Abstract
Hatchery production of Pacific salmon for release into the environment is a common tool for conservation and supplementation of depleted stocks. Hatchery conditions are controlled to support high survival rates; however, once released into the environment, managers have little control over conditions experienced by juveniles. Thus, release timing is a critical decision in hatchery operations. This 3-year study used acoustic telemetry to estimate release timing effects on migration survival of fall run Chinook Salmon from the largest hatchery in California: the Coleman National Fish Hatchery. Juvenile Chinook Salmon implanted with JSATS transmitters were released in two or more groups each year, concomitant with the regular hatchery production. Arrays of acoustic monitors were deployed at six locations within the 517 km migration route to estimate reach-specific survival of each release. Mark-recapture models identified both year and release timing effects on reach-specific survival. In years when release effects were well-supported, survival was higher in the earlier release group. Survival in subsequent releases was similar or significantly lower indicating that conditions in the river may decline or stay the same but improvement was not observed. Survival was consistently high in the first 92 km of the migration route and may be related to predator swamping from the release of millions of hatchery fish. Survival declined, and became more variable, within the lower 425 km of the river with the lowest rates observed in the tidal bay-delta. These results indicate that both year and release timing can strongly influence survival and that time between releases likely influences the magnitude of the effect.
Similar content being viewed by others
Introduction
Production of hatchery fish for release into the environment is a tool used by fisheries managers since the late nineteenth century (Naish et al. 2007). For most of the twentieth century, large scale dam construction in North America resulted in significant habitat constriction for anadromous species such as Pacific salmon (Oncorhynchus spp) that have considerable economic, recreational and cultural value (Nehlsen et al. 1991; Sheer and Steel 2006). Advances in fish husbandry led to hatcheries becoming the primary tool used to mitigate for lost production of these valuable species and many of these facilities continue to support impacted fish populations (McIlwain 2003). However, operational decisions can affect the effectiveness of hatchery programs to meet their goals.
One of the most important aspects of hatchery operations is the timing of juvenile release into the environment (Brown and Day 2002; Karppinen et al. 2014). Releasing fish too early can reduce the cost of feeding and maintaining juveniles in the hatchery. However, survival of smaller fish is often lower and hatchery objectives may not be met (McGurk 1999). If fish are held too long, conditions may deteriorate in the environment where they will be released and reduce survival (Brown and Day 2002). An additional consideration for anadromous species is the timing of ocean entry that can have a significant effect on survival to later life stages (Chittenden et al. 2010; Satterthwaite et al. 2014). Where natural populations still exist, minimizing negative interactions between natural and hatchery populations also must be considered (Kostow 2009). Data are needed to inform decision makers to achieve a balance of meeting hatchery program goals while avoiding impacts to natural populations (Blankenship and Leber 1995).
The Coleman National Fish Hatchery (CNFH) in California, USA was built to mitigate for lost Chinook Salmon (O. tshawytscha) production due to the construction of Shasta Dam on the Sacramento River in 1945. The Sacramento River is the largest in California and historically supported large runs of four different races of Chinook Salmon (Yoshiyama et al. 1998). The river continues to support natural populations at reduced abundances with two races (winter run and spring run) listed under the Federal Endangered Species Act. Fall run Chinook Salmon are currently the most abundant natural population and supports commercial and recreational fisheries. In addition to natural production, CNFH has an annual fall run juvenile production target of 12 million. These fish can make substantial contributions to the commercial fishery in the Pacific Ocean and recreational ocean and freshwater fisheries.
Monitoring of CNFH Chinook Salmon is accomplished through the use of coded wire tags. A few weeks prior to release, 25% of juvenile fall run Chinook Salmon are implanted with a coded wire tag and their adipose fin is clipped to assist in identifying tagged individuals when later captured in agency monitoring efforts and in the fishery. Rates of coded wire tag returns to the hatchery indicated a trend of higher returns for earlier releases (USFWS unpublished data). However, coded wire tags are unable to differentiate between survival during different portions of the Chinook Salmon life cycle (freshwater vs. ocean). Additionally, natural populations of fall run Chinook Salmon are present in the Sacramento River and minimizing overlap between natural and hatchery juvenile migration is a consideration in release timing to avoid competition between these populations.
Here we present the results of a 3-year study of the effect of release timing on reach-specific survival rates of CNFH fall run Chinook Salmon from hatchery release to ocean entry. Our goals were to estimate variation in survival between release groups during their 517 km migration to the ocean. Additionally, we sought to identify specific reaches within the migration corridor that might disproportionality contribute to differences in survival among the release groups. These data can be used by hatchery managers to evaluate release strategies as well as identifying river reaches that may be targeted for actions to improve survival.
