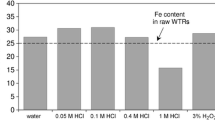
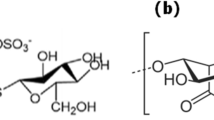
Abstract
The present work analyses the adsorption capacity of lead ions onto two bioadsorbents consisting of biodegraded grape marc (BGM) and free or encapsulated in calcium alginate, forming hybrid beads (alginate–grape marc hybrid beads (AGMHB)). It has been verified that the removal effectiveness of BGM was always lower than that of AGMHB. Moreover, the pH of lead ion solution had a significant influence on the removal efficacy of both bioadsorbents tested. A Box-Behnken factorial design optimized the most influential variables (amount of bioadsorbent, concentration of lead ions and pH solution) involved in the adsorption of lead ions onto AGMHB. Theoretical equations were validated by means of additional experiments, resulting in a good agreement between both experimental results and estimated values. In addition, different kinetic models were evaluated in order to obtain kinetic parameters associated to the bioadsorption process. The intraparticle diffusion model was also employed to interpret the diffusion of lead ions among the particles of the bioadsorbent.





Similar content being viewed by others
References
WHO (World Health Organization), Exposure to lead: a major public health concern, 2010.
Arbabi, M., Hemati, S., & Amiri, M. (2015). Removal of lead ions from industrial wastewater: a review of removal methods. International Journal of Epidemiologic Research, 2(2), 105–109.
FAO (Food and Agriculture Organization), The Environment Protection. Standards for effluent discharge regulations. General Notice No 44, 2003.
WHO (World Health Organization), Lead in drinking water. Background document for development of WHO Guidelines for Drinking-water Quality, 2011.
Barakat, M. (2011). New trends in removing heavy metals from industrial wastewater. Arabian Journal of Chemistry, 4(4), 361–377. https://doi.org/10.1016/j.arabjc.2010.07.019.
Agarwal, R. M., & Singh, K. (2017). Heavy metal removal from wastewater using various adsorbents: a review. Journal of Water Reuse and Desalination, 7(4), 387–410. https://doi.org/10.2166/wrd.2016.104.
Anwar, J., Shafique, U., Zaman, W., Saiman, M., Dar, A., & Anwar, S. (2010). Removal of Pb(II) and Cd(II) from water by adsorption on peels of banana. Bioresource Technology, 101, 1752–1755.
Vilardi, G., Di Palma, L., & Verdone, N. (2018). Heavy metals adsorption by banana peels micro-powder: equilibrium modeling by non-linear models. Chinese Journal of Chemical Engineering, 26, 455–464. https://doi.org/10.1016/j.cjche.2017.06.026.
Goyal, K., & Arora, S. (2016). Equilibrium and kinetic studies of adsorption of lead using low cost adsorbents. Indian Journal of Science and Technology, 9(44), 105258. https://doi.org/10.17485/ijst/2016/v9i44/105258.
Nastaj, J., Przewłocka, A., & Rajkowska-Myśliwiec, M. (2016). Biosorption of Ni(II), Pb(II) and Zn(II) on calcium alginate beads: equilibrium, kinetic and mechanism studies. Polish Journal of Chemical Technology, 18(3), 81–87. https://doi.org/10.1515/pjct-2016-0052.
Pal, P., Edathil, A. A., & Banat, F. (2019). Calcium alginate gel and hard beads for the removal of total organic acid anions and heavy metal ions from industrial lean methyldiethanolamine solvent. Polymer Bulletin, 76, 103–118. https://doi.org/10.1007/s00289-018-2376-0.
Flórez-Fernández, N., Domínguez, H., & Torres, M. D. (2019). A green approach for alginate extraction from Sargassum muticum brown seaweed using ultrasound-assisted technique. International Journal of Biological Macromolecules, 124, 451–459.
Ablouh, E., Essaghraoui, A., Eladlani, N., Rhazi, M., & Taourirte, M. (2019). Uptake of Pb(II) onto nanochitosan/sodium alginate hybrid beads: mechanism and kinetics study. Water Environment Research, 91, 239–249. https://doi.org/10.1002/wer.1050.
