Abstract
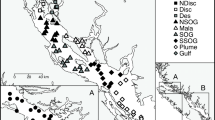
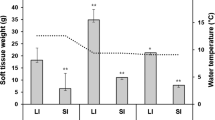
Chum salmon (Oncorhynchus keta) migrate to the ocean in their first spring, and growth during early marine life is critical for survival. We examined the validity of circulating IGF-I and muscle RNA/DNA ratio as indices of growth rate using individually tagged juvenile chum salmon fed or fasted for 10 days. Serum IGF-I level was highly, positively correlated with individual growth rate. Muscle RNA/DNA ratio also showed a positive correlation, but its relation was not as high as that of IGF-I. We next measured these physiological parameters in chum salmon juveniles caught at river, estuary, port and nearshore of the northeastern Hokkaido, Japan, from May to June in 2013 and 2014, respectively. In both years, there was a trend that serum IGF-I levels were high in nearshore fish and low in river/estuarine fish in June. In contrast, muscle RNA/DNA ratio showed no clear temporal and spatial patterns. The present study shows that circulating IGF-I can be used as a growth index in juvenile chum salmon. Monitoring growth status using serum IGF-I suggests that growth of juvenile chum salmon in the survey area was activated when they left the coast.








Similar content being viewed by others
References
Arndt SKA, Benfey TJ, Cunjak RA (1996) Effect of temporary reductions in feeding on protein synthesis and energy storage of juvenile Atlantic salmon. J Fish Biol 49:257–276
Bax NJ (1980) Early marine mortality of marked juvenile chum salmon (Oncorhynchus keta) released into Hood Canal, Puget Sound, Washington, in 1980. Can J Fish Aquat Sci 40:426–435
Beamish RJ, Mahnken C (2001) A critical size and period hypothesis to explain natural regulation of salmon abundance and the linkage to climate and climate change. Prog Oceanogr 49:423–437
Beamish RJ, Mahnken C, Neville CM (2004) Evidence that reduced early marine growth is associated with lower marine survival of coho salmon. Trans Am Fish Soc 133:26–33
Beaudreau AH, Andrews KS, Larsen DA, Young G, Beckman BR (2011) Variation in plasma levels of insulin-like growth factor-I (IGF-I) in lingcod: relationships among season, size, and gonadal steroids. Mar Biol 158:439–450
Beckman BR (2011) Perspectives on concordant and discordant relations between insulin-like growth factor 1 (IGF1) and growth in fishes. Gen Comp Endocrinol 170:233–252
Beckman BR, Larsen DA, Moriyama S, Lee-Pawlak B, Dickhoff WW (1998) Insulin-like growth factor-I and environmental modulation of growth during smoltification of spring chinook salmon (Oncorhynchus tshawytscha). Gen Comp Endocrinol 109:325–335
Beckman BR, Shearer KD, Cooper KA, Dickhoff WW (2001) Relationship of insulin-like growth factor-I and insulin to size and adiposity of under-yearling chinook salmon. Comp Biochem Physiol A 129:585–593
Beckman BR, Fairgrieve W, Cooper KA, Mahnken CVW, Beamish RJ (2004a) Evaluation of endocrine indices of growth in individual postsmolt coho salmon. Trans Am Fish Soc 133:1057–1067
Beckman BR, Shimizu M, Gadberry BA, Cooper KA (2004b) Response of the somatotropic axis of juvenile coho salmon to alterations in plane of nutrition with an analysis of the relationships among growth rate and circulating IGF-I and 41 kDa IGFBP. Gen Comp Endocrinol 135:334–344
Beckman BR, Shimizu M, Gadberry BA, Parkins PJ, Cooper KA (2004c) The effect of temperature change on the relations among plasma IGF-I, 41-kDa IGFBP, and growth rate in postsmolt coho salmon. Aquaculture 241:601–619
Buckley LJ (1984) RNA–DNA ratio: an index of larval fish growth in the sea. Mar Biol 80:291–298
Chícharo MA, Chícharo L (2008) RNA:DNA ratio and other nucleic acid derived indices in marine ecology. Int J Mol Sci 9:1453–1471
Daughaday WH, Rotwein P (1989) Insulin-like growth factors I and II. Peptide, messenger ribonucleic acid and gene structures, serum, and tissue concentrations. Endocr Rev 10:68–91
Dyer AR, Barlow CG, Bransden MP, Carter CG, Glencross BD, Richardson N, Thomas PM, Williams KC, Carragher JF (2004) Correlation of plasma IGF-I concentrations and growth rate in aquacultured finfish: a tool for assessing the potential of new diets. Aquaculture 236:583–592
Farley EV, Moss JH, Beamish RJ (2007) A review of the critical size, critical period hypothesis for juvenile Pacific salmon. N Pac Anadromous Fish Comm Bull 4:311–317
Ferriss BE, Trudel M, Beckman BR (2014) Regional and inter-annual trends in marine growth of juvenile salmon in coastal pelagic ecosystems of British Columbia, Canada. Mar Ecol Prog Ser 503:247–261
Fukuwaka M, Suzuki T (2002) Early sea mortality of mark-recaptured juvenile chum salmon in open coastal waters. J Fish Biol 60:3–12
Grémare A, Vétion G (1994) Comparison of several spectrofluorimetric methods for measuring RNA and DNA concentrations in the deposit-feeding bivalve Abra ovate. Comp Biochem Physiol B 107:297–308
Healey MC (1982) Timing and relative intensity of size-selective mortality of juvenile chum salmon (Oncorhynchus keta) during early sea life. Can J Fish Aquat Sci 39:952–957
Hillgruber N, Zimmerman CE (2009) Estuarine ecology of juvenile salmon in western Alaska: a review. Am Fish Soc Symp 70:183–199