Methods
Study site
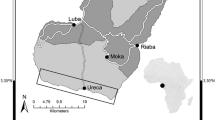
The fall run Chinook Salmon program at CNFH has been in operation since the mid-1940’s, producing fish to mitigate for the construction of Shasta and Keswick Dams. The hatchery was built, and is currently funded by, the US Bureau of Reclamation (USBR) whereas the US Fish and Wildlife Service (USFWS) operates the facility. Although built to mitigate for lost production on the Sacramento River, CNFH is located on the tributary of Battle Creek about 20 miles southeast of the city of Redding, CA and approximately 10 km upstream of the confluence with the Sacramento River (Fig. 1). The broodstock originally selected for CNFH was derived from native Battle Creek and Sacramento River fish (CAHSRG 2012). In most years, fall run juveniles are released directly into Battle Creek and migrate ≈ 517 km to the ocean. In years where conditions in the river are poor (e.g. low flows/high temperatures), portions of the production or the entire production are released directly into the estuary. Release timing is typically a function of when eggs were collected the previous fall, and the time required for juveniles to grow to an average size considered by USFWS as sufficient for release into the environment. In most years, fish are released in two batches between early April and early May from 7 to 21 days apart.
Map depicting the location of the Sacramento River within the state of California and the location of acoustic receiver stations, flow sensors and temperature sensors. Numbers in parentheses after station names are the location in river kilometers from the ocean. The Coleman National Fish Hatchery and cities are represented by star symbols
Below the release site, Battle Creek is a gravel bedded stream of volcanic origin containing riffle-pool sequences interspersed by long runs. Land use is dominated by ranching and farming although riparian forest and floodplain habitats are present. Battle Creek joins the Sacramento River approximately 10 km below the release site. The Sacramento River from the confluence of Battle Creek to Red Bluff [river kilometer (rkm) 462] contains long runs and short riffles with occasional off-channel habitat. From Redd Bluff to Colusa (rkm 314), the river enters a region of intense agricultural land use. The river meanders through this lowland region where riffles are rare and off-channel habitats are common. Downstream from Colusa to the City of Sacramento (rkm 172), the river is increasingly channelized and confined by levees. Banks are often riprapped and there are few off channel habitat features. There are two major flood bypasses in this region and when Sacramento River flows are high enough, water flows into these bypasses and juvenile salmon can access floodplain habitat.
In the City of Sacramento, the river continues to be channelized with rip rapped banks. However, tides begin to influence water level and this is recognized as the beginning of the estuary. Between the City of Sacramento and Chipps Island (rkm 72) is the Sacramento-San Joaquin Delta. This region is a network of channels where water movement changes from tidal but unidirectional at the beginning to fully bidirectional at the end. The main stem Sacramento River leads directly to the end of the Delta; however, fish may enter channels leading to alternate migrations routes. Some of these routes have been found to have poor survival rates relative to the main stem (Perry et al. 2010). West of the Delta at Chipps Island is Suisun Bay and San Pablo Bay where water becomes increasingly saline. Finally, fish enter Central Bay that is directly connected to the ocean at the Golden Gate.
Acoustic tags and receiver array
In all study years, the Juvenile Salmon Acoustic Telemetry System (JSATS) was used to estimate reach specific survival. In 2012, Lotek Wireless model L-AMT-1.1 transmitters were used. These tags weighed 0.3 g in air and allowed tagging fish as small as 6.0 g without exceeding a tag burden of 5% body weight (Ammann et al. 2013; Brown et al. 2011). Acoustic receivers deployed in 2012 were all Lotek Wireless model WHS4000. This array was used by multiple concurrent studies and a subset of six receiver locations was selected for analysis of survival in 2012 based on their delimiting of relevant features (e.g. confluences, long-term monitoring locations, habitat transitions; Fig. 1, Table 1). Additional receiver stations deployed as part of concurrent studies were used to identify potential predator-type detections but were not included in survival models. At most sites, multiple receivers were deployed to provide adequate coverage. The final station located at the Golden Gate Bridge was comprised of two independent receiver lines ~1 km apart to facilitate calculation of detection probabilities at this site and allow estimation of survival through the final reach.
In 2013 and 2016, Advanced Telemetry System (ATS) model SS300 transmitters were used in the study. These transmitters also weighed 0.3 g in air. ATS model SR3000 receivers were added into the array and paired with a Lotek receiver at most sites. Some receiver locations were added between study years and others were removed when data indicated poor detection probabilities at those sites (Fig. 1, Table 1). However, six receiver stations that were consistently deployed in all study years were included in the survival analysis. Additional stations were used only to identify potential predator detections.
A tag life evaluation was performed at CNFH in 2012 and at the National Marine Fisheries Service Laboratory in Santa Cruz, California in 2013 and 2016. In all years the study occurred in an outdoor tank of freshwater measuring 2 m in diameter and 1 m deep. Sample sizes were, 21 (Lotek 2012), 43 (ATS 2013) and 29 (ATS 2016) tags. For the 2012 study, tags were activated and then placed on the bottom of the circular midway between the center and the edge of the tank. A Lotek WHS 4000 JSATS receiver was placed in the tank and the study continued until all tags had failed. In the 2013 and 2016 study, tags were individually taped to a 6.4 mm nylon line about 1 m long. There were multiple lines each with 9 or 10 tags. Tags were spaced about 5 cm apart on the line. The lines were hung in the tank with a Hobo pendant logger that recorded temperature every 15 min. Two Lotek WHS 4000 JSATS receivers were suspended in the center of the tank so the negatively buoyant receivers were off the bottom and below the surface. Both receivers were equipped with beacon transmitters that pinged at 60 s intervals. A small electric submersible pump created a slight circular current in the tank. Receivers were downloaded every 1–4 weeks, at which time the memory was cleared and the receiver’s internal clock was reset. The study continued until all tags had failed.
