Abstract
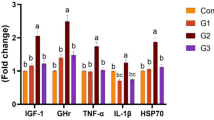
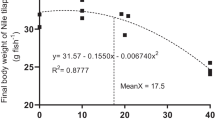
Fucoidans are marine algal sulfated glycans that are widely used as dietary additives in aquaculture. These glycans are recognized as beneficial supplements for their antimicrobial, anti-inflammatory, anticancer, and antiviral properties. Potassium permanganate is another commonly used chemical that is used in aquaculture to treat infections in fish. Despite their widespread use, there are few data available regarding the potential sublethal toxicity associated with fucoidan and potassium permanganate treatments of fish. In this study, we investigated the effect of each compound on the growth, intestinal health, and antioxidant status of Nile tilapia (Oreochromis niloticus). Both compounds affected the growth of experimental fish compared with untreated fish. However, while growth parameters were positively associated with the dose of fucoidan administered, growth was negatively associated with the dose of potassium permanganate in Nile tilapia. Fucoidan treatment was observed to improve the intestinal health of fish based upon increases in intestinal villous area, intestinal villous length and width, and the intraepithelial lymphocyte number and decreases in the total intestinal bacterial count compared with untreated fish. Conversely, potassium permanganate induced intestinal epithelium proliferation and villous branching, a histopathological response typically observed with chemical irritants. Both fucoidan and potassium permanganate decreased levels of oxidative and nitrosative stress markers and enhanced the antioxidant status in multiple organs. Taken together, fucoidan dietary application improved the growth, intestinal health, and antioxidant status in Nile tilapia, supporting the use of this compound as a promising feed additive for aquaculture production. Conversely, potassium permanganate baths have negative effects on fish growth at higher doses and appeared to act as a gastrointestinal irritant in tilapia. This study improves knowledge regarding the biochemical and histological responses in Nile tilapia to two widely used aquaculture-related treatments.










Similar content being viewed by others
References
Abdel-Tawwab M, Hagras A, Elbaghdady H, Monier M (2015) Effects of dissolved oxygen and fish size on Nile tilapia, Oreochromis niloticus (L.): growth performance, whole-body composition, and innate immunity. Aquac Int 23. https://doi.org/10.1007/s10499-015-9882-y
Aguilar-Briseño JA, Cruz-Suarez LE, Sassi J-F, Ricque-Marie D, Zapata-Benavides P, Mendoza-Gamboa E, Rodríguez-Padilla C, Trejo-Avila LM (2015) Sulphated polysaccharides from Ulva clathrata and Cladosiphon okamuranus seaweeds both inhibit viral attachment/entry and cell-cell fusion, in NDV infection. Mar Drugs 13:697–712. https://doi.org/10.3390/md13020697
Aleissa MS, Alkahtani S, Abd Eldaim MA, Ahmed AM, Bungău SG, Almutairi B, Bin-Jumah M, AlKahtane AA, Alyousif MS, Abdel-Daim MM (2020) Fucoidan ameliorates oxidative stress, inflammation, DNA damage, and hepatorenal injuries in diabetic rats intoxicated with aflatoxin B1. In: Oxidative Medicine and Cellular Longevity. https://www.hindawi.com/journals/omcl/2020/9316751/. Accessed 29 Jun 2020
Alejandro M-SD, Catherine C-DI, María V-SY (2019) Biochemical responses and proximate analysis of Piaractus brachypomus (Pisces: Characidae) exposed to phenanthrene. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology 108649 . https://doi.org/10.1016/j.cbpc.2019.108649
