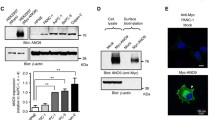
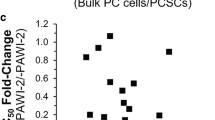
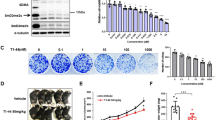
Abstract
Pancreatic cancer is one of the most lethal human neoplasms, and despite advances in the understanding of the molecular complexity involved in the development and progression of this disease, little of this new information has been translated into improvements in therapy and prognosis. Ezrin (EZR) is a protein that regulates multiple cellular functions, including cell proliferation, survival, morphogenesis, adhesion, and motility. In pancreatic cancer, EZR is highly expressed and reflects an unfavorable prognosis, whereas EZR silencing ameliorates the malignant phenotype of pancreatic cancer cells. NSC305787 was identified as a pharmacological EZR inhibitor with favorable pharmacokinetics and antineoplastic activity. Here, we endeavored to investigate the impact of EZR expression on survival outcomes and its associations with molecular and biological characteristics in The Cancer Genome Atlas pancreatic adenocarcinoma cohort. We also assessed the potential antineoplastic effects of NSC305787 in pancreatic cancer cell lines. High EZR expression was an independent predictor of worse survival outcomes. Functional genomics analysis indicated that EZR contributes to multiple cancer-related pathways, including PI3K/AKT/mTOR signaling, NOTCH signaling, estrogen-mediated signaling, and apoptosis. In pancreatic cells, NSC305787 reduced cell viability, clonal growth, and migration. Our exploratory molecular studies identified that NSC305787 modulates the expression and activation of key regulators of the cell cycle, proliferation, DNA damage, and apoptosis, favoring a tumor-suppressive molecular network. In conclusion, EZR expression is an independent prognosis marker in pancreatic cancer. Our study identifies a novel molecular axis underlying the antineoplastic activity of NSC305787 and provides insights into the development of therapeutic strategies for pancreatic cancer.



Similar content being viewed by others
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. CA Cancer J Clin 69:7–34
Stathis A, Moore MJ (2010) Advanced pancreatic carcinoma: current treatment and future challenges. Nat Rev Clin Oncol 7:163–172
TCGA (2017) Integrated Genomic Characterization of Pancreatic Ductal Adenocarcinoma. Cancer Cell 32(185–203):e113
Neisch AL, Fehon RG (2011) Ezrin, Radixin and Moesin: key regulators of membrane-cortex interactions and signaling. Curr Opin Cell Biol 23:377–382
Li N, Kong J, Lin Z, Yang Y, Jin T, Xu M, Sun J, Chen L (2019) Ezrin promotes breast cancer progression by modulating AKT signals. Br J Cancer 120:703–713
Bretscher A, Reczek D, Berryman M (1997) Ezrin: a protein requiring conformational activation to link microfilaments to the plasma membrane in the assembly of cell surface structures. J Cell Sci 110(Pt 24):3011–3018
Kawaguchi K, Yoshida S, Hatano R, Asano S (2017) Pathophysiological Roles of Ezrin/Radixin/Moesin Proteins. Biol Pharm Bull 40:381–390
Yeh TS, Tseng JH, Liu NJ, Chen TC, Jan YY, Chen MF (2005) Significance of cellular distribution of ezrin in pancreatic cystic neoplasms and ductal adenocarcinoma. Arch Surg 140:1184–1190
Cui Y, Li T, Zhang D, Han J (2010) Expression of Ezrin and phosphorylated Ezrin (pEzrin) in pancreatic ductal adenocarcinoma. Cancer Invest 28:242–247
Oda Y, Aishima S, Morimatsu K, Hayashi A, Shindo K, Fujino M, Mizuuchi Y, Hattori M, Tanaka M (2013) Differential ezrin and phosphorylated ezrin expression profiles between pancreatic intraepithelial neoplasia, intraductal papillary mucinous neoplasm, and invasive ductal carcinoma of the pancreas. Hum Pathol 44:1487–1498
Zhou J, Feng Y, Tao K, Su Z, Yu X, Zheng J, Zhang L, Yang D (2014) The expression and phosphorylation of ezrin and merlin in human pancreatic cancer. Int J Oncol 44:2059–2067
Piao J, Liu S, Xu Y, Wang C, Lin Z, Qin Y (2015) Ezrin protein overexpression predicts the poor prognosis of pancreatic ductal adenocarcinomas. Exp Mol Pathol 98:1–6
Quan C, Sun J, Lin Z, Jin T, Dong B, Meng Z, Piao J (2019) Ezrin promotes pancreatic cancer cell proliferation and invasion through activating the Akt/mTOR pathway and inducing YAP translocation. Cancer Manag Res 11:6553–6566
Xu J, Zhang W (2021) EZR promotes pancreatic cancer proliferation and metastasis by activating FAK/AKT signaling pathway. Cancer Cell Int 21:521
Bulut G, Hong SH, Chen K, Beauchamp EM, Rahim S, Kosturko GW, Glasgow E, Dakshanamurthy S, Lee HS, Daar I, Toretsky JA, Khanna C, Uren A (2012) Small molecule inhibitors of ezrin inhibit the invasive phenotype of osteosarcoma cells. Oncogene 31:269–281
Pore D, Bodo J, Danda A, Yan D, Phillips JG, Lindner D, Hill BT, Smith MR, Hsi ED, Gupta N (2015) Identification of Ezrin-Radixin-Moesin proteins as novel regulators of pathogenic B-cell receptor signaling and tumor growth in diffuse large B-cell lymphoma. Leukemia 29:1857–1867
Lipreri da Silva JC, Coelho-Silva JL, Lima K, Vicari HP, Lazarini M, Costa-Lotufo LV, Traina F, Machado-Neto JA (2021) Comprehensive analysis of cytoskeleton regulatory genes identifies ezrin as a prognostic marker and molecular target in acute myeloid leukemia. Cell Oncol (Dordr) 44:1105–1117
Celik H, Bulut G, Han J, Graham GT, Minas TZ, Conn EJ, Hong SH, Pauly GT, Hayran M, Li X, Ozdemirli M, Ayhan A, Rudek MA, Toretsky JA, Uren A (2016) Ezrin Inhibition Up-regulates Stress Response Gene Expression. J Biol Chem 291:13257–13270
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, Sander C, Schultz N (2012) The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2:401–404
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E, Sander C, Schultz N (2013) Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 6:pl1.
