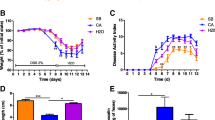
Abstract
Background
The aim of this study was to investigate the effect of the macrolide antibiotic azithromycin on mucosal changes and colonic bacterial load in a murine model of colitis.
Methods
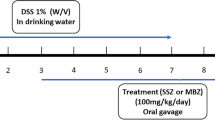
Colitis was induced in CD1 mice using enema of 0.2% solution of dinitrofluorobenzene, combined with skin sensitization. Four experimental groups of animals (N = 10 per group) were treated with 50 mg/kg/day azithromycin (AZ) or metronidazole (MN) perorally, starting 24 h before (AZ−1, MN−1) or 6 h after (AZ+1, MN+1) induction of colitis and for consecutive 5 days. Additional experimental mice group was treated with 10 mg/kg/day methylprednisolone intraperitoneally after induction of experimental colitis in the same manner (MP). Two control groups consisted of healthy animals (C) that received the challenge enema with phosphate-buffered saline (PBS) and animals with experimental colitis (chall) treated with equivolume of PBS perorally. Clinical score (0–5) and histopathologic score (0–30) were used to assess inflammatory changes, and colon washings were used to determine changes in bacterial load.
Results
The anti-inflammatory effect of azithromycin did not differ from the effect of methylprednisolone, when compared with control group with experimental colitis. Metronidazole did not show a significant anti-inflammatory effect. Number of colonic bacteria did not differ significantly between control and experimental groups of animals.
Conclusions
We documented the anti-inflammatory effect of azithromycin in a murine model of acute colitis, suggesting that effects were targeted to oxidative burst and on mucosal/bacterial interface, independent of luminal bacterial load. Further studies should be focused on effect of azithromycin on the role of bacterial biofilm in perpetuation of chronic intestinal inflammation.




Similar content being viewed by others
References
Sands BE. Therapy of inflammatory bowel disease. Gastroenterology. 2000;118:68–82.
Camma C, Giunta M, Rosselli M, Cottone M. Mesalamine in the maintenance treatment of Crohn′s disease: a meta-analysis adjusted for confounding variables. Gastroenterology. 1997;113:1465–1473.
Steinhart AH, Feagan BG, Wong CJ, et al. Combined budesonide and antibiotic therapy for active Crohn’s disease: a randomized controlled trial. Gastroenterology. 2002;123(1):33–40.
Greenbloom SL, Steinhart AH, Greenberg GR. Combination ciprofloxacin and metronidazole for active Crohn’s disease. Can J Gastroenterol. 1998;12(1):53–56.
Labro MT. Anti-inflammatory activity of macrolides: a new therapeutic potential? J Antimicrob Chemother. 1998;41(Suppl 13):37–46.
Shafran I, Kugler L, El-Zaatari FA, Naser SA, Sandoval J. Open clinical trial of rifabutin and clarithromycin therapy in Crohn’s disease. Dig Liver Dis. 2002;34(1):22–28.
Allen SD, Emery CL, Siders JA. Clostridium. U:Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH, eds. Manual of Clinical Microbiology Washington DC: ASM; 1999, pp 654–671.
Brkić T, Banić M, Anić B, et al. A model of inflammatory bowel disease induced by 2, 4-dinitrofluorobenzene in previously sensitized BALB-c mice. Scand J Gastroenterol. 1992;27:184–188.
Banić M, Brkić T, Anić B, et al. Effect of methylprednisolone on small bowell, spleen and liver changes in a murine model of inflammatory bowel disease. Aliment Pharmacol Ther. 1993;7:201–206.
Koneman EW, Allen SD, Janda WM, Schreckenberger PC, Winn WC, eds. The Anaerobic Bacteria Color atlas and textbook of Diagnostic Microbiology. Philadelphia: Lippincott; 1997:709–784.
Hunter MM, Wang A, Hirota CL, McKay DM. Neutralizing Anti-IL-10 antibody blocks the protective effect of tapeworm infection in a murine model of chemically induced colitis. J Immunol. 2005;174:7368–7375.
Anić B, Brkić T, Banić M, et al. Histopathologic features of T-cell mediated colonic injury induced with 2, 4-dinitrofluorobenzene in BALB/c mice. Acta Med Croat. 1997;51:11–14.
Banic M, Anic B, Brkic T, et al. Effect of cyclosporine in a murine model of experimental colitis. Dig Dis Sci. 2002;47(6):1362–1368.
Koneman EW, Allen SD, Janda WM, Schreckenberger PC, Winn WC, eds. Introduction to microbiology: Part I: The role of the Microbiology Laboratory in the Diagnosis of Infectious Diseases:Guidelines to Practice and Management. Color atlas and textbook of Diagnostic Microbiology. Philadelphia: Lippincott; 1997:69–120.
Jousimies-Somer HR, Summanen PH, Finegold SM. Bacteroides, Porphyromonas, Prevotella, Fusobacterium, and Other Anaerobic Gram-Negative Rods and Cocci. In: Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH, eds. Manual of Clinical Microbiology. Washington DC: ASM Press; 1999:690–711.
Hillier SL, Moncla BJ. Peptostreptococcus, Propionibacterium, Lactobacillus, Actinomyces and Other Non-Sporeforming Anaerobic Gram-Positive Bacteria. In: Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH, eds. Manual of Clinical Microbiology. Washington DC: ASM Press; 1999:672–687.
Dawson-Saunders B, Trapp RG. Basic and clinical biostatistics. London, New Jersey: Prentice-Hall; 1994.