Al-Harbi A, Siddiqui AQ (2000) Effects of tilapia stocking densities on fish growth and water quality in tanks. Asian Fish Sci 13
Asase A, Acolatse E, Apraku A, Darko J (2019) Potential for the culture of Tilapia guineensis (Bleeker, 1862): a comparative growth performance with Nile tilapia (Oreochromis niloticus) in a fresh water pond. J Fish Coast Mgt 1:20. https://doi.org/10.5455/jfcom.20190401124703
Bagenal T (1978) Methods for the assessment of fish production in fresh waters, 3 edition. Blackwell Science Inc, Oxford Eng. : Philadelphia
Balta F, Kayis S, Altinok I (2008) External protozoan parasites in three trout species in the Eastern Black Sea region of the Turkey: intensity, seasonality, and their treatments. Bull Eur Assoc Fish Pathol 28:157–162
Besednova NN, Zaporozhets TS, Somova LM, Kuznetsova TA (2015) Review: prospects for the use of extracts and polysaccharides from marine algae to prevent and treat the diseases caused by Helicobacter pylori. Helicobacter 20:89–97. https://doi.org/10.1111/hel.12177
Burdon RH, Gill V, Rice-Evans C (1989) Cell proliferation and oxidative stress. Free Radic Res Commun 7:149–159. https://doi.org/10.3109/10715768909087937
Che N, Ma Y, Xin Y (2016) Protective role of fucoidan in cerebral ischemia-reperfusion Injury through inhibition of MAPK signaling pathway. Biomol Ther 25. https://doi.org/10.4062/biomolther.2016.098
Chen J, Chen Q, Lu X-J, Chen J (2016) The protection effect of LEAP-2 on the mudskipper (Boleophthalmus pectinirostris) against Edwardsiella tarda infection is associated with its immunomodulatory activity on monocytes/macrophages. Fish Shellfish Immunol 59:66–76. https://doi.org/10.1016/j.fsi.2016.10.028
Choi YYH, Hosseindoust A, Goel A, Lee SH, Jha PK, Kwon IK, Chae B-J (2016) Effects of Ecklonia cava as fucoidan-rich algae on growth performance, nutrient digestibility, intestinal morphology and caecal microflora in weanling pigs. Asian-Australasian journal of animal sciences, In
Crespi V, New M (2009) Oreochromis niloticus. FAO. Cultured aquatic species fact sheets
Done HY, Venkatesan AK, Halden RU (2015) Does the recent growth of aquaculture create antibiotic resistance threats different from those associated with land animal production in agriculture? AAPS J 17:513–524. https://doi.org/10.1208/s12248-015-9722-z
El-Boshy M, El-Ashram A, Risha E, Abdelhamid F, Zahran E, Gab-Alla A (2014) Dietary fucoidan enhance the non-specific immune response and disease resistance in African catfish, Clarias gariepinus, immunosuppressed by cadmium chloride. Vet Immunol Immunopathol 162:168–173. https://doi.org/10.1016/j.vetimm.2014.10.001
Elliott JM (1975) The growth rate of brown trout (Salmo trutta L.) fed on maximum rations. J Anim Ecol 44:805–821. https://doi.org/10.2307/3720
El-Sayed A-FM (2006) Tilapia culture. CABI Pub. Wallingford, UK ; Cambridge, MA
Eun-Jeong Kwon I-HP, Kim M-M (2011) Fucoidan with antioxidant effect on DPPH and hydroxyl radical modulates matrix metalloproteinase-9 in HT1080 cell line. Cancer Prev Res 16:33–40
Eurell JAC, Frappier BL, Dellmann H-D (2006) Dellmann’s textbook of veterinary histology. Wiley
Fabrini BC, Fern W, Braga E, Andrade ES, Paula DA de J, Paulino RR, Costa AC, Paiva LV, da SFL, Murgas LDS (2017) Sulfated polysaccharides in diets for Nile tilapia (Oreochromis niloticus) in the initial growth phase. Journal of Aquaculture Research & Development 8(1–6). https://doi.org/10.4172/2155-9546.1000477