Tagging and release procedures
In all years, the average size of production fish at the time of release was less than that required to achieve the maximum tag burden. Thus, fish were sorted prior to the tagging day to obtain individuals of appropriate size. Two different anesthetics were used during the study. In 2012 and 2013 MS-222 was used and in 2016 Aqui-S was used due to a ban on MS-222 by the California Department of Fish and Wildlife for fish being released into the environment before the anesthetic has cleared the experimental fish. Chinook Salmon were individually weighed to determine if they met the weight criteria and then immersed in anesthetic until they lost equilibrium. Fish were then measured to the nearest mm fork length (FL) and transferred to a cradle that held the fish ventral side up while a maintenance dose of anesthetic was pumped over the gills. A small (~1 cm) incision was made anterior to the pelvic girdle and just off of the midline. A JSATS transmitter was inserted and the incision was closed with a single suture. Fish were then transferred to a flow through holding tank where they were allowed to recover. Once fish regained equilibrium, they were transferred to a holding container within a raceway at CNFH and were allowed to recover at least 12 h prior to release.
Twenty-four juvenile Chinook Salmon were implanted with dummy tags in 2012 to evaluate potential tagging-related mortality. These fish went through the same surgical procedures as the fish released with live tags and were placed in a flow-through circular tank at CNFH. Mortality of these fish was evaluated for 30 days which was the approximate anticipated tag life. A comprehensive tag effects study for JSATS transmitters implanted in CNFH fall run was performed in 2013 and is reported in (Singer et al. 2019).
Releases of tagged fish occurred on the day following tagging for all releases except for the final release of 2016. This release was delayed by 1 day due to a last-minute change in the hatchery release schedule. These dates corresponded to the release of production fish from CNFH. As regular production fish were pumped from the raceways into Battle Creek, ten tagged fish were released every 15 min until all fish had been released. The release of tagged fish occurred at the mouth of the pipe where fish were being pumped from the raceway to simulate the conditions experienced by the regular production to the greatest extent possible. Each production release from the hatchery contained 2–6 million juvenile Chinook salmon.
Data analysis
Encounter histories for each tag released were constructed using a series of data filters. Each receiver’s detection data was processed independently of other receivers. First, a time sorted and indexed list of detections for each TagID in the file was created. The date-time of the detection must have been less than the date-time of tagging plus 1.5 times the warranty life of the tag for that tagged fish. Any detection that occurred less than 0.3 s after the previous detection of the TagID was classified as a multipath (echo) and removed. A time window of 16.6 times the pulse rate interval (PRI) was used to find four or more detections of the TagID. The estimated PRI for consecutive detections must be within 20% of the nominal PRI, if not then the first detection is removed, the time window reset, and the search continued. If the four detects passed the above criteria and the standard deviation of the three estimated PRIs was less than 0.025, the four detects are considered valid. This level was chosen by examining standard deviations from known valid detects and from known false positives. Additionally, confirmed detections of tagged Chinook Salmon that had potentially been consumed by predators were identified using a simple algorithm along with tag detections at the six experimental receiver stations and additional JSATS receiver stations in the study area that were deployed for concurrent studies. Tags that always moved in a downstream direction were classified as Chinook Salmon. If a tag made an upstream movement, they had to make a subsequent downstream movement greater than the distance they moved upstream to be classified as Chinook Salmon. If they were classified as a predator, the receiver where they were detected prior to the upstream movement was considered the last detection prior to the predation event.
The effects of year and release timing on reach specific survival and detection probabilities were estimated with Cormack-Jolly-Seber models (CJS) using the encounter histories described above in Program Mark (White and Burnham 1999). Three models were constructed to elucidate the weight of evidence for release timing to explain reach specific survival of Chinook Salmon. The first model estimated a constant survival value (intercept only) for each reach without year or release group represented. The second model included a variable indicating release timing (first, second or third release of the year) but no year effects. The third model included year effects but no release timing variable and the final model included both year and release timing effects. An information theoretic approach was employed to identify the best approximating model. Akaike’s Information Criterion corrected for sample size (AICc) was calculated for each model. The model with the lowest AICc value was considered the best approximating model. The AICc value for this model was subtracted from the AICc value of the other candidate models to calculate ∆AICc. A model with a ∆AICc ≥7.0 was considered to have relatively little support (Burnham and Anderson 2002). Additionally, AICc weights were calculated based on the ∆AICc values. These weights were interpreted as the probability that a given model was the best explanation of the data.
Reach specific survival estimates generated by candidate models were converted to a standardized value of survival (S•10 km−1) to facilitate comparison among reaches of different lengths.
