Abstract
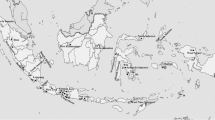
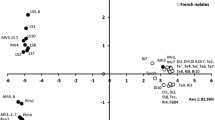
The population structure of the rice blast fungus Pyricularia grisea was analyzed in two major rice-growing provinces of Iran using rep-PCR DNA fingerprinting. A total of 221 monoconidial isolates of the fungus was collected from 12 cultivars at ten regions during 1997–2000. Long-PCR conditions were used to amplify sequences lying between adjacent Pot2 elements. The frequencies of Pot2 lineages (isolates with ≥70% amplicon similarity) and haplotypes within lineages were determined. Phenetic analysis differentiated five Pot2 fingerprint lineages, designated A, B, C, D and E. The most common fingerprint group, Lineage E, was recovered from all rice cultivars sampled and was distributed throughout the region. Haplotype E6, the most common haplotype within lineage E, was recovered from almost all regions. Lineage A, the second most common lineage, was found mainly in the western part of the sampled region. Haplotype A1 was found in most sites in the western province. Lineage A occurred at relatively high frequency on the susceptible local cultivar Binam, suggesting that lineage A is specifically adapted to Binam. To test this hypothesis, 193 additional isolates were recovered from four fields at two sites separated by approximately 100 km. This second, field-specific collection of isolates contained lineages A, C, D, and E. Approximately 64% and 29% of the isolates recovered from Binam (the shared cv. at two sites) grouped into lineages A and E, respectively. The other two susceptible cultivars at these sites were infected by lineage E at frequencies of 100% and 71%. Overall, these data indicated a low level of genetic diversity in the Iranian P. grisea population similar to that reported in other countries.
Similar content being viewed by others
References
Bahrami M and Izadyar M (1998) Introduction of new races of rice blast disease (Pyricularia grisea ). In: Proceeding of the 13th Iranian Plant Protection Congress (p 73) Karaj, Iran
Chen D, Zeigler RS, Leung H and Nelson RJ (1995) Population structure of Pyricularia grisea at two screening sites in the Philippines. Phytopathology 85: 1011-1020
Correa-Victoria FJ, Zeigler RS and Levy M (1994) Virulence characteristics of genetic families of Pyricularia grisea in Colombia. In: Zeigler RS, Leong SA and Teng PS (eds) Rice Blast Disease (pp 211-229) CABI/IRRI, Wallingford, Oxon, UK
Couch BC and Kohn LM (2002) A multilocus gene genealogy concordant with host preference indicates segregation of a new species, Magnaporthe oryzae from M. grisea. Mycologia 94: 683-693
Don LD, Tosa Y, Nakayashiki H and Mayama S (1999) Population structure of the rice blast pathogen in Vietnam. Annals of the Phytopathological Society of Japan 65: 475-479
Fatemi J and Rahimian H (1975) Prevalence and distribution of the rice blast disease in Iran. Plant Disease Reporter 59: 991-994
George MLC, Nelson RJ, Zeigler RS and Leung H (1998) Rapid population analysis of Magnaporthe grisea by using rep-PCR and endogenous repetitive DNA sequences. Phytopathology 88: 223-229
Hamer JE, Farral L, Orbach MJ, Valent B and Chumley F (1989) Host species-specific conservation of a family of repeated DNA sequences in the genome of a fungal plant pathogen. Proceedings of the National Academy of Sciences USA 86: 9981-9985
Izadyar M (1982) Pathogenic races of Pyricularia oryzae in Guilan province of Iran. Iranian Journal of Plant Pathology 18: 52-57
Javan-Nikkhah M and Hedjaroude GA (2000) Study on physiological races of rice blast disease in Guilan, Iran. In: Proceedings of the 14th Iranian Plant Protection Congress (p 245) Isfahan, Iran
Javan-Nikkhah M, Hedjaroude GA, Shari-Tehrani A and Okhovvat SM (2003) Study on pathogenic diversity in population of Magnaporthe grisea, the rice blast fungus in Guilan province, Iran. Iranian Journal of Agricultural Science 34: 647-658
Kachroo P, Leong SA and Chattoo BB (1994) Pot2, an inverted repeat transposon from the rice blast fungus Magnapor-the grisea. Molecular and General Genetics 245: 339-348
Kiyosawa S (1972) Genetics of blast resistance. In: Rice Breeding (pp 203-225) International Rice Research Institute, PO Box 933, Manila, The Philippines
