Abstract
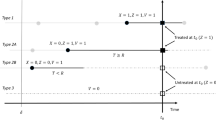
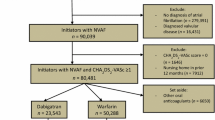
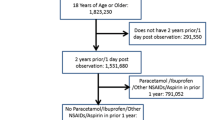
Estimates of the average causal effect (ACE) of warfarin on the risk of bleeding may be confounded by indication as patients at high risk of bleeding are unlikely to be prescribed warfarin. One approach to estimating the ACE is inverse probability of treatment weighting (IPTW). This study was designed to examine the use of IPTW in this setting, and to demonstrate problems with the violation of the positivity assumption. We analyzed a case–control study on 4,028 cases of gastro-intestinal bleeding and 79,239 controls set in the United Kingdom’s General Practice Research Database. Warfarin exposure was defined as a prescription issued in the 90 days before the index date. Secondary analyses were conducted restricted to patients more likely to receive warfarin and with a truncated weight distribution, to exclude subjects highly unlikely to be treated. The estimated association between warfarin use and bleeding was stronger with IPTW [odds ratio (OR): 17.2; 95% confidence interval (CI): 6.5–37.7] than with a standard logistic regression model (OR: 2.1; 95% CI: 1.7–2.5). The presence of large weights (five subjects with stabilized weight >500) indicated a potential violation of the positivity assumption. In the restricted analysis, both IPTW (OR: 2.0; 95% CI: 0.4–9.6) and standard regression (OR: 1.6; 95% CI: 1.3–2.0) were compatible with a meta-analysis of randomized trials inverse probability of treatment weighting is sensitive to the positivity assumption; however, such sensitivity may assist in diagnosing off-support inference.

Similar content being viewed by others
References
Robins JM, Hernan MA, Brumback B. Marginal structural models and causal inference in epidemiology. Epidemiology. 2000;11(5):550–60.
Hernan MA, Brumback B, Robins JM. Marginal structural models to estimate the causal effect of zidovudine on the survival of HIV-positive men. Epidemiology. 2000;11(5):561–70.
Lefebvre G, Delaney JA, Platt RW. Impact of mis-specification of the treatment model on estimates from a marginal structural model. Stat Med. 2008;27(18):3629–42.
Cole SR, Hernan MA, Robins JM, Anastos K, Chmiel J, Detels R, Ervin C, Feldman J, Greenblatt R, Kingsley L, Lai S, Young M, Cohen M, Munoz A. Effect of highly active antiretroviral therapy on time to acquired immunodeficiency syndrome or death using marginal structural models. Am J Epidemiol. 2003;158(7):687–94.
Cole SR, Hernán MA. Constructing inverse probability weights for marginal structural models. Am J Epidemiol. 2008;168(6):656–64.
Mortimer KM, Neugebauer R, van der Laan M, Tager IB. An application of model-fitting procedures for marginal structural models. Am J Epidemiol. 2005;162(4):382–8.
Westreich D, Cole SR. Invited commentary: positivity in practice. Am J Epidemiol. 2010;171(6):674–7.
Hernán MA, Hernández-Diaz S, Robins JM. A structural approach to selection bias. Epidemiology. 2004;15:615–25.
Robins JM, Wang N. Inference for imputation estimators. Biometrika. 2000;87(1):113–24.
Kang JD, Schafer JL. Demystifying double robustness: a comparison of alternative strategies for estimating a population mean from incomplete data. Stat Sci. 2007;22(4):523–39.
Robins JM, Sued M, Lei-Gomez Q, Rotnitzky A. Performance of double-robust estimators when ‘inverse probability’ weights are highly variable. Stat Sci. 2007;22(4):544–59.
Kurth T, Walker AM, Glynn RJ, Chan KA, Gaziano JM, Berger K, Robins JM. Results of multivariable logistic regression, propensity matching, propensity adjustment, and propensity-based weighting under conditions of nonuniform effect. Am J Epidemiol. 2006;163(3):262–70.
Messer LC, Oakes JM, Mason S. Effects of socioeconomic and racial residential segregation on preterm birth: a cautionary tale of structural confounding. Am J Epidemiol. 2010;171(6):664–73.
Imai K, King G, Stuart EA. Misunderstandings between experimentalists and observationalists about causal inference. J R Statist Soc A. 2008;171:481–502.
Delaney JA, Opatrny L, Brophy JM, Suissa S. Drug–drug interactions between antithrombotic medications and the risk of gastrointestinal bleeding. CMAJ. 2007;177(4):347–51.
Kish L. Weighting for unequal Pi. J Off Stat. 1992;8(2):183–200.
Delaney JA, Moodie EE, Suissa S. Modeling blood pressures changes after drug treatment in the General Practice Research Database. Pharmacoepidemiol Drug Saf. 2008;17(6):535–45.
Marston L, Carpenter JR, Walters KR, Morris RW, Nazareth I, Petersen I. Issues in multiple imputation of missing data for large general practice clinical databases. Pharmacoepidemiol Drug Saf. 2010. doi:10.1002/pds.1934 (Epub ahead of print).
Miettinen OS. Estimability and estimation in case-referent studies. Am J Epidemiol. 1976;103(2):226–35.
Newman SC. Causal analysis of case–control data. Epidemiol Perspect Innov. 2006;3:2.
Månsson R, Joffe MM, Sun W, Hennessy S. On the estimation and use of propensity scores in case—control and case—cohort studies. Am J Epidemiol. 2007;166(3):332–9.
Robins JM. Comment on “Choice as an alternative to control in observational studies” by Paul Rosenbaum. Stat Sci. 1999;14:281–93.
Lip GY, Edwards SJ. Stroke prevention with aspirin, warfarin and ximelagatran in patients with non-valvular atrial fibrillation: a systematic review and meta-analysis. Thromb Res. 2006;118(3):321–33.
Filion KB, Delaney JA, Brophy JM, Ernst P, Suissa S. The impact of over-the-counter simvastatin on the number of statin prescriptions in the United Kingdom: a view from the General Practice Research Database. Pharmacoepidemiol Drug Saf. 2007;16(1):1–4.
Ray WA. Evaluating medication effects outside of clinical trials: new-user designs. Am J Epidemiol. 2003;158:915–20.
Delaney JA, Platt RW, Suissa S. The impact of unmeasured baseline effect modification on a marginal structural logistic model: a Monte Carlo study. Eur J Epidemiol. 2009;24:343–9.
Acknowledgments
The authors thank Stephen Cole for comments on an earlier version of this work. This study was funded by the Canadian Institutes of Health Research (CIHR) and the Canadian Foundation for Innovation. Robert Platt holds a Chercheur-boursier award from the Fonds de Recherche en Sante du Quebec (FRSQ) and is a member of the Research Institute of the McGill University Health Centre, which receives operating funds from the FRSQ.
Conflict of interest
The authors declare no conflict of interest.
Ethical review
Ethical review for this study was done by the Independent Scientific Advisory Committee for MHRA database research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Platt, R.W., Delaney, J.A.C. & Suissa, S. The positivity assumption and marginal structural models: the example of warfarin use and risk of bleeding. Eur J Epidemiol 27, 77–83 (2012). https://doi.org/10.1007/s10654-011-9637-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10654-011-9637-7