Environmental data
To describe the environmental conditions experienced by tagged Chinook Salmon during migration, weighted averages of water temperature and river flow were calculated.
where, Pi,j is the weighted average of the parameter of interest (flow or temperature) in year i during release j, Pd is the mean daily value of the parameter, td is the daily proportion of tags passing by the acoustic station nearest to the environmental station and T is the total number of tags that pass the station.
Due to the large size of the migration route and diversity of aquatic habitats, characterizing the environmental conditions experienced by tagged Chinook Salmon could not be limited to a single location. To better describe the conditions experienced through the entire migration route, weighted averages of flow and temperature were calculated at multiple environmental stations. Stations maintained by the United States Geological Survey and the California Department of Water Resources were selected to represent locations of major tributary inputs, diversions and habitat changes (Fig. 1; Table 1). Flow and temperature stations closest to an acoustic receiver station were selected to increase the resolution of fish-environment relationships although this distance could be from 0 to 27 km (Table 1). Not all stations within the migration route contained both temperature and flow sensors (Table 1). Thus, there are a limited number of temperature and flow estimates relative to the number of acoustic receiver locations (Fig. 1).
Results
Releases
In spring 2012, Chinook Salmon implanted with JSATS transmitters were released from CNFH concurrent with the regular hatchery production. The first release occurred on 19 April and the second occurred 12 days later on 1 May. Three hundred fish were tagged with 150 released on each occasion. However, during the surgical procedures, tags were turned on and then placed in a sterilization solution where the activated tags were in close proximity. This resulted in some tags being turned off via acoustic signals from the other activated tags. Following inspection of filtered detection data on receivers at the release site and deployed within Battle Creek upstream of the first station used in the survival model, it was determined that 170 of the tags remained active at the time of release. Thus, we decided the data were sufficient for analysis. No mortality of the dummy tagged fish was observed after 30 days post-surgery. Mean time to tag failure was 28.8 days with a range between 0.2 and 45.6 days. One tag failed within the mean time study fish were observed passing the final receiver station (13.6 days). Methods are available to correct survival estimates for tag failure (Townsend et al. 2006; Holbrook et al. 2013). However, variation in corrected and uncorrected values is low (<1%) when tag survival rates are high as was observed here (Townsend et al. 2006). Thus, we chose not to correct values for the 2012 releases. Tagged Chinook Salmon in each release group were a similar size with a mean FL of 84.5 ± 3.0 mm for the first release group and 85.4 ± 2.9 mm for the second release group. In 2013 two production releases occurred from CNFH. The first occurred on 10 April and the second 14 days later on 24 April. On both dates, 150 JSATS implanted Chinook Salmon were released with the production fish. Mean time to tag failure was 38.6 days with a range between 31.9 and 45.3 days. The minimum value (31.9 d) was greater than the time it took study fish to pass the final receiver line at the Golden Gate Bridge (<20 d). No mortality was observed for fish used in the 2013 tag effects study (Singer et al. 2019; G. Singer, pers. comm.). The size of tagged Chinook Salmon was similar between releases with a mean of 85.1 ± 2.5 mm for the first release and 84.0 ± 2.3 mm for the second release.
Due to drought conditions in the Sacramento River Watershed, a decision was made by the USFWS to perform a single release in 2014 and truck the remaining fish from CNFH to the San Francisco Estuary to avoid poor conditions anticipated in the Sacramento River. In 2015, all fish produced at CNFH were trucked to the estuary. Since no paired releases were made in these years, no releases of JSATS tagged Chinook Salmon were performed.
A total of 597 tags was available in spring 2016 due to the lack of releases during the drought period when fish were trucked to the estuary. This provided an opportunity to increase the sample size within each release. Three releases of JSATS implanted Chinook Salmon occurred along with CNFH production. The first release occurred 7 April (200 tags), the second on 12 April (200 tags) and the third on 29 April (197 tags). The third release on 29 April occurred 1 day after the release of production fish from CNFH due to a last-minute schedule change at the hatchery. Tag life study results indicated the range of days until tag failure (31.6–74.3) was greater than the time it took for any study fish to pass the final receiver location at the Golden Gate Bridge (<20 d). No fish were held to evaluate tagging-related mortality in 2016; however, the same surgical and release procedures were used as in previous years when no mortality was observed. Similar to previous years, the size of tagged Chinook Salmon was similar among releases with a value of 83.8 ± 2.6 mm for the first release, 82.9 ± 2.0 mm for the second release and 83.7 ± 3.0 mm for the third release.
Survival models
The model selection exercise identified two models that were well supported by the data. The best model included both year and release timing effects on reach specific survival (Table 2). This model had an AICc weight of 0.75 suggesting there was good support for this model being the best. The second model included only year effects and had a ∆AIC of 2.2 and an AICc weight of 0.25 (Table 2). Although this model had less support than the model with year and release effects, there was enough support that the model could not be eliminated from consideration. No other candidate model had a ∆AICc <10.5.
In 2012, the coefficient for a release effect had good support with a 95% confidence interval that did not include zero (Table 3). The value of the coefficient was negative indicating that survival of tagged Chinook Salmon was lower in release two relative to release one. In 2013, there was not a well-supported release effect with a 95% confidence interval for the coefficient that included zero (Table 3). For the 2016 releases, the coefficient for the effect of being in the second release group relative to the first had poor support with a 95% confidence that included zero. However, the coefficient for the effect of being in the third release group relative to the first had good support. This coefficient was negative indicating survival of tagged Chinook Salmon was lower in the third group relative to the first (Table 3).