Thiele C, Hirschfeld G (2020) cutpointr: Improved Estimation and Validation of Optimal Cutpoints in R. arXiv:2002.09209
Heagerty PJ, Zheng Y (2005) Survival model predictive accuracy and ROC curves. Biometrics 61:92–105
Subramanian A, Tamayo P, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A, Pomeroy SL, Golub TR, Lander ES, Mesirov JP (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci U S A 102:15545–15550
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Hua H, Kong Q, Zhang H, Wang J, Luo T, Jiang Y (2019) Targeting mTOR for cancer therapy. J Hematol Oncol 12:71
Tang Y, Sun X, Yu S, Bie X, Wang J, Ren L (2019) Inhibition of Ezrin suppresses cell migration and invasion in human nasopharyngeal carcinoma. Oncol Lett 18:553–560
Wan X, Mendoza A, Khanna C, Helman LJ (2005) Rapamycin inhibits ezrin-mediated metastatic behavior in a murine model of osteosarcoma. Cancer Res 65:2406–2411
Gao J, Long B, Wang Z (2017) Role of Notch signaling pathway in pancreatic cancer. Am J Cancer Res 7:173–186
Ranganathan P, Weaver KL, Capobianco AJ (2011) Notch signalling in solid tumours: a little bit of everything but not all the time. Nat Rev Cancer 11:338–351
Xue J, Yao Y, Yao Q, Tian X, Feng Y, Su H, Kong D, Cui C, Yan L, Hao C, Zhou T (2020) Important roles of estrogen receptor alpha in tumor progression and anti-estrogen therapy of pancreatic ductal adenocarcinoma. Life Sci 260:118302
Saygideger-Kont Y, Minas TZ, Jones H, Hour S, Celik H, Temel I, Han J, Atabey N, Erkizan HV, Toretsky JA, Uren A (2016) Ezrin Enhances EGFR Signaling and Modulates Erlotinib Sensitivity in Non-Small Cell Lung Cancer Cells. Neoplasia 18:111–120
Giacinti C, Giordano A (2006) RB and cell cycle progression. Oncogene 25:5220–5227
Kreis NN, Louwen F, Yuan J (2019) The Multifaceted p21 (Cip1/Waf1/CDKN1A) in Cell Differentiation, Migration and Cancer Therapy. Cancers (Basel) 11
Abukhdeir AM, Park BH (2008) P21 and p27: roles in carcinogenesis and drug resistance. Expert Rev Mol Med 10:e19
Zhan Q, Antinore MJ, Wang XW, Carrier F, Smith ML, Harris CC, Fornace AJ Jr (1999) Association with Cdc2 and inhibition of Cdc2/Cyclin B1 kinase activity by the p53-regulated protein Gadd45. Oncogene 18:2892–2900
Melino G, Bernassola F, Ranalli M, Yee K, Zong WX, Corazzari M, Knight RA, Green DR, Thompson C, Vousden KH (2004) p73 Induces apoptosis via PUMA transactivation and Bax mitochondrial translocation. J Biol Chem 279:8076–8083
Youle RJ, Strasser A (2008) The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 9:47–59
Villunger A, Michalak EM, Coultas L, Mullauer F, Bock G, Ausserlechner MJ, Adams JM, Strasser A (2003) p53- and drug-induced apoptotic responses mediated by BH3-only proteins puma and noxa. Science 302:1036–1038
Acknowledgements
The authors acknowledge all the research participants contributing to The Cancer Genome Atlas (TCGA) resource for providing high-quality data for analysis.
Funding
This study was supported by grant #2019/23864–7, #2019/25421–5, #2020/12909–7, and #2020/12748–3 from the São Paulo Research Foundation (FAPESP). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Contributions
Execution of experiments, data analysis and interpretation, and manuscript writing and editing [J.C.L.S., M.F.L.C., L.B.L.M., B.O.A., K.L.]. Conceptualization, formal analysis, supervision, manuscript writing and editing [J.A.M.-N]. All authors reviewed and approved the submitted manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflicts of interest
All authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lipreri da Silva, J.C., Carvalho, M.F.L., de Miranda, L.B.L. et al. NSC305787, a pharmacological ezrin inhibitor, exhibits antineoplastic activity in pancreatic cancer cells. Invest New Drugs 40, 728–737 (2022). https://doi.org/10.1007/s10637-022-01249-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-022-01249-z