcriptional responses to abscisic acid and osmotic, salt, and drought stress in the moss, Physcomitrella patens. New Phytol 176:275–287
Frank W, Ratnadewi D, Reski R (2005) Physcomitrella patens is highly tolerant against drought, salt and osmotic stress. Planta 220:384–394
Goode JA, Stead AD, Duckett JG (1993) Redifferentiation of moss protonemata: an experimental and immunofluorescence study of brood cell formation. Can J Bot 74:726–734
Guschina IA, Harwood JL, Smith M, Beckett RP (2002) Abscisic acid modifies the changes in lipids brought about by water stress in the moss Atrichum androgynum. New Phytol 156:255–264
Hoekstra FA, Golovina EA, Buitink J (2001) Mechanisms of plant desiccation tolerance. Trends Plant Sci 6:431–438
Khandelwal A, Cho SH, Marella H, Sakata Y, Perroud P-F, Pan A, Quatrano RS (2010) Role of ABA and ABI3 in desiccation tolerance. Science 29:327–546
Koster KL, Bryant G (2006) Dehydration in model membranes and protoplasts: contrasting effects at low, intermediate and high hydrations. In: Chen THH, Uemura M, Fujikawa S (eds) Cold hardiness in plants. CAB International Press, Wallingford, pp 219–234
Koster KL, Lei YP, Anderson M, Martin S, Bryant G (2000) The effect of vitrified and nonvitrified sugars on phosphatidylcholine fluid-to-gel phase transitions. Biophys J 78:1932–1946
Machuka J, Bashiardes S, Ruben E, Spooner K, Cuming A, Knight C, Cove D (1999) Sequence analysis of expressed sequence tags from an ABA-treated cDNA library identifies stress response genes in the moss Physcomitrella patens. Plant Cell Physiol 40:378–387
Minami A, Nagao M, Arakawa K, Fujikawa S, Takezawa D (2003) Abscisic acid-induced freezing tolerance in the moss Physcomitrella patens is accompanied by increased expression of stress-related genes. J Plant Physiol 160:475–483
Nagao M, Minami A, Arakawa K, Fujikawa S, Takezawa D (2005) Rapid degradation of starch in chloroplasts and concomitant accumulation of soluble sugars associated with ABA-induced freezing tolerance in the moss Physcomitrella patens. J Plant Physiol 162:169–180
Nagao M, Oku K, Minami A, Mizuno K, Sakurai M, Arakawa K, Fujikawa S, Takezawa D (2006) Accumulation of theanderose in association with development of freezing tolerance in the moss Physcomitrella patens. Phytochem 67:702–709
Oldenhof H, Wolkers WF, Bowman JL, Tablin F, Crowe JH (2006) Freezing and desiccation tolerance in the moss Physcomitrella patens: an in situ Fourier transform infrared spectroscopic study. Biochim Biophys Acta 1760:1226–1234
Oliver MJ (2007) Lessons on dehydration tolerance from desiccation tolerant plants. In: Jenks MA, Wood AJ (eds) Plant desiccation tolerance. CAB International Press, Wallingford, pp 11–50
Oliver MJ (2009) Biochemical and molecular mechanisms of desiccation tolerance in bryophytes. In: Goffinet B, Shaw J (eds) Bryophyte biology, 2nd edn. Cambridge University Press, New York, pp 269–298
Oliver MJ, Mishler BD, Quisenberry JE (1993) Comparative measures of desiccation-tolerance in the Tortula ruralis complex. I. Variation in damage control and repair. Am J Bot 80:127–136
Pressel S, Duckett JG (2010) Cytological insights into the desiccation biology of a model system: moss protonemata. New Phytol 185:944–963
Proctor MCF (1980) Diffusion resistance in bryophytes. In: Grace J, Ford ED, Jarvis PG (eds) Plants and their atmospheric environment. Blackwell Scientific, Oxford, pp 219–229
Quatrano RS, McDaniel SF, Khandelwal A, Perroud P-F, Cove DJ (2007) Physcomitrella patens: mosses enter the genomic age. Curr Opin Plant Biol 10:182–189
Rensing SA, Lang D, Zimmer AD, Terry A, Salamov A, Shapiro H, Nishiyama T, Perroud PF, Lindquist EA, Kamisugi Y, Tanahashi T, Sakakibara K, Fujita T, Oishi K, Shin IT, Kuroki Y, Toyoda A, Suzuki Y, Hashimoto S, Yamaguchi K, Sugano S, Kohara Y, Fujiyama A, Anterola A, Aoki S, Ashton N, Barbazuk WB, Barker E, Bennetzen JL, Blenkenship R, Cho SH, Dutcher SK, Estelle M, Fawcett JA, Gundlach H, Hanada K, Heyl A, Hicks KA, Hughes J, Lohr M, Mayer K, Melkozernov A, Murata T, Nelson DR, Pils B, Prigge M, Reiss B, Renner T, Rombauts S, Rushton PJ, Sanderfoot A, Schween G, Shiu SH, Stueber K, Theodoulou FL, Tu H, Van de Peer Y, Verrier PJ, Waters E, Wood A, Yang LX, Cove DJ, Cuming AC, Hasebe M, Lucas S, Mishler BD, Reski R, Grigoriev IV, Quatrano RS, Boore JL (2008) The Physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 319:64–69