França JG, Ranzani-Paiva MJT, Lombardi JV, de Carvalho S, Filipak-Neto F, Oliveira-Ribeiro CA (2013) Toxic effect of potassium permanganate on Oreochromis niloticus based on hematological parameters and biomarkers of oxidative stress. Int J Fish Aquac 5:1–6. https://doi.org/10.5897/IJFA12.050
Gao W, Guo Y, Wang L, Jiang Y, Liu Z, Lin H (2020) Ameliorative and protective effects of fucoidan and sodium alginate against lead-induced oxidative stress in Sprague Dawley rats. Int J Biol Macromol 158:662–669. https://doi.org/10.1016/j.ijbiomac.2020.04.192
Hakalahti-Sirén T, Mikheev VN, Valtonen ET (2008) Control of freshwater fish louse Argulus coregoni: a step towards an integrated management strategy. Dis Aquat Org 82:67–77. https://doi.org/10.3354/dao01971
Han Y, Lee JH, Lee SH (2015a) Antitumor effects of fucoidan on human colon cancer cells via activation of Akt signaling. Biomol Ther (Seoul) 23:225–232. https://doi.org/10.4062/biomolther.2014.136
Han Y-S, Lee JH, Lee SH (2015b) Fucoidan inhibits the migration and proliferation of HT-29 human colon cancer cells via the phosphoinositide-3 kinase/Akt/mechanistic target of rapamycin pathways. Mol Med Rep 12:3446–3452. https://doi.org/10.3892/mmr.2015.3804
Hobbs MS, Grippo RS, Farris JL, Griffin BR, Harding LL (2006) Comparative acute toxicity of potassium permanganate to nontarget aquatic organisms. Environ Toxicol Chem 25:3046–3052. https://doi.org/10.1897/05-453R2.1
Isnansetyo A, Fikriyah A, Kasanah N, Murwantoko (2016a) Non-specific immune potentiating activity of fucoidan from a tropical brown algae (Phaeophyceae), Sargassum cristaefolium in tilapia (Oreochromis niloticus). Aquac Int 24:465–477. https://doi.org/10.1007/s10499-015-9938-z
Isnansetyo A, Lutfia F, Nursid M, Trijoko T, Susidarti R (2016b) Cytotoxicity of fucoidan from three tropical brown algae against breast and colon cancer cell lines. Pharm J 9:14–20. https://doi.org/10.5530/pj.2017.1.3
Jones TC, Hunt RD, King NW (1997) Veterinary pathology, 6th edn. Williams & Wilkins, Baltimore, Md. ; London
Kaer LV, Olivares-Villagómez D (2018) Development, homeostasis, and functions of intestinal intraepithelial lymphocytes. J Immunol 200:2235–2244. https://doi.org/10.4049/jimmunol.1701704
Kamaruzzaman N, Nguyen NH, Hamzah A, Ponzoni RW (2009) Growth performance of mixed sex, hormonally sex reversed and progeny of YY male tilapia of the GIFT strain, Oreochromis niloticus. Aquac Res 40:720–728. https://doi.org/10.1111/j.1365-2109.2008.02152.x
Keihani R (2016) The effect of Laminaria hyperborea and its bioactive components on the intestinal health of Atlantic salmon. Norwegian University of Life Sciences, Master’s Thesis
Kim K-W, Kim S-S, Khosravi S, Rahimnejad S, Lee K-J (2014a) Evaluation of Sargassum fusiforme and Ecklonia cava as dietary additives for olive flounder (Paralichthys olivaceus). Turk J Fish Aquat Sci 14:321–330. https://doi.org/10.4194/1303-2712-v14_2_03
Kim M-J, Jeon J, Lee J-S (2014b) Fucoidan prevents high-fat diet-induced obesity in animals by suppression of fat accumulation. Phytother Res 28:137–143. https://doi.org/10.1002/ptr.4965
Klein JA, Ackerman SL (2003) Oxidative stress, cell cycle, and neurodegeneration. J Clin Invest 111:785–793. https://doi.org/10.1172/JCI200318182
