Abstract
Background
Studies on diet and gastric intestinal metaplasia (GIM) risk are lacking in US populations.
Aim
To determine the associations of dietary factors and risk of GIM among a US population with typical American diet.
Methods
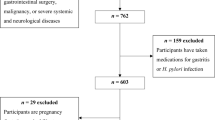
We analyzed data from a cross-sectional study of veterans attending primary care and endoscopy clinics at the Houston VA Medical Center. Patients completed a 110-item Block Food Frequency Questionnaire then underwent upper endoscopy with gastric mapping biopsies. We compared cases defined by GIM on ≥ 1 non-cardia gastric biopsy to controls without GIM. Associations of dietary factors and GIM were estimated using logistic regression models as odds ratios (OR) and 95% confidence intervals (CI).
Results
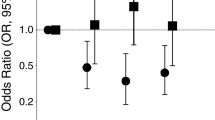
Among 423 GIM cases and 1796 controls, cases were older (62.1 vs. 59.9 years) and more likely to be male (97.2% vs. 90.8%) and non-White (58.6% vs. 39.0%). GIM cases had lower fat intake (percent kcal from fat tertile 1: 43.6% vs. 33.4%) and higher carbohydrate intake (percent kcal from carbohydrate T3: 41.8% vs. 33.3%) than controls. Adjusting for age, gender, race, smoking, and Helicobacter pylori, percent kcal from carbohydrates (T3 vs. T1: OR 1.35, 95% CI 1.08–1.67), fruit intake (T3 vs. T1: OR 1.28, 95% CI 1.02–1.61), and fiber intake (T3 vs. T1: OR 1.37, 95% CI 1.04–1.80) were associated with GIM. In subgroup analyses, these associations were primarily seen in non-White patients.
Conclusions
Few dietary factors, including high carbohydrate intake, are associated with increased risk of GIM in US populations, independent of H. pylori or smoking.
Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Howlader N NA, Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA (eds). SEER Cancer Statistics Review, 1975–2016, National Cancer Institute. Bethesda, MD, https://seer.cancer.gov/csr/1975_2016/, based on November 2018 SEER data submission, posted to the SEER web site, April 2019.
Wang Z, Graham DY, Khan A, et al. Incidence of gastric cancer in the USA during 1999 to 2013: a 50-state analysis. Int J Epidemiol. 2018;47:966–975.
Graham DY. Helicobacter pylori update: gastric cancer, reliable therapy, and possible benefits. Gastroenterology. 2015;148:719-31.e3.
Oh S, Kim N, Yoon H, et al. Risk factors of atrophic gastritis and intestinal metaplasia in first-degree relatives of gastric cancer patients compared with age-sex matched controls. J Cancer Prev. 2013;18:149–160.
Kim N, Park YS, Cho SI, et al. Prevalence and risk factors of atrophic gastritis and intestinal metaplasia in a Korean population without significant gastroduodenal disease. Helicobacter. 2008;13:245–255.
Leung WK, Ng EK, Chan WY, et al. Risk factors associated with the development of intestinal metaplasia in first-degree relatives of gastric cancer patients. Cancer Epidemiol Biomarkers Prev. 2005;14:2982–2986.
Tan MC, Mallepally N, Liu Y, et al. Demographic and lifestyle risk factors for Gastric Intestinal Metaplasia among US Veterans. Am J Gastroenterol 2019; accepted manuscript (in press).
Jedrychowski W, Popiela T, Drews M, et al. Effect of Helicobacter pylori infection, smoking and dietary habits on the occurrence of antrum intestinal metaplasia. Clinico-epidemiological study in Poland. Pol J Pathol Off J Pol Soc Pathol. 1999;50:289–295.
Russo A, Maconi G, Spinelli P, et al. Effect of lifestyle, smoking, and diet on development of intestinal metaplasia in H. pylori-positive subjects. Am J Gastroenterol. 2001;96:1402–1408.
Marcos-Pinto R, Carneiro F, Dinis-Ribeiro M, et al. First-degree relatives of patients with early-onset gastric carcinoma show even at young ages a high prevalence of advanced OLGA/OLGIM stages and dysplasia. Aliment Pharmacol Ther. 2012;35:1451–1459.
Rokkas T, Sechopoulos P, Pistiolas D, et al. Helicobacter pylori infection and gastric histology in first-degree relatives of gastric cancer patients: a meta-analysis. Eur J Gastroenterol Hepatol. 2010;22:1128–1133.
Song JH, Kim YS, Heo NJ, et al. High salt intake is associated with atrophic gastritis with intestinal metaplasia. Cancer Epidemiol Biomark Prev. 2017;26:1133–1138.