Reski R (1998) Physcomitrella and Arabidopsis: the David and Goliath of reverse genetics. Trends Plant Sci 3:209–210
Rice SK, Collins D, Anderson AM (2001) Functional significance of variation in bryophyte canopy structure. Am J Bot 88:1568–1576
Rockland LB (1960) Saturated salt solutions for static control of relative humidity between 5°C and 40°C. Anal Chem 32:1375–1376
Rowntree JK, Duckett JG, Mortimer CL, Ramsay MM, Pressel S (2007) Formation of specialized propagules resistant to desiccation and cryopreservation in the threatened moss Ditrichum plumbicola (Ditrichales, Bryopsida). Ann Bot 100:483–496
Saavedra L, Svensson J, Carballo V, Izmendi D, Welin B, Vidal S (2006) A dehydrin gene in Physcomitrella patens is required for salt and osmotic stress tolerance. Plant J 45:237–249
Schaefer DG (2001) Gene targeting in Physcomitrella patens. Curr Opin Plant Biol 4:143–150
Schonbeck MW, Bewley JD (1981) Responses of the moss Tortula ruralis to desiccation treatments. I. Effects of minimum water content and rates of dehydration and rehydration. Can J Bot 59:2698–2706
Steponkus PL, Uemura M, Webb MS (1993) A contrast of the cryostability of the plasma membrane of winter rye and spring oat–two species that differ widely in their freezing tolerance and plasma membrane lipid composition. In: Steponkus PL (ed) Advances in low temperature biology, vol 2. JAI Press, London, pp 211–312
Sun WQ (2002) Methods for the study of water relations under desiccation stress. In: Black M, Pritchard H (eds) Desiccation and survival in plants: drying without dying. CAB International Press, Wallingford, pp 47–92
Uemura M, Steponkus PL (2003) Modification of the intracellular sugar content alters the incidence of freeze-induced membrane lesions of protoplasts isolated from Arabidopsis thaliana leaves. Plant Cell Environ 26:1083–1096
Walters C, Koster KL (2007) Structural dynamics and desiccation damage in plant reproductive organs. In: Jenks MA, Wood AJ (eds) Plant desiccation tolerance. CAB International Press, Wallingford, pp 251–280
Walters C, Farrant JM, Pammenter NW, Berjak P (2002) Desiccation stress and damage. In: Black M, Pritchard H (eds) Desiccation and survival in plants: drying without dying. CAB International Press, Wallingford, pp 263–291
Wang XQ, Yang PF, Liu Z, Liu WZ, Hu Y, Chen H, Kuang TY, Peo ZM, Shen SH, He YK (2009) Exploring the mechanism of Physcomitrella patens desiccation tolerance through a proteomic strategy. Plant Physiol 149:1739–1750
Werner O, Espin RMR, Bopp M, Atzorn R (1991) Abscisic acid-induced drought tolerance in Funaria hygrometrica Hedw. Planta 186:99–103
Winston PW, Bates DH (1960) Saturated solutions for the control of humidity in biological research. Ecol 41:232–237
Wood AJ (2007) The nature and distribution of vegetative desiccation-tolerance in hornworts, liverworts and mosses. Bryologist 110:163–177
Acknowledgments
We thank Dr. G. Esteban Fernandez and Stephanie Boyle of the Molecular Cytology Core at the University of Missouri for assistance with microscopy and Jim Elder of the USDA-ARS for general lab support. This work was supported by grants from the USDA National Institute of Food and Agriculture to KLK (grant no. 2008-35100-19240), MJO (grant no. 2007-55100-18374), and RAB (grant no. 2008-35100-04413).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Koster, K.L., Balsamo, R.A., Espinoza, C. et al. Desiccation sensitivity and tolerance in the moss Physcomitrella patens: assessing limits and damage. Plant Growth Regul 62, 293–302 (2010). https://doi.org/10.1007/s10725-010-9490-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-010-9490-9