Lange KW, Hauser J, Nakamura Y, Kanaya S (2015) Dietary seaweeds and obesity. Food Sci Human Wellness 4:87–96. https://doi.org/10.1016/j.fshw.2015.08.001
Li Y, Zhao W, Wang L, Chen Y, Zhang H, Wang T, Yang X, Xing F, Yan J, Fang X (2019) Protective effects of fucoidan against hydrogen peroxide-induced oxidative damage in porcine intestinal epithelial cells. Animals (Basel) 9. https://doi.org/10.3390/ani9121108
Liang Z, Liu Z, Sun X, Tao M, Xiao X, Yu G, Wang X (2019) The effect of fucoidan on cellular oxidative stress and the CatD-Bax signaling axis in MN9D cells damaged by 1-methyl-4-phenypyridinium. Front Aging Neurosci 10. https://doi.org/10.3389/fnagi.2018.00429
Lim JD, Lee SR, Kim T, Jang S-A, Kang SC, Koo HJ, Sohn E, Bak JP, Namkoong S, Kim HK, Song IS, Kim N, Sohn E-H, Han J (2015) Fucoidan from Fucus vesiculosus protects against alcohol-induced liver damage by modulating inflammatory mediators in mice and HepG2 cells. Mar Drugs 13:1051–1067. https://doi.org/10.3390/md13021051
Lobo G, Pereira LF, Gonçalves JFM, Peixoto MJ, Ozório ROA (2018) Effect of dietary seaweed supplementation on growth performance, antioxidant and immune responses in European seabass (Dicentrarchus labrax) subjected to rearing temperature and salinity oscillations. Int Aquat Res 10:321–331. https://doi.org/10.1007/s40071-018-0208-3
Mamun S, Das K, Uddin M (2017) Microbiological analysis and determination of antibacterial activity of apple samples collected from local markets in Dhaka city, Bangladesh. Stamford J Microbiol 6:11. https://doi.org/10.3329/sjm.v6i1.33511
McCullough JS, Ratcliffe B, Mandir N, Carr KE, Goodlad RA (1998) Dietary fibre and intestinal microflora: effects on intestinal morphometry and crypt branching. Gut 42:799–806. https://doi.org/10.1136/gut.42.6.799
Mengistu SB, Mulder HA, Benzie JAH, Komen H (2020) A systematic literature review of the major factors causing yield gap by affecting growth, feed conversion ratio and survival in Nile tilapia (Oreochromis niloticus). Rev Aquac 12:524–541. https://doi.org/10.1111/raq.12331
Mitchell AJ, Darwish A, Fuller A (2008) Comparison of tank treatments with copper sulfate and potassium permanganate for sunshine bass with ichthyobodosis - Mitchell - 2008 - Journal of Aquatic Animal Health - Wiley Online Library. J Aquat Anim Health 20:202–206. https://doi.org/10.1577/H07-048.1
Mohammed HH, Arias CR (2015) Potassium permanganate elicits a shift of the external fish microbiome and increases host susceptibility to columnaris disease. Vet Res 46. https://doi.org/10.1186/s13567-015-0215-y
Nagamine T, Nakazato K, Tomioka S, Iha M, Nakajima K (2015) Intestinal absorption of fucoidan extracted from the brown seaweed, Cladosiphon okamuranus. Marine Drugs 13:48–64. https://doi.org/10.3390/md13010048
Oh J-Y, Kim E-A, Kang SI, Yang H-W, Ryu B, Wang L, Lee J-S, Jeon Y-J (2020) Protective effects of fucoidan isolated from celluclast-assisted extract of Undaria pinnatifida sporophylls against AAPH-induced oxidative stress in vitro and in vivo zebrafish model. Molecules 25. https://doi.org/10.3390/molecules25102361
Peixoto MJ, Salas-Leitón E, Pereira LF, Queiroz A, Magalhães F, Pereira R, Abreu H, Reis PA, Gonçalves JFM, de Ozório ROA (2016) Role of dietary seaweed supplementation on growth performance, digestive capacity and immune and stress responsiveness in European seabass (Dicentrarchus labrax). Aquac Rep 3:189–197. https://doi.org/10.1016/j.aqrep.2016.03.005