Taborda AG, Prolla JC. Alimentary factors in the development of gastric intestinal metaplasia in functional dyspeptic patients. Arquivos de Gastroenterologia. 2012;49:208–213.
Dias-Neto M, Pintalhao M, Ferreira M, et al. Salt intake and risk of gastric intestinal metaplasia: systematic review and meta-analysis. Nutr Cancer. 2010;62:133–147.
Fontham E, Zavala D, Correa P, et al. Diet and chronic atrophic gastritis: a case-control study. J Natl Cancer Inst. 1986;76:621–627.
Garnweidner LM, Terragni L, Pettersen KS, et al. Perceptions of the host country’s food culture among female immigrants from Africa and Asia: aspects relevant for cultural sensitivity in nutrition communication. J Nutr Educ Behav. 2012;44:335–342.
Park SY, Paik HY, Skinner JD, et al. Mothers’ acculturation and eating behaviors of Korean American families in California. J Nutr Educ Behav. 2003;35:142–147.
Yang EJ, Chung HK, Kim WY, et al. Chronic diseases and dietary changes in relation to Korean Americans’ length of residence in the United States. J Am Diet Assoc. 2007;107:942–950.
Thapa S, Fischbach LA, Delongchamp R, et al. Association between dietary salt intake and progression in the gastric precancerous process. Cancers. 2019;11:467.
Nguyen TH, Thrift AP, Ramsey D, et al. Risk factors for Barrett’s esophagus compared between African Americans and non-Hispanic Whites. Am J Gastroenterol. 2014;109:1870–1880.
Subar AF, Thompson FE, Kipnis V, et al. Comparative validation of the Block, Willett, and National Cancer Institute food frequency questionnaires: the Eating at America’s Table Study. Am J Epidemiol. 2001;154:1089–1099.
Dixon MF, Genta RM, Yardley JH, et al. Classification and grading of gastritis. The updated Sydney System. International workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161–1181.
Shiota S, Thrift AP, Green L, et al. Clinical manifestations of Helicobacter pylori-negative gastritis. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc. 2017;15:1037-46.e3.
Jansen MC, Bueno-de-Mesquita HB, Rasanen L, et al. Consumption of plant foods and stomach cancer mortality in the seven countries study. Is grain consumption a risk factor? Seven Countries Study Research Group. Nutr Cancer. 1999;34:49–55.
Jedrychowski W, Popiela T, Steindorf K, et al. Nutrient intake patterns in gastric and colorectal cancers. Int J Occup Med Environ Health. 2001;14:391–395.
Risch HA, Jain M, Choi NW, et al. Dietary factors and the incidence of cancer of the stomach. Am J Epidemiol. 1985;122:947–959.
Fang X, Wei J, He X, et al. Landscape of dietary factors associated with risk of gastric cancer: a systematic review and dose-response meta-analysis of prospective cohort studies. Eur J Cancer. 2015;51:2820–2832.
Larsson SC, Bergkvist L, Wolk A. Glycemic load, glycemic index and carbohydrate intake in relation to risk of stomach cancer: a prospective study. Int J Cancer. 2006;118:3167–3169.
Kodama M, Kodama T, Suzuki H, et al. Effect of rice and salty rice diets on the structure of mouse stomach. Nutr Cancer. 1984;6:135–147.
Stemmermann GN, Nomura AM, Chyou PH, et al. Impact of diet and smoking on risk of developing intestinal metaplasia of the stomach. Dig Dis Sci. 1990;35:433–438.
Brawner KM, Morrow CD, Smith PD. Gastric microbiome and gastric cancer. Cancer J (Sudbury, Mass). 2014;20:211–216.
Eun CS, Kim BK, Han DS, et al. Differences in gastric mucosal microbiota profiling in patients with chronic gastritis, intestinal metaplasia, and gastric cancer using pyrosequencing methods. Helicobacter. 2014;19:407–416.
Stillwell WG, Glogowski J, Xu HX, et al. Urinary excretion of nitrate, N-nitrosoproline, 3-methyladenine, and 7-methylguanine in a Colombian population at high risk for stomach cancer. Cancer Res. 1991;51:190–194.
Tan MC, Graham DY. Proton pump inhibitor therapy after Helicobacter pylori eradication may increase the risk of gastric cancer. BMJ Evid Based Med. 2018;23:111–112.
Miftahussurur M, Yamaoka Y, Graham DY. Helicobacter pylori as an oncogenic pathogen, revisited. Expert Rev Mol Med. 2017;19:e4.
Gancz H, Jones KR, Merrell DS. Sodium chloride affects Helicobacter pylori growth and gene expression. J Bacteriol. 2008;190:4100–4105.