Strober W, Fuss IJ, Blumberg RS. The immunology of mucosal models of inflammation. Annu Rev Immunol. 2002;20:495–549.
Elson CO, Sartor RB, Tennyson GS, Riddell RH. Experimental models of inflammatory bowel disease. Gastroenterology. 1995;109:1344–1367.
Xiao WB, Liu YL. Changes of CD8+CD28- T regulatory cells in rat model of colitis induced by 2, 4-dinitrofluorobenzene. World J Gastroenterol. 2003;9(11):2528–2532.
Barnich Nicolas, Darfeuille-Michaud Arlette. Role of bacteria in the etiopathogenesis of inflammatory bowel disease. World J Gastroenterol. 2007;13(42):5571–5576.
Sartor RB. The influence of normal microbial flora on the development of chronic mucosal inflammation. Res Immunol. 1997;148:567–576.
Blondeau JM, DeCarolis E, Metzler KL, Hansen GT. The macrolides. Expert Opin Investig Drugs. 2002;11(2):189–215.
Labro MT. Interference of antibacterial agents with phagocyte functions: immunomodulation or “immuno-fairy tales”? Clin Microbiol Rev. 2000;13(4):615–650.
Parnham MJ, Čulic O, Eraković V, et al. Modulation of neutrophil and inflammation markers in chronic obstructive pulmonary disease by short-term azithromycin treatment. Eur J Pharmacol. 2005;517:132–143.
Labro MT. Interactions entre les agents anti-infectieux et les phagocytes. Presse Med. 1995;24:992–998.
Rennick DM, Fort MM. Lessons from genetically engineered animal models. XII. IL-10-deficient (IL-10(-/-) mice and intestinal inflammation. Am J Physiol Gastrointest Liver Physiol. 2000;278(6):G829–G833.
Arabi Y, Dimock F, Burdon DW, Alexander-Wiliams J, Keighley MR. Influence of neomycin and metronidazole on colonic microflora of volunteers. J Antimicrob Chemother. 1979;5:531–537.
Sartor RB. The influence of normal microbial flora on the development of chronic mucosal inflammation. Res Immunol. 1997;148:567–576.
Mahgoub A, El-Medany A, Mustafa A, Arafah M, Moursi M. Azithromycin and erythromycin ameliorate the extent of colonic d+amage induced by acetic acid in rats. Toxicol Appl Pharmacol. 2005;205(1):43–52.
Menozzi A, Pozzoli C, Poli E, et al. Effect of the macrolide antibacterial drug, tylosin, on TNBS-induced colitis in the rat. Pharmacology. 2005;74(3):135–142.
Yamada T, Deitch E, Specian RD, Perry MA, Sartor RB, Grisham MB. Mechanisms of acute and chronic intestinal inflammation induced by indomethacin. Inflammation. 1993;17:641–662.
Matute AJ, Schurink CA, Krijnen RM, Florijn A, Rozenberg-Arska M, Hoepelman IM. Double-blind, placebo-controlled study comparing the effect of azithromycin with clarithromycin on oropharyngeal and bowel microflora in volunteers. Eur J Clin Microbiol Infect Dis. 2002;21(6):427–431.
Canbakan B, Sentürk H, Tahan V, Yildirim B, Özbay G, Hatemi I, Oktay E: Neutrophil oxidative burst: A possible pathogenic mechanism for tissue damage in ulcerative colitis. Abstract book. Inflammatory bowel disease: Translation from basic research to clinical practice, Dubrovnik: Falk Symposium 140; 2004, A43.
Rainis T, Maor I, Lanir A, Lavy A. Oxidative stress and neutrophil superoxide release in patients with Crohn’s disease: Distinction between active and non-active disease. Abstract book. Inflammatory bowel disease: Translation from basic research to clinical practice, Dubrovnik: Falk Symposium 140; 2004, A44.
Popovic M, Janicijevic-Hudomal S, Kaurinovic B, Rasic J, Trivic S. Antioxidant effects of some drugs on ethanol-induced ulcers. Molecules. 2009;14:816–826.
Swidsinski A, Weber J, Loening-Baucke V, Hale LP, Lochs H. Spatial organization and composition of the mucosal flora in patients with inflammatory bowel disease. J Clin Microbiol. 2005;43(7):3380–3389.
Barnich N, Darfeuille-Michaud A. Role of bacteria in the etiopathogenesis of inflammatory bowel disease. World J Gastroenterol. 2007;13(42):5571–5576.
Giamarellos-Bourboulis EJ. Macrolides beyond the conventional antimicrobials: a class of potent immunomodulators. Int J Antimicrob Agents. 2008;31(1):12–20.
Macfarlane S, Dillon JF. Microbial biofilms in the human gastrointestinal tract. J Appl Microbiol. 2007;102(5):1187–1196.
Giamarellos-Bourboulis EJ. Macrolides beyond the conventional antimicrobials: a class of potent immunomodulators. Int J Antimicrob Agents. 2008;31(1):12–20.
Author information
Authors and Affiliations
Corresponding author
Additional information
Statement
The manuscript “Effect of Azithromycin on Acute Inflammatory Lesions and Colonic Bacterial Load in A Murine model of Experimental Colitis” has been approved by all authors. None of the material in this manuscript has been published previously in any form and none of this material is currently under consideration for publication elsewhere.
Rights and permissions
About this article
Cite this article
Pleško, S., Banić, M., Plečko, V. et al. Effect of Azithromycin on Acute Inflammatory Lesions and Colonic Bacterial Load in a Murine Model of Experimental Colitis. Dig Dis Sci 55, 2211–2218 (2010). https://doi.org/10.1007/s10620-009-1034-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-009-1034-7