Peng Y, Song Y, Wang Q, Hu Y, He Y, Ren D, Wu L, Liu S, Cong H, Zhou H (2019) In vitro and in vivo immunomodulatory effects of fucoidan compound agents. Int J Biol Macromol 127:48–56. https://doi.org/10.1016/j.ijbiomac.2018.12.197
Pereira L (2011) A review of the nutrient composition of selected edible seaweeds. In: Seaweed: Ecology, Nutrient Composition and Medicinal Uses, pp 15–47
Picón-Camacho SM, Marcos-Lopez M, Bron JE, Shinn AP (2012) An assessment of the use of drug and non-drug interventions in the treatment of Ichthyophthirius multifiliis Fouquet, 1876, a protozoan parasite of freshwater fish. Parasitology 139:149–190. https://doi.org/10.1017/S0031182011001867
Pomin VH (2015) Sulfated glycans in inflammation. Eur J Med Chem 92:353–369. https://doi.org/10.1016/j.ejmech.2015.01.002
Purbomarto C, Isnansetyo A, . Murwantoko and Triyanto (2019) Dietary fucoidan from Padina boergesenii to enhance non-specific immune of catfish (Clarias sp.). J Biol Sci 19:173–180 . https://doi.org/10.3923/jbs.2019.173.180
Rintamäki-Kinnunen P, Rahkonen M, Mannermaa-Keränen A, Suomalainen L, Mykrä H, Valtonen E (2005) Treatment of ichthyophthiriasis after malachite green. I. Concrete tanks at salmonid farms. Dis Aquat Org 64:69–76. https://doi.org/10.3354/dao064069
Ryu MJ, Chung HS (2016) Fucoidan reduces oxidative stress by regulating the gene expression of HO-1 and SOD-1 through the Nrf2/ERK signaling pathway in HaCaT cells. Mol Med Rep 14:3255–3260. https://doi.org/10.3892/mmr.2016.5623
Sanchez-Martínez JG, Pérez-Castañeda R, Rábago-Castro JL, Aguirre-Guzmán G, Vázquez-Sauceda ML (2008) A preliminary study on the effects on growth, condition, and feeding indexes in channel catfish, Ictalurus punctatus , after the prophylactic use of potassium permanganate and oxytetracycline. J World Aquacult Soc 39:664–670. https://doi.org/10.1111/j.1749-7345.2008.00195.x
Schlenk D, Colley WC, El-Alfy A, Kirby R, Griffin BR (2000) Effects of the oxidant potassium permanganate on the expression of gill metallothionein mRNA and its relationship to sublethal whole animal endpoints in channel catfish. Toxicol Sci 54:177–182. https://doi.org/10.1093/toxsci/54.1.177
Schneider T, Ehrig K, Liewert I, Alban S (2015) Interference with the CXCL12/CXCR4 axis as potential antitumor strategy: superiority of a sulfated galactofucan from the brown alga Saccharina latissima and Fucoidan over heparins. Glycobiology 25:812–824. https://doi.org/10.1093/glycob/cwv022
Setyawan A, Isnansetyo A, Murwantoko M, Soedarmanto I, Handayani CR (2018) Comparative immune response of dietary fucoidan from three Indonesian brown algae in white shrimp Litopenaeus vannamei. AACL Bioflux 11:1707–1723
Shaw D, Gohil K, Basson MD (2012) Intestinal mucosal atrophy and adaptation. World J Gastroenterol 18:6357–6375. https://doi.org/10.3748/wjg.v18.i44.6357
Shi H, Chang Y, Gao Y, Wang X, Chen X, Wang Y, Xue C, Tang Q (2017) Dietary fucoidan of Acaudina molpadioides alters gut microbiota and mitigates intestinal mucosal injury induced by cyclophosphamide. Food Funct 8:3383–3393. https://doi.org/10.1039/C7FO00932A
Simon CMFRCP (2013) Underwood’s pathology: a clinical approach. In: 6 edition. Churchill Livingstone, Edinburgh
Sivagnanavelmurugan M, Ramnath GK, Thaddaeus BJ, Palavesam A, Immanuel G (2015) Effect of Sargassum wightii fucoidan on growth and disease resistance to Vibrio parahaemolyticus in Penaeus monodon post-larvae. Aquac Nutr 21:960–969. https://doi.org/10.1111/anu.12217
