Abstract
Background and Aims
Colitis-associated cancer (CAC) is one of the most serious complications in patients with inflammatory bowel disease. Sphingosine kinase 1 (Sphk1) is a key enzyme in the sphingolipid pathway and has oncogene potential for inducing both initiation and progression of tumors. The aim of this work is to characterize the role of epithelial Sphk1 in mouse colitis and CAC models.
Methods
We investigated the roles of Sphk1 in CAC by conditional deletion of Sphk1 in intestinal epithelial cells (IECs).
Results
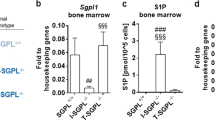
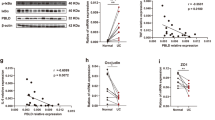
CAC was induced in both Sphk1ΔIEC/ApcMin/+ and Sphk1IEC/ApcMin/+ mice by administration of 2% dextran sodium sulfate (DSS) for 7 days. Genetic deletion of Sphk1 significantly reduced the number and size of tumors in ApcMin/+ mice. Histologic grade was more severe in Sphk1ΔIEC/ApcMin/+ mice compared with Sphk1IEC/ApcMin/+ mice (invasive carcinoma, 71% versus 13%, p < 0.05). Deletion of Sphk1 decreased mucosal proliferation and inhibited STAT3 activation and genetic expression of cyclin D1 and cMyc in tumor cells. Conditional deletion of Sphk1 using CRISPR-Cas9 in HCT 116 cells inhibited interleukin (IL)-6-mediated STAT3 activation.
Conclusions
Epithelial conditional deletion of Sphk1 inhibits CAC in ApcMin/+-DSS models in mice by inhibiting STAT3 activation and its target signaling pathways.






Similar content being viewed by others
References
Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420:860–867.
Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539–545.
Bernstein CN, Blanchard JF, Kliewer E, et al. Cancer risk in patients with inflammatory bowel disease—a population-based study. Cancer. 2001;91:854–862.
Munkholm P. Review article: the incidence and prevalence of colorectal cancer in inflammatory bowel disease. Aliment Pharmacol Ther. 2003;18:1–5.
Ng WK, Wong SH, Ng SC. Changing epidemiological trends of inflammatory bowel disease in Asia. Intest Res. 2016;14:111–119.
Le Stunff H, Milstien S, Spiegel S. Generation and metabolism of bioactive sphingosine-1-phosphate. J Cell Biochem. 2004;92:882–899.
Hait NC, Maiti A. The role of sphingosine-1-phosphate and ceramide-1-phosphate in inflammation and cancer. Mediat Inflamm. 2017;2017:4806541.
Liu SQ, Xu CY, Wu WH, et al. Sphingosine kinase 1 promotes the metastasis of colorectal cancer by inducing the epithelial–mesenchymal transition mediated by the FAK/AKT/MMPs axis. Int J Oncol. 2019;54:41–52.
Li W, Yu CP, Xia JT, et al. Sphingosine kinase 1 is associated with gastric cancer progression and poor survival of patients. Clin Cancer Res. 2009;15:1393–1399.
Liu SQ, Su YJ, Qin MB, et al. Sphingosine kinase 1 promotes tumor progression and confers malignancy phenotypes of colon cancer by regulating the focal adhesion kinase pathway and adhesion molecules. Int J Oncol. 2013;42:617–626.
Dirisina R, Katzman RB, Goretsky T, et al. p53 and PUMA independently regulate apoptosis of intestinal epithelial cells in patients and mice with colitis. Gastroenterology. 2011;141:1036–1045.
Lee SD, Choe JW, Lee BJ, et al. Butein effects in colitis and interleukin-6/signal transducer and activator of transcription 3 expression. World J Gastroenterol. 2015;21:465.
Tanaka T, Kohno H, Suzuki R, et al. Dextran sodium sulfate strongly promotes colorectal carcinogenesis in Apc(Min/+) mice: inflammatory stimuli by dextran sodium sulfate results in development of multiple colonic neoplasms. J Cancer. 2006;118:25–34.
Bollrath J, Phesse TJ, von Burstin VA, et al. gp130-mediated Stat3 activation in enterocytes regulates cell survival and cell-cycle progression during colitis-associated tumorigenesis. Cancer Cell. 2009;15:91–102.
Pulkoski-Gross MJ, Obeid LM. Molecular mechanisms of regulation of sphingosine kinase 1. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863:1413–1422.
Vadas M, Xia P, McCaughan G, et al. The role of sphingosine kinase 1 in cancer: oncogene or non-oncogene addiction? Biochim Biophys Acta. 2008;1781:442–447.
Nemoto S, Nakamura M, Osawa Y, et al. Sphingosine kinase isoforms regulate oxaliplatin sensitivity of human colon cancer cells through ceramide accumulation and Akt activation. J Biol Chem. 2009;284:10422–10432.
