Summary
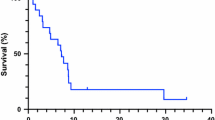
Background Anti-vascular endothelial growth factor receptor (VEGFR) tyrosine kinase inhibitors (TKI) combined with mTOR inhibitors, like everolimus, result in significant responses and prolonged progression-free survival (PFS) among patients with renal cell carcinoma (RCC) [1]. However, everolimus doses >5 mg are often not tolerated when combined with other TKIs2,3. Vorolanib (X-82), an oral anti-VEGFR/platelet derived growth factor receptor (PDGFR)/colony stimulating factor 1 receptor (CSF1R) multitarget TKI, has a short half-life and limited tissue accumulation. We conducted a Phase 1 study of vorolanib with everolimus (10 mg daily) in patients with solid tumors. Methods A 3 + 3 dose escalation design was utilized to determine dose limiting toxicities (DLT) and recommended Phase 2 dose (RP2D) of vorolanib/everolimus. Oral vorolanib at 100, 150, 200, 300, or 400 mg was combined with 10 mg oral everolimus daily. The phase 2 portion was terminated after enrolling two patients due to funding. Results Eighteen patients were evaluable for DLT among 22 treated subjects. Observed DLTs were grade 3 fatigue, hypophosphatemia, and mucositis. The RP2D is vorolanib 300 mg with everolimus 10 mg daily. In 15 patients evaluable for response, three had partial response (PR; 2 RCC, 1 neuroendocrine tumor [NET]) and eight had stable disease (SD; 2 RCC, 6 NET). Conclusions Vorolanib can safely be combined with everolimus. Encouraging activity is seen in RCC and NET. Further studies are warranted. Trial Registration Number: NCT01784861.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study may be available from the corresponding author on reasonable request.
References
Motzer RJ, Hutson TE, Glen H, Michaelson MD, Molina A, Eisen T, Jassem J, Zolnierek J, Maroto JP, Mellado B, Melichar B, Tomasek J, Kremer A, Kim HJ, Wood K, Dutcus C, Larkin J (2015) Lenvatinib, everolimus, and the combination in patients with metastatic renal cell carcinoma: a randomised, phase 2, open-label, multicentre trial. Lancet Oncol 16(15):1473–1482. https://doi.org/10.1016/S1470-2045(15)00290-9
Cohen MM Jr (2009) Judah Folkman, MD, 1933-2008: father of angiogenesis. J Craniofac Surg 20(Suppl 1):590–591. https://doi.org/10.1097/SCS.0b013e3181927e8a
Inoue M, Hager JH, Ferrara N, Gerber HP, Hanahan D (2002) VEGF-A has a critical, nonredundant role in angiogenic switching and pancreatic beta cell carcinogenesis. Cancer Cell 1(2):193–202
Hansel DE, Rahman A, Hermans J, de Krijger RR, Ashfaq R, Yeo CJ, Cameron JL, Maitra A (2003) Liver metastases arising from well-differentiated pancreatic endocrine neoplasms demonstrate increased VEGF-C expression. Mod Pathol 16(7):652–659. https://doi.org/10.1097/01.MP.0000077416.68489.50
Casanovas O, Hicklin DJ, Bergers G, Hanahan D (2005) Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumors. Cancer Cell 8(4):299–309. https://doi.org/10.1016/j.ccr.2005.09.005
Yao JC, Phan A, Hoff PM, Chen HX, Charnsangavej C, Yeung SC, Hess K, Ng C, Abbruzzese JL, Ajani JA (2008) Targeting vascular endothelial growth factor in advanced carcinoid tumor: a random assignment phase II study of depot octreotide with bevacizumab and pegylated interferon alpha-2b. J Clin Oncol 26(8):1316–1323. https://doi.org/10.1200/JCO.2007.13.6374
Rini BI, Halabi S, Rosenberg JE, Stadler WM, Vaena DA, Ou SS, Archer L, Atkins JN, Picus J, Czaykowski P, Dutcher J, Small EJ (2008) Bevacizumab plus interferon alfa compared with interferon alfa monotherapy in patients with metastatic renal cell carcinoma: CALGB 90206. J Clin Oncol 26(33):5422–5428. https://doi.org/10.1200/JCO.2008.16.9847
Escudier B, Bellmunt J, Negrier S, Bajetta E, Melichar B, Bracarda S, Ravaud A, Golding S, Jethwa S, Sneller V (2010) Phase III trial of bevacizumab plus interferon alfa-2a in patients with metastatic renal cell carcinoma (AVOREN): final analysis of overall survival. J Clin Oncol 28(13):2144–2150. https://doi.org/10.1200/JCO.2009.26.7849
Vignot S, Faivre S, Aguirre D, Raymond E (2005) mTOR-targeted therapy of cancer with rapamycin derivatives. Ann Oncol 16(4):525–537. https://doi.org/10.1093/annonc/mdi113