Standardized survival estimates (S•10 km−1) revealed a general trend of higher survival in the first three reaches (CNFH to Woodson Bridge) relative to the last three reaches (Woodson Bridge to the Golden Gate Bridge (Fig. 2). Within Battle Creek between CNFH and the confluence with the Sacramento River, mean S•10 km−1 was ≥94.4% in all study years. In the second reach, between the Battle Creek Confluence and Salt Creek, survival was consistently high among all years (≥98.9%•10 km−1). Survival was also relatively high in the third reach between Salt Creek and Woodson Bridge but also more variable than reach two (Fig. 2). The reach between Woodson Bridge and Freeport had the lowest survival of all the non-tidal reaches and was variable among study years. Survival in the tidal freshwater portion of the estuary (Freeport to Benicia Bridge) was consistently lower than any of the reaches upstream though confidence intervals overlapped and there was variation among study years. The lowest survival rate was observed between Benicia Bridge and the Golden Gate in 2013. However, low sample sizes in this final reach resulted in uncertain estimates during all study years.
Reach specific standardized survival estimates (S•10 km−1) and 95% confidence intervals in each of the three study years. Reaches were defined by the acoustic receiver stations listed in Table 1
Environmental conditions during migration
There was a general pattern of increasing water temperature from the most upstream station at Balls Ferry (rkm 515) to the most downstream station at Martinez (rkm 52; Fig. 3). Temperature variation between releases >1 °C occurred at the Red Bluff Diversion Dam (rkm 462) station in 2012 (1.6 °C warmer during release 1) and in 2016 (1.3 and 1.7 °C warmer in releases 1 and 3 relative to release 2). The greatest temperature variation among releases occurred in 2013. During the second 2013 release weighted average water temperature was 3.1 and 1.6 °C warmer at the Freeport and Martinez stations, respectively.
Weighted average water temperature at four stations within the migration route of Chinook Salmon juveniles. Temperature was weighted by the proportion of tags from each release passing by the receiver array located closest to each station. The location of stations by river kilometer is listed in Table 1
Flow magnitude generally increased from upstream to downstream in all years with the greatest increases occurring below the confluence of the Feather River and American River with the Sacramento River (Freeport station). During 2012, flow magnitude was 29–37% higher at the Sacramento River stations during the first release (Fig. 4). Battle Creek was the only station where flow magnitude was greater during the second release (19%). Flow variation among releases was more varied in 2013. At two stations (Battle Creek and Hamilton City) flow was greater during the first release and at two stations it was greater during the second release (Bend Bridge and Freeport; Fig. 4). The largest difference occurred in Battle Creek where flow was ~25% higher in the first release. The greatest between-release variation in flow occurred in 2016. There were only small differences between releases 1 and 2 (<11.5%). However, flows during release three were substantially lower than the first two releases (Fig. 4).
Weighted average river flow at four stations within the migration route of Chinook Salmon juveniles. Flow was weighted by the proportion of tags from each release passing by the receiver array located closest to each station. The location of stations by river kilometer is listed in Table 1
Discussion
Mark-recapture modeling of acoustically tagged CNFH fall run Chinook Salmon indicated that both year and release timing were important influences on migration survival between release and ocean entry. Release effects were well supported in 2012, and between release 1 and 3 in 2016. In both cases, survival was higher for fish released in the first group of the year. These releases were made 12 and 22 days apart in 2012 and 2016 respectively. Release effects were not well supported in 2013, or between releases 1 and 2 in 2016 and time between these releases was 14 days and 5 days, respectively. This pattern suggests conditions for survival in the river are better earlier in the spring and decline or stay the same during subsequent releases. Weber and Fausch (2004) examined natural-origin Chinook Salmon densities in the Sacramento River and Battle Creek before during and after production releases from CNFH. They found that densities of Chinook Salmon peaked prior to the first hatchery release. This may suggest that natural-origin fish initiate migration when conditions for survival are more favorable (earlier in spring) and hatchery releases are occurring as environmental conditions are declining.
Water temperature has been shown to have a significant influence on juvenile Chinook Salmon survival probabilities during migration (Connor et al. 2003; Newman 2003; Zeug and Cavallo 2013). Tagged Chinook Salmon in this study did not experience temperatures within the range where growth becomes impaired (≥21 °C; Marine and Cech 2004). However, Marine and Cech (2004) reported higher susceptibility of juvenile Chinook Salmon to predation at temperatures between 17 and 24 °C. Temperatures ≥17 °C were never observed at the two upstream temperature stations whereas tagged Chinook Salmon frequently experienced these temperatures within the tidal bay-delta (Freeport and Martinez stations). Although water temperature varied among stations within the migration corridor, there was little variation at the same station between releases suggesting that temperature was unlikely to drive observed differences in survival among release groups.