Loh JT, Friedman DB, Piazuelo MB, et al. Analysis of Helicobacter pylori cagA promoter elements required for salt-induced upregulation of CagA expression. Infect Immun. 2012;80:3094–3106.
Kato I, Vivas J, Plummer M, et al. Environmental factors in Helicobacter pylori-related gastric precancerous lesions in Venezuela. Cancer Epidemiol Biomark Prev. 2004;13:468–476.
Jenab M, Thompson LU. Phytic acid in wheat bran affects colon morphology, cell differentiation and apoptosis. Carcinogenesis. 2000;21:1547–1552.
World Cancer Research Fund American Institute for Cancer Research. Diet, nutrition, physical activity and stomach cancer: World Cancer Research Fund International; 2018. Retrieved from https://www.wcrf.org/sites/default/files/Stomach-cancer-report.pdf Available from: https://www.wcrf.org/sites/default/files/Stomach-cancer-report.pdf.
Salganik RI. The benefits and hazards of antioxidants: controlling apoptosis and other protective mechanisms in cancer patients and the human population. J Am Coll Nutr. 2001;20:464S–472S. discussion 73S-75S.
Nouraie M, Pietinen P, Kamangar F, et al. Fruits, vegetables, and antioxidants and risk of gastric cancer among male smokers. Cancer Epidemiol Biomark Prev. 2005;14:2087–2092.
Threapleton DE, Greenwood DC, Evans CE, et al. Dietary fibre intake and risk of cardiovascular disease: systematic review and meta-analysis. BMJ (Clin Res Ed). 2013;347:f6879.
Aune D, Giovannucci E, Boffetta P, et al. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int J Epidemiol. 2017;46:1029–1056.
Ma Y, Hu M, Zhou L, et al. Dietary fiber intake and risks of proximal and distal colon cancers: a meta-analysis. Medicine (Baltimore). 2018;97:e11678.
Da B, Jani N, Gupta N, et al. High-risk symptoms do not predict gastric cancer precursors. Helicobacter. 2019;24:e12548.
Simko V, Anand N, Ginter E. Gastric intestinal metaplasia—age, ethnicity and surveillance for gastric cancer. Bratislavske lekarske listy. 2015;116:3–8.
Almouradi T, Hiatt T, Attar B. Gastric intestinal metaplasia in an underserved population in the USA: prevalence, epidemiologic and clinical features. Gastroenterol Res Pract. 2013;2013:856256.
Capelle LG, Haringsma J, de Vries AC, et al. Narrow band imaging for the detection of gastric intestinal metaplasia and dysplasia during surveillance endoscopy. Dig Dis Sci. 2010;55:3442–3448.
Pimentel-Nunes P, Libanio D, Lage J, et al. A multicenter prospective study of the real-time use of narrow-band imaging in the diagnosis of premalignant gastric conditions and lesions. Endoscopy. 2016;48:723–730.
Sobrino-Cossío S, Abdo Francis JM, Emura F, et al. Efficacy of narrow-band imaging for detecting intestinal metaplasia in adult patients with symptoms of dyspepsia. Revista de Gastroenterología de México (English Edition). 2018;83:245–252.
Fennerty MB, Emerson JC, Sampliner RE, et al. Gastric intestinal metaplasia in ethnic groups in the southwestern United States. Cancer Epidemiol Biomark Prev. 1992;1:293–296.
Tannenbaum SR, Weisman M, Fett D. The effect of nitrate intake on nitrite formation in human saliva. Food Cosmet Toxicol. 1976;14:549–552.
Acknowledgments
This work was supported in part by National Institutes of Health grant P30 DK056338 (Study Design and Clinical Research Core), which supports the Texas Medical Center Digestive Diseases Center. This research was supported in part with resources at the VA HSR&D Center for Innovations in Quality, Effectiveness and Safety (#CIN 13-413), at the Michael E. DeBakey VA Medical Center, Houston, TX. The opinions expressed reflect those of the authors and not necessarily those of the Department of Veterans Affairs, the US government or Baylor College of Medicine. Mimi C. Tan, MD, MPH is guarantor of the article.
Author information
Authors and Affiliations
Contributions
APT, HBE, and MCT analyzed the data, designed the research study, wrote the paper, and contributed to the design of the study. NM collected the data and contributed to the design of the study. QH wrote the paper. YL analyzed the data and contributed to the design of the study. All authors have approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no competing interests for this publication.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tan, M.C., Mallepally, N., Ho, Q. et al. Dietary Factors and Gastric Intestinal Metaplasia Risk Among US Veterans. Dig Dis Sci 66, 1600–1610 (2021). https://doi.org/10.1007/s10620-020-06399-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-020-06399-9