Saxton RA, Sabatini DM (2017) mTOR signaling in growth, metabolism, and disease. Cell 168(6):960–976. https://doi.org/10.1016/j.cell.2017.02.004
Motzer RJ, Escudier B, Oudard S, Hutson TE, Porta C, Bracarda S, Grunwald V, Thompson JA, Figlin RA, Hollaender N, Urbanowitz G, Berg WJ, Kay A, Lebwohl D, Ravaud A, Group R-S (2008) Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet 372(9637):449–456. https://doi.org/10.1016/S0140-6736(08)61039-9
Yao JC, Shah MH, Ito T, Bohas CL, Wolin EM, Van Cutsem E, Hobday TJ, Okusaka T, Capdevila J, de Vries EG, Tomassetti P, Pavel ME, Hoosen S, Haas T, Lincy J, Lebwohl D, Oberg K, Rad001 in Advanced Neuroendocrine Tumors TTSG (2011) Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med 364 (6):514–523. doi:https://doi.org/10.1056/NEJMoa1009290
Yao JC, Pavel M, Lombard-Bohas C, Van Cutsem E, Voi M, Brandt U, He W, Chen D, Capdevila J, de Vries EGE, Tomassetti P, Hobday T, Pommier R, Oberg K (2016) Everolimus for the treatment of advanced pancreatic neuroendocrine tumors: overall survival and circulating biomarkers from the randomized, phase III RADIANT-3 study. J Clin Oncol 34(32):3906–3913. https://doi.org/10.1200/JCO.2016.68.0702
Bendell JC, Patel MR, Moore KN, Chua CC, Arkenau HT, Dukart G, Harrow K, Liang C (2019) Phase I, first-in-human, dose-escalation study to evaluate the safety, tolerability, and pharmacokinetics of Vorolanib in patients with advanced solid tumors. Oncologist 24(4):455–e121. https://doi.org/10.1634/theoncologist.2018-0740
Molina AM, Feldman DR, Voss MH, Ginsberg MS, Baum MS, Brocks DR, Fischer PM, Trinos MJ, Patil S, Motzer RJ (2012) Phase 1 trial of everolimus plus sunitinib in patients with metastatic renal cell carcinoma. Cancer 118(7):1868–1876. https://doi.org/10.1002/cncr.26429
Bitting RL, Healy P, Creel PA, Turnbull J, Morris K, Wood SY, Hurwitz HI, Starr MD, Nixon AB, Armstrong AJ, George DJ (2014) A phase Ib study of combined VEGFR and mTOR inhibition with vatalanib and everolimus in patients with advanced renal cell carcinoma. Clin Genitourin Cancer 12(4):241–250. https://doi.org/10.1016/j.clgc.2013.11.020
Hobday TJ, Qin R, Reidy-Lagunes D, Moore MJ, Strosberg J, Kaubisch A, Shah M, Kindler HL, Lenz HJ, Chen H, Erlichman C (2015) Multicenter phase II trial of Temsirolimus and Bevacizumab in pancreatic neuroendocrine tumors. J Clin Oncol 33(14):1551–1556. https://doi.org/10.1200/JCO.2014.56.2082
Yao JC, Phan AT, Hess K, Fogelman D, Jacobs C, Dagohoy C, Leary C, Xie K, Ng CS (2015) Perfusion computed tomography as functional biomarker in randomized run-in study of bevacizumab and everolimus in well-differentiated neuroendocrine tumors. Pancreas 44(2):190–197. https://doi.org/10.1097/MPA.0000000000000255
Sheng X, Yan X, Chi Z, Cui C, Si L, Tang B, Li S, Mao L, Lian B, Wang X, Bai X, Zhou L, Kong Y, Dai J, Ding L, Mao L, Guo J (2020) Phase 1 trial of vorolanib (CM082) in combination with everolimus in patients with advanced clear-cell renal cell carcinoma. EBioMedicine 55:102755. https://doi.org/10.1016/j.ebiom.2020.102755
Acknowledgments
We wish to acknowledge Abhi Acharya for providing data support during manuscript preparation.
Funding
Xcovery.
Author information
Authors and Affiliations
Contributions
A.W.G., B.T., and C.L. contributed to the study conception and design. Material preparation, data collection and analysis were performed by all authors except C.L. The first draft of the manuscript was written by K.P. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the Institutional Review Boards of Washington University in St. Louis and Vanderbilt University.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Patients signed informed consent regarding publishing their data and imaging.
Conflicts of interest
C.L.: Xcovery employee. No other relevant conflicts of interest to report by other authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pedersen, K.S., Grierson, P.M., Picus, J. et al. Vorolanib (X-82), an oral anti-VEGFR/PDGFR/CSF1R tyrosine kinase inhibitor, with everolimus in solid tumors: results of a phase I study. Invest New Drugs 39, 1298–1305 (2021). https://doi.org/10.1007/s10637-021-01093-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-021-01093-7