Flow magnitude has also been related to survival of Chinook Salmon smolts during migration (Giorgi et al. 1997; Smith et al. 2002; Zeug et al. 2014; Henderson et al. 2018). In release pairs with significant timing effects (release 1 vs 2 in 2012 and release 1 vs 3 in 2016), flow magnitude was 12.8% to 53.5% greater in the Sacramento River during the release with higher survival. In release pairs with no significant effect (release 1 vs 2 in 2013 and release 1 vs 2 in 2016), flow varied between 20.3% lower and 9% higher during the first release. The magnitude of flow variation that occurred during this 3-year study was less than that observed in previous studies of Chinook Salmon survival in the Sacramento River when quantitative relationships were identified (Michel et al. 2015; Henderson et al. 2018). Additionally, tributary accretions and large water diversions result in substantial changes in flow magnitude throughout the migration route, complicating attempts to identify the spatial location of flow effects. This study was not designed to specifically test for flow effects. However, the results presented here suggest additional studies directed at spatially explicit flow-survival relationships are needed to identify release tactics that may be effective to increase survival of hatchery releases (e.g., performing releases at flows that maximize survival).
In addition to differences in freshwater survival between releases observed here, the shift in timing of ocean entry resulting from variable release timing can influence survival to later life stages. Muir et al. (2006) evaluated smolt-to-adult return rates for Chinook Salmon in the Columbia River that had been transported by barge or migrated naturally. They reported that the difference in timing of ocean entry (2–4 weeks) was the most parsimonious explanation for differential survival to adult return. This difference in timing is similar to that experienced by release groups of CNFH Chinook Salmon in the current study (5–22) days. Satterthwaite et al. (2014) reported release timing was a good predictor of ocean recoveries of coded wire tagged hatchery-origin Chinook Salmon with higher rates for earlier releases. In that study fish were released directly into the San Francisco Estuary. The results presented here suggest that differences in freshwater survival could further magnify release effects on survival to later life stages. Irvine et al. (2013) found an interaction between smolt size and release timing for some hatchery populations of coho Salmon (Oncorhynchus kisutch) and suggested optimal release timing may be a moving target. Although our results demonstrated differences in freshwater survival, ocean survival may be the major driver of differential adult return rates during some years.
Modeling of survival data revealed variation among reaches within the migration corridor that provides information useful for consideration of migration success. Survival was consistently high in the first ~92 km (rkm 517–425) following release despite variations in flow up to 28% between some releases. Study fish were released with large numbers of hatchery production fish (2–6 million) which may have resulted in a predator swamping effect (Ims 1990; Petersen and DeAngelis 2000). Furey et al. (2016) reported that survival of juvenile sockeye salmon (Oncorhynchus nerka) increased with the density of conspecifics in the Chiko River, British Columbia. In all years, survival declined between Woodson Bridge (rkm 425) and Freeport (rkm 152) though the magnitude of decline varied among years. Michel et al. (2015) reported the opposite pattern for late-fall Chinook Salmon in the Sacramento River where survival was lower in upper reaches and consistently higher in the lower reaches. Late-fall Chinook salmon in the Michel et al. (2015) study migrate during the winter as yearlings whereas fall run in the current study migrate as young-of-the-year in spring. Striped bass (Morone saxatalis) aggregate in the downstream reaches during their spring spawning migration which overlaps with the fall run migration period (DuBois and Harris 2016). Higher abundance of striped bass has been linked to lower survival probabilities for migrating Chinook Salmon in the Mokelumne River, California (Sabal et al. 2016). Thus, variation between the reach-specific survival results presented here and previous studies may be related to differences in piscivore abundance within the river and susceptibility of individuals (large yearlings vs young-of-the-year).
Several sources of uncertainty in this study should be considered when interpreting the results. First, size restrictions needed to meet tag burden requirements resulted in a tagged sample of Chinook Salmon that was larger than the untagged hatchery production group. Thus, it is unknown how much observed survival rates may deviate from those of the untagged population. During the 2012 study, a single tag failure was observed within the time required to complete migration which biased the survival estimates low. Previous research indicates the observed failure rates would have little effect on the accuracy of estimates (Townsend et al. 2006) but should still be considered. Survival estimates in the tidal estuary had greater uncertainty than in more upstream reaches. Mortality upstream reduced the available sample size for estimation with only 105 of the 1067 tagged fish released in Battle Creek arriving at the boundary of the Delta at Freeport on the Sacramento River (Supplement 1). If differences in survival among releases are strong in the Delta (or bay), the small number of fish arriving there from CNFH and the large confidence intervals resulting from low sample size likely obscure any difference. Regardless, it was clear that survival rates in tidal areas were often low and variable; a pattern similar to that reported in other studies (Perry et al. 2010; Hearn et al. 2014; Buchanan et al. 2018). Future efforts should consider larger sample sizes at release or paired releases at downstream locations to increase the precision of survival estimates in these tidal reaches. The results presented here indicate that release timing can have an effect on juvenile Chinook Salmon survival during migration to the ocean; however, this effect is variable among years and the chance of observing an effect within years likely increases with time between releases. The underlying mechanisms of Chinook Salmon mortality within the migration route are not well understood and likely include both biological and environmental components. Future studies may improve predictability of survival by focusing on these mechanisms at smaller spatial scales; particularly in reaches where low and variable survival was observed. Hatchery production will continue to be a tool to support fall run Chinook Salmon stocks in the Sacramento River and throughout the species’ range. Better understanding survival and behavior of juveniles following release can help achieve hatchery goals while minimizing impacts on natural stocks.
