Abstract
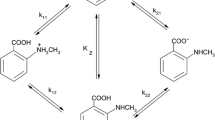
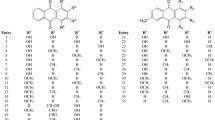
The 9,10–1,10-anthraquinoid tautomer was found to be characteristic of physcion and emodin and their analogs in solutions using spectrophotometric, quantum-chemical, and correlation methods. Ionization of these compounds was accompanied by a shift in tautomeric equilibrium. In alkaline solutions 1,10-anthraquinoid anions with a single α-hydroxy that were stabilized by an intramolecular H-bond were formed. Tautomerism occurred in both the ground and excited states of the molecules.
Similar content being viewed by others
REFERENCES
V. Ya. Fain, B. E. Zaitsev, and M. A. Ryabov, Khim. Prir. Soedin., 119 (2005).
R. H. Thomson, Naturally Occurring Quinones, Vol. II, Academic Press, London and New York (1971); Vol. III, Chapman and Hall, London and New York (1987), p. 350.
R. A. Muzychkina, Natural Anthraquinones. Bioloogical Properties and Physicochemical Characteristics [in Russian], G. A. Tolstikov, ed., Fazis, Moscow (1998).
V. Ya. Fain, Electronic Absorption Spectra and Structure of 9,10-Anthraquinones. I. 9,10-Anthraquinone and Its Monosubstituted Derivatives [in Russian], Sputnik+, Moscow (2003).
V. Ya. Fain, Electronic Absorption Spectra and Structure of 9,10-Anthraquinones. II. Disubstituted 9,10-Anthraquinones [in Russian], Sputnik+, Moscow (2003).
V. Ya. Fain, 9,10-Anthraquinones and Their Use [in Russian], Photochemistry Center of the Russian Academy of Sciences, Moscow (1999).
V. Ya. Fain, Correlation Analysis of Electronic Absorption Spectra [in Russian], Sputnik+, Moscow (2002).
V. Ya. Fain, B. E. Zaitsev, and M. A. Ryabov, Zh. Obshch. Khim., 73, 1688 (2003).
V. Ya. Fain, B. E. Zaitsev, and M. A. Ryabov, Zh. Obshch. Khim., 73, 2035 (2003).
M. J. S. Dewar, Molecular Orbital Method in Organic Chemistry [Russian translation], Mir, Moscow (1972).
K. Nishimoto and L. S. Forster, Theor. Chim. Acta, 4, 155 (1966).
N. K. Utkina and O. B. Maksimov, Khim. Prir. Soedin., 636 (1977).
A. M. Tessier, P. Delaveau, and B. Champion, Planta Med., 41, 337 (1981).
A. Amonkar, C.-J. Chang, and J. M. Cassady, Experientia, 37, 1138 (1981).
P. N. Verma, D. R. Lohar, and A. K. Satsangi, Pharm. Weekbl., 118, 432 (1983).
S. Gopolakrishnan, S. Neelakantan, and P. V. Raman, J. Indian Chem. Soc., 67, 390 (1990).
N. K. Utkina and O. B. Maksimov, Khim. Prir. Soedin., 651 (1978).
J. W. Fairbairn and F. J. El-Muhtadi, Phytochemistry, 11, 263 (1972).
M. Coskun, N. Tanker, A. Sakushima, S. Kitagawa, and S. Nishibe, Phytochemistry, 23, 1485 (1984).
A. F. Kovalev and V. I. Litvinenko, Khim.-Farm. Zh., 3, 22 (1969).
H. J. Banks, D. W. Cameron, and M. J. Crossley, Aust. J. Chem., 29, 2231 (1976).
H. Auterhoff and S. Gedeon, Arch. Pharm. Ber. Dtsch. Pharm. Ges., 304, 241 (1971).
T. Pal and N. Jana, Analyst, 118, 1337 (1993); A. G. Gonzalez, J. D. Martin, and C. Perez, An. Quim. Real. Soc. Esp. Fis. Quim., 69, 805 (1973).
H. W. Meyer and W. D. Treadwell, Helv. Chim. Acta, 35, 1460 (1952).
Author information
Authors and Affiliations
Additional information
__________
Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 411–415, September–October, 2005.
Rights and permissions
About this article
Cite this article
Fain, V.Y., Zaitsev, B.E. & Ryabov, M.A. Tautomerism of the Natural Anthraquinones Physcion and Emodin and Their Analogs. Chem Nat Compd 41, 501–507 (2005). https://doi.org/10.1007/s10600-005-0193-9
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10600-005-0193-9