Johnson MW, Rooker JR, Gatlin DM III, Holt GJ (2002) Effects of variable ration levels on direct and indirect measures of growth in juvenile red drum (Sciaenops ocellatus). J Exp Mar Biol Ecol 274:141–157
Kawaguchi K, Kaneko N, Fukuda M, Nakano Y, Kimura S, Hara A, Shimizu M (2013) Responses of insulin-like growth factor (IGF)-I and two IGF-binding protein-1 subtypes to fasting and re-feeding, and their relationships with individual growth rates in yearling masu salmon (Oncorhynchus masou). Comp Biochem Physiol A 165:191–198
Kocik JF, Hawkes JP, Sheehan TF, Music PA, Beland KF (2009) Assessing estuarine and coastal migration and survival of wild Atlantic salmon smolts from the Narraguagus River, marine using ultrasonic telemetry. Am Fish Soc Symp 69:293–310
Larsen DA, Beckman BR, Dickhoff WW (2001) The effect of low temperature and fasting during the winter on growth and smoltification of coho salmon. N Am J Aquac 63:1–10
Le Roith D, Bondy C, Yakar S, Liu J, Butler A (2001) The somatomedin hypothesis: 2001. Endocr Rev 22:53–74
Maclean SA, Caldarone EM, St Onge-burns JM (2008) Estimating recent growth rates of Atlantic salmon smolts using RNA–DNA ratios from nonlethally sampled tissues. Trans Am Fish Soc 137:1279–1284
Magnusson A, Hilborn R (2003) Estuarine influence on survival rates of coho (Oncorhynchus kisutch) and chinook salmon (Oncorhynchus tshawytscha) released from hatcheries on the U.S. Pacific coast. Estuaries 26:1094–1103
McCormick SD, Sheehan TF, Th BjörnssonB, Lipsky C, Kocik JF, Regish AM, O’Dea MF (2013) Physiological and endocrine changes in Atlantic salmon smolts during hatchery rearing, downstream migration, and ocean entry. Can J Fish Aquat Sci 70:105–118
Miyakoshi Y, Nagata M, Kitada S, Kaeriyama M (2013) Historical and current hatchery programs and management of chum salmon in Hokkaido, northern Japan. Rev Fish Sci 21:469–479
Nagata M, Miyakoshi Y, Ando D, Fujiwara M, Sawada M, Shimada H, Asami H (2007) Influence of coastal seawater temperature on the distribution and growth of juvenile chum salmon, with recommendations for altered release strategies. N Pac Anadromous Fish Comm Bull 4:223–235
Ohlsson C, Mohan S, Sjögren K, Tivesten Ä, Isgaard J, Isaksson O, Jansson J, Svensson J (2009) The role of liver-derived insulin-like growth factor-I. Endocr Rev 30:494–535
Picha ME, Turano MJ, Beckman BR, Borski RJ (2008) Endocrine biomarkers of growth and applications to aquaculture: a minireview of growth hormone, insulin-like growth factor (IGF)-I, and IGF-binding proteins as potential growth indicators in fish. N Am J Aquac 70:196–211
Pierce AL, Shimizu M, Beckman BR, Baker DM, Dickhoff WW (2005) Time course of the GH/IGF axis response to fasting and increased ration in chinook salmon (Oncorhynchus tshawytscha). Gen Comp Endocrinol 140:192–202
Salo EO (1991) Life history of chum salmon (Oncorhynchus keta). In: Croot C, Margolis L (eds) Pacific salmon life history. UBC Press, Vancouver, pp 233–309
Shimizu M, Swanson P, Fukada H, Hara A, Dickhoff WW (2000) Comparison of extraction methods and assay validation for salmon insulin-like growth factor-I using commercially available components. Gen Comp Endocrinol 119:26–36
Small BC, Peterson BC (2005) Establishment of a time-resolved fluoroimmunoassay for measuring plasma insulin-like growth factor I (IGF-I) in fish: effect of fasting on plasma concentrations and tissue mRNA expression of IGF-I and growth hormone (GH) in channel catfish (Ictalurus punctatus). Domest Anim Endocrinol 28:202–215
Stefansson SO, Haugland M, Björnsson BTh, McCormick SD, Holm M, Ebbesson LOE, Holst JC, Nilsen TO (2012) Growth, osmoregulation and endocrine changes in wild Atlantic salmon smolts and post-smolts during marine migration. Aquaculture 362–363:127–136
Thorstad EB, Whoriskey F, Uglem I, Moore A, Rikardsen AH, Finstad B (2012) A critical life stage of the Atlantic salmon Salmo salar: behavior and survival during the smolt and initial post-smolt migration. J Fish Biol 81:500–542
Wedemeyer GA, Saunders RL, Clarke WC (1980) Environmental factors affecting smoltification and early marine survival of anadromous salmonids. Mar Fish Rev 42:1–14
Wertheimer AC, Thrower FP (2007) Mortality rates of chum salmon during their early marine residency. Am Fish Soc Symp 57:1–15
Acknowledgments
We thank Yuji Yoshida and staffs of the Abashiri Fisheries Cooperative Association, the Abashiri City Science Center and the East Branch of Fisheries Technical Guidance Office for supporting field surveys. We also thank staffs of the Kitami Salmon Enhancement Programs Association for providing juvenile chum salmon for tank experiment. This work was supported by Grant-in-Aids for Scientific Research from Japan Society for the Promotion of Science (#25450289) and Hokusui Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kaneko, N., Taniyama, N., Inatani, Y. et al. Circulating insulin-like growth factor I in juvenile chum salmon: relationship with growth rate and changes during downstream and coastal migration in northeastern Hokkaido, Japan. Fish Physiol Biochem 41, 991–1003 (2015). https://doi.org/10.1007/s10695-015-0064-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-015-0064-7