Abstract
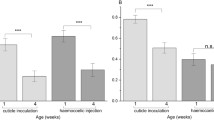
Arguments from life-history theory predict that other things being equal females are likely to invest more in defence against parasites and pathogens than males. This is either because males and females differ in behaviour or, more importantly, because the variance in mating success is typically higher in males than in females. Such effects are likely to be most pronounced in those developmental stages where sex differences are greatest. In most organisms, but especially in holometabolous insects, this will be the adult stage. We explored sex-specific resistance to four natural enemies of Drosophila melanogaster that attack the insect at different developmental stages: the larval parasitoid Asobara tabida, the pupal parasitoid Pachycrepoideus vindemiae; and the adult pathogens Beauvaria bassiana (a fungus) and Tubulinosema kingi (a microsporidian). Measures of resistance were designed to reflect the four species’ natural history. Female larvae were able to defend themselves more strongly against A. tabida than males and there was weak evidence that adult females suffered less from microsporidian attack than males. No differences were found for the other two species. Our results provide some support for lower investment in defences in males, and we discuss why the strongest effect was found at the larval rather than the adult stage contrary to our prediction.

Similar content being viewed by others
References
van Alphen JJM, Drijver RAB (1982) Host selection by Asobara tabida Nees (Braconidae; Alysiinae) a larval parasitoid of fruit inhabiting Drosophila species. I. Host stage selection with Drosophila melanogaster as host species. Neth J Zool 32:215–231
van Alphen JJM, Thunnissen I (1983) Host selection and sex allocation by Pachycrepoideus vindemiae Rondani (Pteromalidae) as a facultative hyperparasitoid of Asobara tabida Nees (Braconidae; Alysiinae) and Leptopilina heterotoma (Thomson)(Cynipoidea; Eucoilidae). Neth J Zool 33:497–514
Armstrong E, Bass L (1989a) Effects of Nosema kingi on the development and weight of adult Drosophila melanogaster (OR-R-Strain). J Invertebr Pathol 53:102–106
Armstrong E, Bass L (1989b) Nosema kingi: effects on fecundity, fertility and longevity of Drosophila melanogaster. J Exp Zool 250:82–86
Bateman AJ (1948) Intra-sexual selection in Drosophila. Heredity 2:349–368
Becnel JJ, Andreadis TG (1999) Microsporidia in insects. In: Wittner M, Weiss LM (eds) The microsporidia and microsporidiosis. ASM Press, Washington, DC
Braune P, Rolff J (2001) Parasitism and survival in a damselfly: does host sex matter? Proc R Soc Lond B 268:1133–1137
David L, Weiser J (1994) Role of hemocytes in the propagation of a microsporidian infection in larvae of Galleria mellonella. J Invertebr Pathol 63:212–213
Eslin P, Prévost G (2000) Racing against host’s immunity defenses: a likely strategy for passive evasion of encapsulation in Asobara tabida parasitoids. J Ins Phys 46:1161–1167
Fellowes MDE, Masnatta P, Kraaijeveld AR, Godfray HCJ (1998) Pupal parasitoid attack influences the relative fitness of Drosophila that have encapsulated larval parasitoids. Ecol Ent 23:281–284
Feng MG, Poprawski TJ, Khachatourians GG (1994) Production, formulation and application of the entomopathogenic fungus Beauveria bassiana for insect control: current status. Biocontrol Sci Techn 4:3–34
Folstad I, Karter A (1992) Parasites, bright males, and the immunocompetence handicap. Am Nat 139:603–622
Franzen C, Futerman PH, Schroeder J, Salzberger B, Kraaijeveld AR (2006) An ultrastructural and molecular study of Tubulinosema kingi Kramer (Microsporidia: Tubulinosematidae) from Drosophila melanogaster (Diptera: Drosophilidae) and its parasitoid Asobara tabida (Hymenoptera: Braconidae). J Invertebr Pathol 91:158–167