Smith SM, Nager RG, Costantini D (2016) Meta-analysis indicates that oxidative stress is both a constraint on and a cost of growth. Ecol Evol 6:2833–2842. https://doi.org/10.1002/ece3.2080
Sony NM, Ishikawa M, Hossain MS, Koshio S, Yokoyama S (2019) The effect of dietary fucoidan on growth, immune functions, blood characteristics and oxidative stress resistance of juvenile red sea bream, Pagrus major. Fish Physiol Biochem 45:439–454. https://doi.org/10.1007/s10695-018-0575-0
Stone N, Thomforde H (2003) Understanding your fish pond water analysis report
Stone N, Shelton JL, Haggard BE, Thomforde HK (2013) Interpretation of water analysis reports for fish culture. Southern Regional Aquaculture Center SRAC Publication No 4606:12
Straus DL, Griffin BR (2001) Prevention of an initial infestation of Ichthyophthirius multifiliis in channel catfish and blue tilapia by potassium permanganate treatment. N Am J Aquac 63:11–16. https://doi.org/10.1577/1548-8454(2001)063<0011:POAIIO>2.0.CO;2
Straus DL, Griffin BR (2002) Efficacy of potassium permanganate in treating ichthyophthiriasis in channel catfish. J Aquat Anim Health 14:145–148. https://doi.org/10.1577/1548-8667(2002)014<0145:EOPPIT>2.0.CO;2
Tengdelius M, Gurav D, Konradsson P, Påhlsson P, Griffith M, Oommen OP (2015) Synthesis and anticancer properties of fucoidan-mimetic glycopolymer coated gold nanoparticles. Chem Commun 51:8532–8535. https://doi.org/10.1039/C5CC02387D
Thomas-Jinu S, Goodwin AE (2004) Acute columnaris infection in channel catfish, Ictalurus punctatus (Rafinesque): efficacy of practical treatments for warmwater aquaculture ponds. J Fish Dis 27:23–28. https://doi.org/10.1046/j.1365-2761.2003.00504.x
Tieman DM, Goodwin AE (2001) Treatments for ich infestations in channel catfish evaluated under static and flow-through water conditions. N Am J Aquac 63:293–299. https://doi.org/10.1577/1548-8454(2001)063<0293:TFIIIC>2.0.CO;2
Tomori M, Nagamine T, Miyamoto T, Iha M (2019) Evaluation of the immunomodulatory effects of fucoidan derived from Cladosiphon okamuranus tokida in mice. Mar Drugs. https://doi.org/10.3390/md17100547
Traifalgar R, Serrano A Jr, Corre V, Kira H, Tung H, Michael F, Kader M, Laining A, Yokoyama S, Ishikawa M, Koshio S (2009) Evaluation of dietary fucoidan supplementation effects on growth performance and vibriosis resistance of Penaeus monodon postlarvae. Aquaculture Science 57
Trejo-Avila LM, Morales-Martínez ME, Ricque-Marie D, Cruz-Suarez LE, Zapata-Benavides P, Morán-Santibañez K, Rodríguez-Padilla C (2014) In vitro anti-canine distemper virus activity of fucoidan extracted from the brown alga Cladosiphon okamuranus. Virusdisease 25:474–480. https://doi.org/10.1007/s13337-014-0228-6
Tuller J, Santis CD, Jerry DR (2014) Dietary influence of fucoidan supplementation on growth of Lates calcarifer (Bloch). Aquac Res 45:749–754. https://doi.org/10.1111/are.12029
Vetvicka V, Vetvickova J (2017) Fucoidans stimulate immune reaction and suppress cancer growth. Anticancer Res 37:6041–6046
Vignesh R, Bagavathy Shanmugam K, Periyasamy N, Devanathan K (2011) Antibiotics in aquaculture: an overview. South Asian J Exp Biol 3
Vinay KMMFRCP, Abul KAM, Jon CAMP (2017) Robbins basic pathology, 10 edition. In: Elsevier. Pennsylvania, Philadelphia
Wang Y, Xing M, Cao Q, Ji A, Liang H, Song S (2019) Biological activities of fucoidan and the factors mediating its therapeutic effects: a review of recent studies. Marine Drugs 17:183. https://doi.org/10.3390/md17030183