References
Ammann AJ, Michel CJ, MacFarlane RB (2013) The effects of surgically implanted acoustic transmitters on laboratory growth, survival and tag retention in hatchery yearling Chinook salmon. Environ Biol Fish 96:135–143
Blankenship HL, Leber KM (1995) A responsible approach to marine stock enhancement. Am Fish Soc Symp 15:167–175
Brown C, Day RL (2002) The future of stock enhancements: lessons for hatchery practice from conservation biology. Fish Fish 3:79–94
Brown RS, Harnish RA, Carter CM, Boyd JW, Deters KA, Eppard MB (2011) An evaluation of the maximum tag burden for implantation of acoustic transmitters in juvenile Chinook Salmon. N Am J Fish Manag 30:499–505
Buchanan RA, Brandes PL, Skalski JR (2018) Survival of juvenile fall-run Chinook Salmon through the San Joaquin River Delta, California, 2010-2015. N Am J Fish Manag 38:663–679
Burnham KP, Anderson DR (2002) Model selection and multimodel inference; a practical information-theoretic approach, 2nd edn. Springer, New York, p 488
California Hatchery Scientific Review Group: CHSRG (2012) Prepared for the United States fish and wildlife service and Pacific states Marine fisheries commission, Sacramento, CA pp100
Chittenden CM, Jensen JLA, Ewart D, Anderson S, Balfry S, Downey E, Eaves A, Saksida S, Smith B, Vincent S, Welch D, McKinley RS (2010) Recent salmon declines: a result of lost feeding opportunities due to bad timing? PLoS One 5(8):e12423. https://doi.org/10.1371/journal.pone.0012423
Connor WP, Burge HL, Yearsley JR, Bjornn TC (2003) Influence of flow and temperature on survival of wild subyearling fall Chinook Salmon in the Snake River. N Am J Fish Manag 23:362–375
DuBois J, Harris MD (2016) 2016 adult striped bass tagging cruise report. California Department of Fish and Wildlife. Bay Delta Region, Stockton, CA http://www.dfg.ca.gov/delta/data/stripedbass/bibliography.asp
Furey NB, Hinch SG, Bass AL, Middleton CT, Minke-Martin V, Lotto AG (2016) Predator swamping reduced predation risk during nocturnal migration of juvenile salmon in a high-mortality landscape. J Anim Ecol 85:948–959
Giorgi AE, Hillman TW, Stevenson JR, Hays SG, Peven CM (1997) Factors that influence the downstream migration rates of juvenile salmon and steelhead through the hydroelectric system in the mid-Columbia River basin. N Am J Fish Manag 17:268–282
Hearn AR, Chapman ED, Singer GP, Brostoff WN, LaCivita PE, Klimley AP (2014) Movements of out-migrating late-fall run Chinook salmon (Oncorhynchus tshawytscha) smolts through the San Francisco Bay estuary. Environ Biol Fish 97:851–863
Henderson MJ, Iglesias IS, Michel CJ, Ammann AJ, Huff DD (2018) Estimating spatial-temporal differences in Chinook Salmon outmigration survival with habitat- and predation-related covariates. Can J Fish Aquat Sci 76:1549–1561
Holbrook CM, Perry RW, Brandes PL, Adams NS (2013) Adjusting survival estimates for premature transmitter failure: a case study from the Sacramento-san Joaquin Delta. Environ Biol Fish 96:165–173
Ims RA (1990) On the adaptive value of reproductive synchrony as a predator-swamping strategy. Am Nat 136:485–498
Irvine JR, O’Neill M, Godbout L, Schnute J (2013) Effects of smolt release timing and size on the survival of hatchery-origin coho salmon in the strait of Georgia. Prog Oceanogr 115:111–118
Karppinen P, Jounela P, Huusko R, Erkinaro J (2014) Effects of release timing on migration behavior and survival of hatchery-reared Atlantic salmon smolts in a regulated river. Ecol Freshw Fish 23:438–452
Kostow K (2009) Factors that contribute to the ecological risks of salmon and steelhead hatchery programs and some mitigating strategies. Rev Fish Biol Fish 19:9–31
Marine KR, Cech JJ (2004) Effects of high water temperature on growth, smoltification, and predator avoidance in juvenile Sacramento River Chinook salmon. N Am J Fish Manag 24:198–210
McGurk MD (1999) Size dependence of natural mortality rate of sockeye salmon and kokanee in freshwater. N Am J Fish Manag 19:376–396
McIlwain TD (2003) NMFS involvement with stock enhancement as a management tool. In: Nakamura Y, JP MV, Leber K, Neidig C, Fox S, Churchill K (eds) Ecology of aquaculture species and enhancement of stocks. Proceedings of the thirtieth U.S. – Japan meeting on aquaculture. Sarasota, Florida, 3-4 December. UJNR technical report no. 30. Mote Marine Laboratory, Sarasota
Michel CJ, Ammann AJ, Lindley ST, Sandstrom PT, Chapman ED, Thomas MJ, Singer GP, Klimley AP, MacFarlane RB (2015) Chinook salmon outmigration survival in wet and dry years in California’s Sacramento River. Can J Fish Aquat Sci 72:1749–1759