Futerman PH, Layen SJ, Franzen C, Kraaijeveld AR, Godfray HCJ (2006) Fitness effects and transmission routes of a microsporidian parasite infecting Drosophila and its parasitoids. Parasitology 132:479–492
Fytrou A, Schofield PG, Kraaijeveld AR, Hubbard SF (2006) Wolbachia infection suppresses both host defence and parasitoid counter-defence. Proc R Soc Lond B 273:791–796
Gray DA (1998) Sex differences in susceptibility of house crickets, Acheta domesticus, to experimental infection with Serratia liquefaciens. J Invertebr Pathol 71:288–289
Hazarika LK, Gupta AP (1987) Variations in hemocyte populations during various developmental stages of Blatella germanica (L.) (Dictyoptera, Blatellidae). Zool Sci 4:307–313
Hoch G, Solter LF, Schopf A (2004) Hemolymph melanization and alterations in hemocyte numbers in Lymantria dispar larvae following infections with different entomopathogenic microsporidia. Entomol Exp Appl 113:77–86
Hosken DJ (2001) Sex and death: microevolutionary trade-offs between reproductive and immune investment in dung flies. Curr Biol 11:R379–R380
Iwasa Y, Haccou P (1994) ESS emergence pattern of male butterflies in stochastic environment. Evol Ecol 8:503–523
Joop G, Mitschke A, Rolff J, Siva-Jothy M (2006) Immune function and parasite resistance in male and polymorphic female Coenagrion puella. BMC Evol Biol 6:19
Joop G, Rolff J (2004) Plasticity of immune function and condition under the risk of predation and parasitism. Evol Ecol Res 6:1051–1062
Kraaijeveld AR, van Alphen JJM (1995) Geographical variation in encapsulation ability of Drosophila melanogaster larvae and evidence for parasitoid-specific components. Evol Ecol 9:10–17
Kraaijeveld AR, Ferrari J, Godfray HCJ (2002) Costs of resistance in insect-parasite and insect-parasitoid interactions. Parasitology 125:S71–S82
Kraaijeveld AR, Godfray HCJ (1997) Trade-off between parasitoid resistance and larval competitive ability in Drosophila melanogaster. Nature 389:278–280
Kraaijeveld AR, Godfray HCJ (2003) Potential life-history costs of parasitoid avoidance in Drosophila melanogaster. Evol Ecol Res 5:1251–1261
Kraaijeveld AR, van der Wel NN (1994) Geographic variation in reproductive success of the parasitoid Asobara tabida in larvae of several Drosophila species. Ecol Entomol 19:221–229
Kurtz J, Sauer K (2001) Gender differences in phenoloxidase activity of Panorpa vulgaris hemocytes. J Invertebr Pathol 78:53–55
Kurtz J, Wiesner A, Götz P, Sauer K (2000) Gender differences and individual variation in the immune system of the scorpionfly Panorpa vulgaris (Insecta: Mecoptera). Dev Comp Immunol 24:1–12
Lanot R, Zachary D, Holder F, Meister M (2001) Postembryonic hematopoiesis in Drosophila. Dev Biol 230:243–257
Lavine MD, Strand MR (2002) Insect hemocytes and their role in immunity. Ins Biochem Mol Biol 32:1295–1309
Lemaitre B, Reichhart J-M, Hoffmann JA (1997) Drosophila host defense: differential induction of antimicrobial peptide genes after infection by various classes of microorganisms. Proc Natl Acad Sci USA 94:14614–14619
Lenski RE (1988) Experimental studies of pleiotropy and epistasis in Escherichia coli. I. Variation in competitive fitness among mutants resistant to virus T4. Evolution 42:425–432
Meister M, Lagueux M (2003) Drosophila blood cells. Cell Microbiol 5:573–580
Mohammed-Ali Q, Elwali NE, Abdelhameed AA, Mergani A, Rahoud S, Elagib KE, Saeed OK, Abel L, Magzoub MM, Dessein AJ (1999) Susceptibility to periportal (Symmers) fibrosis in human Schistosoma mansoni infections: evidence that intensity and duration of infection, gender, and inherited factors are critical in disease progression. J Infect Dis 180:1298–1306
Moore SL, Wilson K (2002) Parasites as a viability cost of sexual selection in natural populations of mammals. Science 297:2015–2018
Nappi AJ (1981) Cellular immune response of Drosophila melanogaster against Asobara tabida. Parasitology 83:319–324
