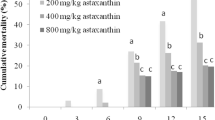
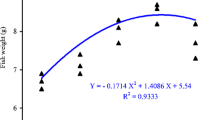
Abstract
Healthy crucian carp (Carassius auratus) were treated by intraperitoneal (i.p.) injection of crude cyanobacterial extracts at two doses, 50 and 200 μg MC-LR equiv kg−1 BW. High mortality (100%) was observed within 60 h post injection in the high-dose group. In the treated fish, activities of four plasma enzymes, alanine aminotransferase (ALT), alkaline phosphatase (ALP), aspartate aminotransferase (AST), and lactate dehydrogenase (LDH), all showed substantial increases, with both dose and time-dependent effects. These increases of enzyme activity indicate severe impairment occurred in the liver of crucian carp over time. Plasma concentrations of energy-related biomolecules including glucose (GLU), cholesterol (CHO), triglyceride (TG), and total protein (TP) showed marked changes in the high-dose group, possibly a nutritional imbalance correlated with the liver injury caused by intraperitoneal exposure to crude cyanobacterial extracts.


Similar content being viewed by others
References
Adhikari S, Sarkar B, Chatterjee A, Mahapatra CT, Ayyappan S (2004) Effects of cypermethrin and carbofuran on certain haematological parameters and prediction of their recovery in a freshwater teleost., Labeo rohita (Hamilton). Ecotoxicol Environ Saf 58:220–226
Andenen DE, Reid SD, Moon TW, Perry SF (1992) Metabolic effects associated with chronically elevated cortisol in rainbow trout (Oncorhynchus mykiss). Can J Fish Aquat Sci 48:1811–1817
Beasley VR, Lovell RA, Holmes KR, Walcott HE, Schaeffer DJ, Walcott HE, Schaeffer DJ (2000) Microcystin-LR decreases hepatic and renal perfusion, and causes circulatory shock, severe hypoglycemia, and terminal hyperkalemia in intravascularly dosed swine. J Toxicol Env Health Part A 61:281–303
Bernet D, Schmidt H, Wahli T, Burkhardt HP (2001) Effluent from a sewage treatment works causes changes in serum chemistry of brown trout (Salmo trutta L.). Ecotox Environ Safe 48:140–147
Bergmeyer HU, Gawehn K, Grassl M (1974) Enzymes as biochemical reagents. In: Bergmeyer HU (ed) Methods of enzymatic analysis, vol 1. Academic Press, New York, pp 481–482
Bergmeyer HU, Bowers GN, Horder M, Moss DW (1976) Provisional recommendations on IFCC methods for the measurement of catalytic concentrations of enzymes. Clin Chim Acta 70:F12, FM
Best JH, Pflugmacher S, Wiegand C, Eddy FB, Metcalf JS, Codd GA (2002) Effects of enteric bacterial and cyanobacterial lipopolysaccharides, and of microcystin-LR, on glutathione-S-transferase activities in zebra fish (Danio rerio). Aquat Toxicol 60:223–231
Burýšková B, Hilscherová K, Babica P, Vršková D, Maršálek B, Bláha L (2006) Toxicity of complex cyanobacterial samples and their fractions in Xenopus laevis embryos and the role of microcystins. Aquat Toxicol 80:346–354
Ding XS, Li YY, Duan HY, Chung IK, Lee JA (2006) Toxic effects of Microcystis cell extracts on the reproductive system of male mice. Toxicon 48:973–979
Falconer IR (2007) Cyanobacterial toxins present in Microcystis aeruginosa extracts—more than microcystins. Toxicon 50:585–588
Fischer WJ, Dietrich DR (2000) Pathological and biochemical characterization of microcystin-induced hepatopancreas and kidney damage in carp (Cyprinus carpio). Toxicol Appl Pharmacol 164:73–81
Gupta N, Pant SC, Vijayaraghavan R, Lakshmana Rao PV (2003) Comparative toxicity evaluation of cyanobacterial cyclic peptide toxin microcystin variants (LR, RR, YR) in mice. Toxicology 188:285–296
Hawk PB, Oser BL, Summersion WH (1954) Practical physiological chemistry. McGraw–Hill, New York