Arshada, F., Selvaraja, M., Zaina, J., Banata, F., & Haijab, M. A. (2019). Polyethylenimine modified graphene oxide hydrogel composite as an efficient adsorbent for heavy metal ions. Separation and Purification Technology, 209, 870–880. https://doi.org/10.1016/j.seppur.2018.06.035.
Lu, M., Liu, Y., Hu, X., Ben, Y., Zeng, X., Li, T., & Wang, H. (2013). Competitive adsorption of Cu(II) and Pb(II) ions from aqueous solutions by Ca-alginate immobilized activated carbon and Saccharomyces cerevisiae. Journal of Central South University, 20, 2478–2488. https://doi.org/10.1007/s11771-013-1760-z.
Bustos, G., Calvar, S., Vecino, X., Cruz, J. M., & Moldel, A. B. (2018). Industrial symbiosis between the winery and environmental industry through the utilization of grape marc for water desalinitation containing copper (II). Water, Air, and Soil Pollution, 229, 36. https://doi.org/10.1007/s11270-018-3697-1.
Vecino, X., Devesa-Rey, R., Moldes, A. B., & Cruz, J. M. (2014). Formulation of an alginate-vineyard pruning waste composite as a new eco-friendly adsorbent to remove micronutrients from agroindustrial effluents. Chemosphere, 111, 24–31. https://doi.org/10.1016/j.chemosphere.2014.03.004.
Moldes, A. B., Vázquez, M., Domínguez, J. M., Díaz-Fierros, F., & Barral, M. T. (2007). Evaluation of mesophilic biodegraded grape marc as soil fertilizer. Applied Biochemistry and Biotechnology, 141, 27–36. https://doi.org/10.1007/s12010-007-9208-2.
Vecino, X., Devesa-Rey, R., Cruz, J. M., & Moldes, A. B. (2013). Entrapped peat in alginate beads as green adsorbent for the elimination of dye compounds from vinasses. Water, Air, and Soil Pollution, 224(3), 1448.
Box, G. E., Hunter, J. S., & Hunter, W. G. (2005). Statistics for experimenters: design, innovation and discovery (2nd ed.). New Yersey: Wiley and Sons.
Lagergren, S. (1898). About the theory of so-called adsorption of soluble substances. Kungliga Svenska Vetenskapsakademiens. Handlingar, 24(4), 1–39.
Ho, Y. S., & McKay, G. (1999). Pseudo-second order model for sorption processes. Process Biochemistry, 34(5), 451–465.
Chien, S. H., & Clayton, W. R. (1980). Application of Elovich equation to the kinetics of phosphate release and sorption in soils. Soil Science Society of America Journal, 44(2), 265–268.
Weber, W. J., & Morris, J. C. (1962). Advances in water pollution research: removal of biologically resistant pollutants from waste waters by adsorption. Proceedings of International Conference on Water Pollution Symposium, 2, 231–266.
Ucun, H., Bayhan, Y. K., Kaya, Y., Cakici, A., & Algur, O. F. (2003). Biosorption of lead (II) from aqueous solution by cone biomass of Pinus sylvestris. Desalination, 154, 233–238. https://doi.org/10.1016/S0011-9164(03)80038-3.
Vecino, X., Devesa-Rey, R., Villagrasa, S., Cruz, J. M., & Moldes, A. B. (2015). Kinetic and morphology study of alginate-vineyard pruning waste biocomposite vs. non modified vineyard pruning waste for dye removal. Journal of Environmental Sciences (China), 38, 158–167. https://doi.org/10.1016/j.jes.2015.05.032.
Funding
This study was supported by the Xunta de Galicia under the project ED431B 2017/77.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iglesias, A.M., Cruz, J.M., Moldes, A. et al. Efficient Adsorption of Lead Ions onto Alginate–Grape Marc Hybrid Beads: Optimization and Bioadsorption Kinetics. Environ Model Assess 25, 677–687 (2020). https://doi.org/10.1007/s10666-020-09692-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10666-020-09692-z