Kumar J, Nelson RJ and Zeigler RS (1999) Population structure and dynamics of Magnaporthe grisea in the Indian Himalayas. Genetics 152: 971-984
Lee SB, Milgroom MG and Taylor JW (1988) A rapid, high yield mini-prep method for isolation of total genomic DNA from fungi. Fungal Genetics Newsletter 35: 23-34
Leung H and Taga M (1988) Magnaporthe grisea (Pyricularia grisea ), the blast fungus. Advances in Plant Pathology 6: 175-188
Levy M, Romao J, Marchetti MA and Hamer JE (1991) DNA ngerprinting with a dispersed repeated sequence resolves pathotypic diversity in the rice blast fungus. The Plant Cell 3: 95-102
Levy M, Correa-Victoria FJ, Zeigler RS, Xu S and Hamer JE (1993) Genetic diversity of the rice blast fungus in a blast disease nursery in Colombia. Phytopathology 83: 1427-1433
Ling KC and Ou SH (1969) Standardization of the international race numbers of Pyricularia oryzae. Phytopathology 59: 339-342
Louws FJ, Rademaker JLW and de Bruijn (1999) The three Ds of PCR-based genomic analysis of phytobacteria: Diversity, detection, and disease diagnosis. Annual Review of Phytopathology 37: 81-125
Nei M (1975) Molecular Population Genetics and Evolution. American Elsevier, New York, USA
Nei M and Li WH (1979) Mathematical model for studying genetic variation in terms of restriction endonucleases. Proceedings of the National Academy of Sciences of the USA 76: 5269-5273
Ou SH (1979) Breeding of rice for resistance to blast, a critical review. In: Proceeding of the rice blast workshop (pp 81-137) International Rice Research Institute, PO Box 933, Manila, The Philippines
Ou SH (1985) Rice Diseases, 2nd ed. Commonwealth Mycological Institute/Association of Applied Biology, Kew, Surrey, England
Rademaker JLW and de Bruijn FJ (1997) Characterization and classi cation of microbes by rep-PCR genomic ngerprinting and computer assisted pattern analysis. In: Caetano-Anolle _ G and Gressho. PM (eds) DNA Markers: Protocols, Applications, and Overviews (pp 151-171) Wiley and Sons, Inc, New York, NY, USA
Rossman AY, Howard RJ and Valent B (1990) Pyricularia grisea, the correct name for the rice blast disease fungus. Mycologia 82: 509-512
Roumen E, Levy M and Notteghem JL (1997) Characterisation of the European pathogen population of Magnaporthe grisea by DNA ngerprinting and pathotype analysis. European Journal of Plant Pathology 103: 363-371
Sharif Gh and Ershad J (1966) A list of infecting fungi on crops, trees and shrubs in Iran. Institute of Pest and Disease Research of Iran, Tehran, Iran
Shull V and Hamer JE (1996) Genetic differentiation in the rice blast fungus revealed by the distribution of the fosbury retrotransposon. Fungal Genetics and Biology 20: 59-69
Swofford DL (2000) PAUP 4. 0 BETA, Phylogenetic Analysis Using Parsimony and Other Methods, Sinauer Associates, Sunderland, Massachusetts, USA
van Etten JL and Daly JM (1980) Production of host-specific toxins by Helminthosporium victoriae and H. maydis in liquid shake culture. Phytopathology 70: 727-729
Xia JQ, Correll JC, Lee FN, Marchetti MA and Rhoads DD (1993) DNA ngerprinting to examine microgeographic variation in the Magnaporthe grisea (Pyricularia grisea ) population in two rice elds in Arkansas. Phytopathology 83: 1029-1035
Xia JQ, Correll JC, Lee FN and Ross WJ (2000) Regional population diversity of Pyricularia grisea in Arkanasas and the in. uence of host selection. Plant Disease 84: 877-844
Zeigler RS, Thome J, Nelson R, Levy M and Correa-Victoria FJ (1994) Lineage exclusion: A proposal for linking blast population analysis to resistance breeding. In: Zeigler RS, Leong RS and Teng PS (eds) Rice Blast Disease (pp 267-292) CABI/IRRI, Wallingford, Oxon, UK
Zeigler RS, Cuoc LX, Scott RP, Bernardo MA, Chen D, Valent B and Nelson RJ (1995) The relationship between phylogeny and virulence in Pyricularia grisea in the Philippines. Phytopathology 85: 443-451
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Javan-nikkhah, M., Mcdonald, B., Banke, S. et al. Genetic structure of Iranian Pyricularia grisea populations based on rep-PCR fingerprinting. European Journal of Plant Pathology 110, 909–919 (2004). https://doi.org/10.1007/s10658-004-5570-x
Issue Date:
DOI: https://doi.org/10.1007/s10658-004-5570-x