Muir WD, Marsh DM, Sandford BP, Smith SG, Williams JG (2006) Post-hydropower system delayed mortality of transported Snake River stream-type Chinook salmon: unraveling the mystery. Trans Am Fish Soc 135:1523–1534
Naish KA, Taylor JE III, Levin PS, Quinn TP, Winton JR, Huppert D, Hilborn R (2007) An evaluation of the effects of conservation and fishery enhancement hatcheries on wild populations of salmon. Adv Mar Biol 53:61–194
Nehlsen W, Williams JE, Lichatowich JA (1991) Pacific salmon at the crossroads: stocks at risk from California, Oregon, Idaho and Washington. Fisheries 16:4–21
Newman KB (2003) Modelling paired release-recovery data in the presence of survival and capture heterogeneity with application to marked juvenile salmon. Stat Model 3:157–177
Perry RW, Skalski JR, Brandes PL, Sandstrom PT, Klimley AP, Ammann A, MacFarlane B (2010) Estimating survival and migration route probabilities of juvenile Chinook salmon in the Sacramento-san Joaquin Delta. N Am J Fish Manag 30:142–156
Petersen JH, DeAngelis DL (2000) Dynamics of prey moving through a predator field: a model of migrating juvenile salmon. Math Biosci 165:97–114
Sabal M, Hayes S, Merz J, Setka J (2016) Habitat Alterations and a nonnative predator, the Striped Bass, increase native Chinook Salmon mortality in the Central Valley, California. N Am J Fish Manag 36:309-320
Satterthwaite WH, Carlson SM, Allen-Moran SD, Vincenzi S, Bograd SJ, Wells BK (2014) Match-mismatch dynamics and the relationship between ocean-entry timing and relative ocean recoveries of Central Valley fall run Chinook Salmon. Mar Ecol Prog Ser 511:237–248
Sheer MB, Steel EA (2006) Lost watersheds: barriers, aquatic habitat connectivity, and salmon persistence in the Willamette and lower Columbia River basins. Trans Am Fish Soc 135:1654–1669
Singer GP, Hansen MJ, Ho KV, Lee KW, Cocherell DE, Klimley AP, Rypel AL, Fangue NA (2019) Behavioral response of juvenile Chinook Salmon to surgical implantation of micro-acoustic transmitters. Trans Am Fish Soc 148:480–492
Smith SG, Muir WD, Williams JG (2002) Factors associated with travel time and survival of migrant yearling Chinook Salmon and steelhead in the lower Snake River. N Am J Fish Manag 22:385–405
Townsend RL, Skalski JR, Dillingham STW (2006) Correcting bias in survival estimation resulting from tag failure in acoustic and radiotelemetry studies. J Agric Biol Environ Stat 11:1–14
Weber ED, Fausch KD (2004) Abundance and size distribution of ocean-type juvenile Chinook salmon in the upper Sacramento River margin before and after hatchery releases. N Am J Fish Manag 24:1447–1455
White GC, Burnham KP (1999) Program MARK: survival estimation from populations of marked animals. Bird Study 46:120–138
Yoshiyama RM, Fisher FW, Moyle PB (1998) Historical abundance and decline of Chinook salmon in the Central Valley region of California. N Am J Fish Manag 18:487–521
Zeug SC, Cavallo BJ, (2013) Influence of estuary conditions on the recovery rate of coded-wire-tagged Chinook salmon ( ) in an ocean fishery. Ecol Freshw Fish 22(1):157–168
Zeug SC, Sellheim K, Watry C, Wikert JD, Merz J (2014) Response of juvenile Chinook salmon to managed flow: lessons learned from a population at the southern extent of their range in North America. Fish Manag Ecol 21:155–168
Acknowledgments
We thank the United States Fish and Wildlife Service and the staff of Coleman National Fish Hatchery for their support and assistance with this study. Funding was provided by the California Department of Fish and Wildlife Ecosystem Restoration Program. Staff from the UC Davis Biotelemetry Laboratory and the National Marine Fisheries Service, deployed and maintained the receiver array throughout the migration corridor and participated in surgeries to implant JSATS transmitters. This study would not have been possible without the assistance of many staff members from Cramer Fish Sciences, USFWS, UC Davis and NMFS.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 31 kb)
Rights and permissions
About this article
Cite this article
Zeug, S.C., Null, R., Brodsky, A. et al. Effect of release timing on apparent survival of juvenile fall run Chinook Salmon from Coleman National Fish Hatchery. Environ Biol Fish 103, 411–423 (2020). https://doi.org/10.1007/s10641-020-00968-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10641-020-00968-7