Abstract
Background/Aim
Lenvatinib and sorafenib are currently available to treat patients with advanced hepatocellular carcinoma (HCC). However, since the clinical trials evaluating the efficacy of lenvatinib and sorafenib included only patients with Child–Pugh class A, little is known about the effectiveness of the treatments in patients with hepatic decompensation. We compared the effectiveness of lenvatinib and sorafenib in decompensated patients with unresectable HCC.
Methods
Consecutive patients who were classified as Child–Pugh class B or C and received lenvatinib or sorafenib as first-line systemic therapy for unresectable HCC between November 2018 and April 2020 at a tertiary referral center were included in this retrospective study. The primary outcome was overall survival (OS), and the secondary outcomes were progression-free survival (PFS), time-to-progression, best overall tumor response, and safety profiles.
Results
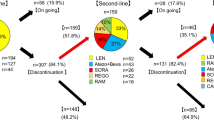
Among 94 patients, 34 received lenvatinib and 60 received sorafenib. The median OS was 4.1 months (95% confidence interval [CI], 2.9–5.2): 4.2 months (95% CI, 2.9–5.3) for lenvatinib and 4.1 months (95% CI, 2.7–6.4) for sorafenib. The treatment regimen was not associated with significant improvement in OS after adjusting for covariables (adjusted hazard ratio [aHR], 0.92; 95% CI, 0.54–1.54; P = 0.74). The treatment regimen was not an independent predictor of PFS (lenvatinib vs. sorafenib; aHR, 0.77; 95% CI, 0.48–1.24; P = 0.28). HRs were maintained even after balancing with the inverse probability treatment weighting method. Objective response rates were 11.8% and 6.7% in patients receiving lenvatinib and sorafenib, respectively (P = 0.45). Ten patients in both groups (five in the lenvatinib group and five in the sorafenib group) underwent dose modification due to adverse events, and significant difference was not observed between the treatment groups (P = 0.49).
Conclusion
The effectiveness of lenvatinib and sorafenib was comparable for the treatment of unresectable HCC in decompensated patients.



Similar content being viewed by others
Abbreviations
- aHR:
-
Adjuvant hazard ratio
- BCLC:
-
Barcelona clinic liver cancer
- BOR:
-
Best overall response
- CI:
-
Confidence interval
- HCC:
-
Hepatocellular carcinoma
- HR:
-
Hazard ratio
- IPTW:
-
Inverse probability treatment weighting
- IQR:
-
Interquartile range
- mRECIST:
-
Modified response evaluation criteria in solid tumors
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- TTP:
-
Time-to-progression
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019;380:1450–1462.
Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390.
Abou-Alfa GK, Niedzwieski D, Knox JJ, et al. Phase III randomized study of sorafenib plus doxorubicin versus sorafenib in patients with advanced hepatocellular carcinoma (HCC): CALGB 80802 (Alliance). J Clin Oncol. 2016;34:192–192.
Cainap C, Qin S, Huang WT, et al. Linifanib versus sorafenib in patients with advanced hepatocellular carcinoma: results of a randomized phase III trial. J Clin Oncol. 2015;33:172–179.
Zhu AX, Rosmorduc O, Evans TR, et al. SEARCH: a phase III, randomized, double-blind, placebo-controlled trial of sorafenib plus erlotinib in patients with advanced hepatocellular carcinoma. J Clin Oncol. 2015;33:559–566.
Johnson PJ, Qin S, Park JW, et al. Brivanib versus sorafenib as first-line therapy in patients with unresectable, advanced hepatocellular carcinoma: results from the randomized phase III BRISK-FL study. J Clin Oncol. 2013;31:3517–3524.
Cheng AL, Kang YK, Lin DY, et al. Sunitinib versus sorafenib in advanced hepatocellular cancer: results of a randomized phase III trial. J Clin Oncol. 2013;31:4067–4075.
Kudo M, Finn RS, Qin S, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163–1173.
Pressiani T, Boni C, Rimassa L, et al. Sorafenib in patients with Child–Pugh class A and B advanced hepatocellular carcinoma: a prospective feasibility analysis. Ann Oncol. 2013;24:406–411.
Marrero JA, Kudo M, Venook AP, et al. Observational registry of sorafenib use in clinical practice across Child–Pugh subgroups: the GIDEON study. J Hepatol. 2016;65:1140–1147.