Xue M, Ji X, Liang H, Liu Y, Wang B, Sun L, Li W (2018) The effect of fucoidan on intestinal flora and intestinal barrier function in rats with breast cancer. Food Funct 9:1214–1223. https://doi.org/10.1039/C7FO01677H
Yang Q, Yang R, Li M, Zhou Q, Liang X, Elmada ZC (2014) Effects of dietary fucoidan on the blood constituents, anti-oxidation and innate immunity of juvenile yellow catfish (Pelteobagrus fulvidraco). Fish Shellfish Immunol 41:264–270. https://doi.org/10.1016/j.fsi.2014.09.003
Yoo HJ, You D-J, Lee K-W (2019) Characterization and immunomodulatory effects of high molecular weight fucoidan fraction from the sporophyll of Undaria pinnatifida in cyclophosphamide-induced immunosuppressed mice. Mar Drugs 17. https://doi.org/10.3390/md17080447
Yu S-H, Wu S-J, Wu J-Y, Wen D-Y, Mi F-L (2015) Preparation of fucoidan-shelled and genipin-crosslinked chitosan beads for antibacterial application. Carbohydr Polym 126:97–107. https://doi.org/10.1016/j.carbpol.2015.02.068
Zhang W, Oda T, Yu Q, Jin J-O (2015) Fucoidan from Macrocystis pyrifera has powerful immune-modulatory effects compared to three other fucoidans. Mar Drugs 13:1084–1104. https://doi.org/10.3390/md13031084
Zhang T, Yang Y, Liang Y, Jiao X, Zhao C (2018) Beneficial effect of intestinal fermentation of natural polysaccharides. Nutrients 10. https://doi.org/10.3390/nu10081055
Acknowledgment
The authors wish to thank Dr. Marwa Ahmed (Department of Animal Hygiene and Zoonotic Diseases, Faculty of Veterinary Medicine, Mansoura University, Egypt) and Dr. Amr Abd El-Wahab (Department of Animal Nutrition, Faculty of Veterinary Medicine, Mansoura University, Egypt) for their help in the fish purchase and fish diet design, respectively. This study was executed as a collaboration between Pathology Department, Department of Biochemistry and Chemistry of Nutrition, and Department of Animal Husbandry (Faculty of Veterinary Medicine, Mansoura University, Egypt) and Centre for Environmental and Human Toxicology, Department of Physiological Sciences, College of Veterinary Medicine, University of Florida.
Funding
This project was partly funded by the Binational Fulbright Commission in Egypt and Mansoura University, as a postdoctoral research visit at the Centre of Environmental and Human Toxicology, Department of Physiological Sciences, College of Veterinary Medicine, University of Florida, awarded in July 2018 and executed between February and November 2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The experiments were conducted under the Guide for the Care and Use of Laboratory Animals approved by the Ethics Committee of the Faculty of Veterinary Medicine, Mansoura University.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hebatallah A. Mahgoub is previously affiliated with Center for Environmental and Human Toxicology, Department of Physiological Sciences, College of Veterinary Medicine, University of Florida
Rights and permissions
About this article
Cite this article
Mahgoub, H.A., El-Adl, M.A.M., Ghanem, H.M. et al. The effect of fucoidan or potassium permanganate on growth performance, intestinal pathology, and antioxidant status in Nile tilapia (Oreochromis niloticus). Fish Physiol Biochem 46, 2109–2131 (2020). https://doi.org/10.1007/s10695-020-00858-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-020-00858-w