Liu SQ, Huang JA, Qin MB, et al. Sphingosine kinase 1 enhances colon cancer cell proliferation and invasion by upregulating the production of MMP-2/9 and uPA via MAPK pathways. Int J Colorectal Dis. 2012;27:1569–1578.
Luo C, Zhang H. The role of proinflammatory pathways in the pathogenesis of colitis-associated colorectal cancer. Mediat Inflamm. 2017;2017:5126048.
Grivennikov SI. Inflammation and colorectal cancer: colitis-associated neoplasia. Semin Immunopathol. 2013;35:229–244.
Shafik NM, Gaber RA, Mohamed DA, et al. Hesperidin modulates dextran sulfate sodium-induced ulcerative colitis in rats: targeting sphingosine kinase-1- sphingosine 1 phosphate signaling pathway, mitochondrial biogenesis, inflammation, and apoptosis. J Biochem Mol Toxicol. 2019;33:e22312.
Xi M, Ge J, Wang X, et al. Development of hydroxy-based sphingosine kinase inhibitors and anti-inflammation in dextran sodium sulfate induced colitis in mice. Bioorg Med Chem. 2016;24:3218–3230.
Pulkoski-Gross MJ, Uys JD, Orr-Gandy KA, et al. Novel sphingosine kinase-1 inhibitor, LCL351, reduces immune responses in murine DSS-induced colitis. Prostaglandins Other Lipid Mediat. 2017;130:47–56.
Snider AJ, Kawamori T, Bradshaw SG, et al. A role for sphingosine kinase 1 in dextran sulfate sodium-induced colitis. FASEB J. 2009;23:143–152.
Yuza K, Nagahashi M, Shimada Y, et al. Upregulation of phosphorylated sphingosine kinase 1 expression in colitis-associated cancer. J Surg Res. 2018;231:323–330.
Liang J, Nagahashi M, Kim EY, et al. Sphingosine-1-phosphate links persistent STAT3 activation, chronic intestinal inflammation, and development of colitis-associated cancer. Cancer Cell. 2013;23:107–120.
Corvinus FM, Orth C, Moriggl R, et al. Persistent STAT3 activation in colon cancer is associated with enhanced cell proliferation and tumor growth. Neoplasia. 2005;7:545–555.
Musteanu M, Blaas L, Mair M, et al. Stat3 is a negative regulator of intestinal tumor progression in Apc(Min) mice. Gastroenterology. 2010;138:1003-U257.
Han J, Theiss AL. Stat3: friend or foe in colitis and colitis-associated cancer? Inflamm Bowel Dis. 2014;20:2405–2411.
Sugimoto K. Role of STAT3 in inflammatory bowel disease. World J Gastroenterol. 2008;14:5110–5114.
Li W, Lee MR, Kim T, et al. Activated STAT3 may participate in tumor progression through increasing CD133/survivin expression in early stage of colon cancer. Biochem Biophys Res Commun. 2018;497:354–361.
Mitsuyama K, Matsumoto S, Masuda J, et al. Therapeutic strategies for targeting the IL-6/STAT3 cytokine signaling pathway in inflammatory bowel disease. Anticancer Res. 2007;27:3749–3756.
Kusaba T, Nakayama T, Yamazumi K, et al. Activation of STAT3 is a marker of poor prognosis in human colorectal cancer. Oncol Rep. 2006;15:1445–1451.
Wang Y, Shen YC, Wang SN, et al. The role of STAT3 in leading the crosstalk between human cancers and the immune system. Cancer Lett. 2018;415:117–128.
Grivennikov S, Karin E, Terzic J, et al. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell. 2009;15:103–113.
Fukuda T, Naganuma M, Kanai T. Current new challenges in the management of ulcerative colitis. Intest Res. 2019;17:36–44.
Lee SH, Kwon JE, Cho ML. Immunological pathogenesis of inflammatory bowel disease. Intest Res. 2018;16:26–42.
Acknowledgments
This work is supported by grants from the National Research Foundation of Korea (no. NRF-2011-0014256) and by experimental techniques from the Core Laboratory for Convergent Translational Research of Korea University College of Medicine (grant no. K1421417).
Author information
Authors and Affiliations
Contributions
CBI and PSB contributed equally to this work. Conceptualization: LBJ. Formal analysis: KNJ, PSB, CBI, JYA, LHJ. Investigation: KNJ, PSB, CBI, JYA. Project administration: NYS. Supervision: LBJ. Visualization: CBI, JYA, KNJ. Writing—original draft: PSB, CBI, LBJ. Writing—review: PJJ, JMK, KJS, KHS. Writing—editing: LBJ, CBI.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Park, S.B., Choi, Bi., Lee, B.J. et al. Intestinal Epithelial Deletion of Sphk1 Prevents Colitis-Associated Cancer Development by Inhibition of Epithelial STAT3 Activation. Dig Dis Sci 65, 2284–2293 (2020). https://doi.org/10.1007/s10620-019-05971-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-05971-2