McNamara MG, Slagter AE, Nuttall C, et al. Sorafenib as first-line therapy in patients with advanced Child–Pugh B hepatocellular carcinoma-a meta-analysis. Eur J Cancer. 2018;105:1–9.
Labeur TA, Achterbergh R, Takkenberg B, Van Delden O, Mathot R, Klumpen HJ. Sorafenib for patients with hepatocellular carcinoma and Child–Pugh B liver cirrhosis: lessons learned from a terminated study. Oncologist. 2019;25:E1274.
Heimbach JK, Kulik LM, Finn RS, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–380.
Kudo M, Matsui O, Izumi N, et al. Transarterial chemoembolization failure/refractoriness: JSH-LCSGJ criteria 2014 update. Oncology. 2014;87:22–31.
Llovet JM, Bru C, Bruix J. Prognosis of hepatocellular carcinoma: the BCLC staging classification. Semin Liver Dis. 1999;19:329–338.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–247.
Common Terminology Criteria for Adverse Events (CTCAE) Version 5. Published: November 27. US Department of Health and Human Services, National Institutes of Health, National Cancer Institute.
Shapiro SS, Wilk MB. An analysis of variance test for normality (complete samples)†. Biometrika. 1965;52:591–611.
Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70:41–55.
Curtis LH, Hammill BG, Eisenstein EL, Kramer JM, Anstrom KJ. Using inverse probability-weighted estimators in comparative effectiveness analyses with observational databases. Med Care. 2007;45:S103-107.
Yamashita T, Kudo M, Ikeda K, et al. REFLECT-a phase 3 trial comparing efficacy and safety of lenvatinib to sorafenib for the treatment of unresectable hepatocellular carcinoma: an analysis of Japanese subset. J Gastroenterol. 2020;55:113–122.
Kuzuya T, Ishigami M, Ito T, et al. Sorafenib versus lenvatinib as first-line therapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis. Anticancer Res. 2020;40:2283–2290.
Cheon J, Chon HJ, Bang Y, et al. Real-world efficacy and safety of lenvatinib in Korean patients with advanced hepatocellular carcinoma: a multicenter retrospective analysis. Liver Cancer. 2020;9:613–624.
Giannini EG, Farinati F, Ciccarese F, et al. Prognosis of untreated hepatocellular carcinoma. Hepatology. 2015;61:184–190.
Raoul JL, Bruix J, Greten TF, et al. Relationship between baseline hepatic status and outcome, and effect of sorafenib on liver function: SHARP trial subanalyses. J Hepatol. 2012;56:1080–1088.
Funding
This study was supported by research grants from the Liver Research Foundation of Korea as part of Bio Future Strategies Research Project.
Author information
Authors and Affiliations
Contributions
The corresponding author (YBL) had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Study concept and design were carried out by YBL. Acquisition, analysis, and interpretation of data were done by all authors. Drafting of the manuscript was done by MKP and YBL. Critical revision of the manuscript for important intellectual content was done by YJK and J-HY. Statistical analysis was carried out by MKP, HM, and YBL. Obtained funding was done by YBL. Administrative, technical, or material support was given by NRC, MAK, HJ, JYN, EJC, J-HL, and SJY. Supervision: J-HY.
Corresponding author
Ethics declarations
Conflict of interest
Dr. Lee YB reports receiving research grant from Samjin Pharmaceuticals and Yuhan Pharmaceuticals; Dr. Lee JH reports receiving research grant from Yuhan Pharmaceuticals and lecture fees from GreenCross Cell, Daewoong Pharmaceuticals, and Gilead Korea; Dr. Yu SJ reports receiving research grants from Yuhan Pharmaceuticals and Daewoong Pharmaceuticals; Dr. Kim YJ reports receiving research grants from AstraZeneca, Boston Scientific, Daewoong Pharmaceuticals, Yuhan Pharmaceuticals, and Samjin Pharmaceuticals, and lecture fees from Bayer HealthCare Pharmaceuticals, Gilead Science, and MSD Korea; Dr. Yoon JH reports receiving research grants from AstraZeneca, Daewoong Pharmaceuticals, and Hanmi Pharmaceuticals. No other potential conflicts of interest relevant to this article are reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Park, M.K., Lee, Y.B., Moon, H. et al. Effectiveness of Lenvatinib Versus Sorafenib for Unresectable Hepatocellular Carcinoma in Patients with Hepatic Decompensation. Dig Dis Sci 67, 4939–4949 (2022). https://doi.org/10.1007/s10620-021-07365-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-021-07365-9