Kotak BG, Semalulu S, Fritz DL, Prepas EE, Hrudey SE, Coppock RW (1996) Hepatic and renal pathology of intraperitoneally administered microcystin-LR in rainbow trout (Oncorhynchus mykiss). Toxicon 34:517–525
Li L, Xie P, Chen J (2005) In vivo studies on toxin accumulation in liver and ultrastructural changes of hepatocytes of the phytoplanktivorous bighead carp i.p.-injected with extracted microcystins. Toxicon 46:533–545
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:269–275
Malbrouck C, Trausch G, Devos P, Kestemont P (2003) Hepatic accumulation and effects of microcystin-LR on juvenile goldfish Carassius auratus L. Comp Biochem Physiol Part C 135:39–48
Malbrouck C, Kestemont P (2006) Effects of microcystins on fish. Environ Toxicol Chem 25:72–86
McComb RR, Bowers GN Jr (1972) Study of optimal buffer conditions for measuring alkaline phosphatase activity in human serum. Clin Chem 18:97–104
Molina R, Moreno I, Pichardo S, Jos A, Moyano R, Monterde JG (2005) Acid and alkaline phosphatase activities and pathological changes induced in Tilapia fish (Oreochromis sp.) exposed subchronically to microcystins from toxic cyanobacterial blooms under laboratory conditions. Toxicon 46:725–735
Oberemm A, Fastner J, Steinberg CEW (1997) Effects of microcystins-LR and cyanobacterial crude extracts on embryo-larval development of zebrafish (Danio rerio). Water Res 31:2918–2921
Oberemm A, Becker J, Codd GA, Steinberg C (1999) Effects of cyanobacterial toxins and aqueous crude extracts of cyanobacteria on the development of fish and amphibians. Environ Toxicol 14:77–88
Palíková M, Krejčí R, Hilscherová K, Babica P, Navrátil S, Kopp R, Bláha L (2007) Effect of different cyanobacterial biomasses and their fractions with variable microcystin content on embryonal development of carp (Cyprinus carpio L.) Aquat Toxicol 81:312–318
Rěhulka J (2000) Influence of astaxanthin on growth rate, condition, and some blood indices of rainbow trout, Oncorhynchus mykiss. Aquaculture 190:27–47
Råbergh CMI, Bylund G, Eriksson JE (1991) Histopathological effects of microcystin-LR, a cyclic peptide toxin from the cyanobacterium (blue-green alga) microcystis aeruginosa, on common carp (Cyprinus carpio L.). Aquat Toxicol 20:131–146
Vajcová V, Navrati S, Palíková M (1998) The effect of intraperitoneally applied pure microcystin-LR on haematological, biochemical and morphological indices of silver carp (Hypophthalmichthys molitrix Val.). Acta Vet Brno 67:281–287
Xie L, Xie P, Ozawa K (2004) Dynamics of microcystins-LR and -RR in the phytoplanktivorous silver carp in a sub-chronic toxicity experiment. Environ Pollut 127:431–439
Xie P (2006) Microcystins in aquatic animals with potential risk to human health. Science Press, Beijing
Zhang X, Xie P, Li D, Shi Z (2007) Haematological and plasma biochemical responses of crucian carp (Carassius auratus) to intraperitoneal injection of extracted microcystins with the possible mechanisms of anemia. Toxicon 49:1150–1157
Acknowledgements
We would like to express our sincere thanks to Dr H.J.T. Goos and the anonymous reviewer for their useful comments and suggestions on the manuscript. This study is supported by a fund for lecture professor from Huazhong Agricultural University and the National Natural Science Foundation of China (30530170).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, X., Xie, P., Wang, W. et al. Plasma biochemical responses of the omnivorous crucian carp (Carassius auratus) to crude cyanobacterial extracts. Fish Physiol Biochem 34, 323–329 (2008). https://doi.org/10.1007/s10695-007-9191-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-007-9191-0