Nigam Y, Maudlin I, Welburn NA (1997) Detection of phenoloxidase activity in the hemolymph of tsetse flies, refractory and susceptible to infection with Trypanosoma brucei rhodesiense. J Invertebr Pathol 69:279–281
Nunney L (1983) Sex differences in larval competition in Drosophila melanogaster: The testing of a competition model and its relevance to frequency-dependent selection. Am Nat 121:67–93
Nunney L (1996) The response to selection for fast development in Drosophila melanogaster and its effect on adult weight. Evolution 50:1193–1204
Offner PJ, Moore EE, Biffl WL (1999) Male gender is a risk factor for major infections after surgery. Arch Surg 134:935–938
Poulin R (1996) Sexual inequalities in helminth infections: a cost of being a male? Am Nat 147:287–295
Radhika M, Abdul Nazar AK, Munuswamy N, Nellaiappan K (1998) Sex-linked differences in phenol oxidase in the fairy shrimp Streptocephalus dichotomus Baird and their possible role (Crustacea: Anostraca). Hydrobiologia 377:161–164
Rheins LA, Karp RD (1985) Effect of gender on the inducible humoral immune response to honeybee venom in the American cockroach (Periplaneta americana). Dev Comp Immunol 9:41–49
Rigby MC, Jokela J (2000) Predator avoidance and immune defence: costs and trade-offs in snails. Proc R Soc Lond B 267:171–176
Rolff J (2002) Bateman’s principle and immunity. Proc R Soc Lond B 269:867–872
Roxström-Lindquist K, Terenius O, Faye I (2004) Parasite-specific immune response in adult Drosophila melanogaster: a genomic study. EMBO Rep 5:207–212
Schmid-Hempel P, Müller C, Schmid-Hempel R, Shykoff JA (1990) Frequency and ecological correlates of parasitism by conopid flies (Conopidae, Diptera) in populations of bumblebees. Insectes Sociaux 37:14–30
Sheldon B, Verhulst S (1996) Ecological immunity: costly parasite defences and trade-offs in evolutionary ecology. Trends Ecol Evol 11:317–321
Sheridan LAD, Poulin R, Ward DF, Zuk M (2000) Sex differences in parasitic infections among arthropod hosts: is there a male bias? Oikos 88:327–334
Stearns SC (1992) The evolution of life histories. Oxford University Press, Oxford
van der Veen IT, Kurtz J (2002) To avoid or eliminate: cestode infections in copepods. Parasitology 124:465–474
Verhulst S, Dieleman SJ, Parmentier HK (1999) A trade-off between immunocompetence and sexual ornamentation in domestic fowl. Proc Natl Acad Sci USA 96:4478–4481
Webster JP, Woolhouse MEJ (1999) Cost of resistance: relationship between reduced fertility and increased resistance in a snail-schistosome host-parasite system. Proc R Soc Lond B 266:391–396
Wedekind C, Jacobsen PJ (1998) Male-biased susceptibility to helminth infection: an experimental test with a copepod. Oikos 81:458–462
Wertheim B, Kraaijeveld AR, Schuster E, Blanc E, Hopkins M, Pletcher SD, Strand MR, Partridge L, Godfray HCJ (2005) Genome-wide gene expression in response to parasitoid attack in Drosophila. Genome Biol 6:R94
Zuk M, McKean KA (1996) Sex differences in parasite infections: patterns and processes. Int J Parasitol 26:1009–1023
Acknowledgements
We are grateful to Meirion Hopkins and Martha Kotzen for maintaining all the cultures and to Kim Dobson and Meirion Hopkins for help in constructing the cages for the fungal experiment. Franco Pennacchio (Università Basilicata) collected the AV fly strain. Gé Boskamp (Universiteit Leiden) supplied the pupal parasitoids and Marie Meister (Institut de Biologie Moleculaire et Cellulaire, Strasbourg) the fungus. Carly Whittaker and Meirion Hopkins assisted with the fungus experiment, Sophie Layen with the microsporidian experiment. Comments by Jens Rolff improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kraaijeveld, A.R., Barker, C.L. & Godfray, H.C.J. Stage-specific sex differences in Drosophila immunity to parasites and pathogens. Evol Ecol 22, 217–228 (2008). https://doi.org/10.1007/s10682-007-9171-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-007-9171-y