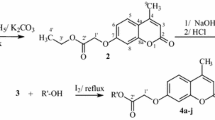
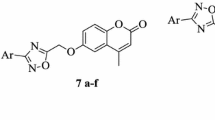
A series of novel coumarin derivatives were synthesized from commercially available chemical agents. All prepared compounds were evaluated for their in vitro antiproliferative activity against five selected human cancer cell lines (EC109, MGC-803, PC-3, MCF-7, and EC9706) and their in vitro antimicrobial activity against E. coli and M. albicans. 4-(3-Bromopropoxy)-2H-chromen-2-one exhibited the highest growth inhibition against MGC-803 cell line (IC50 47.7 μM) and 7-(2-bromoethoxy)-2H-chromen-2-one exhibited the highest growth inhibition against MCF-7 cell line (IC50 48.3 μM). The latter compound was also the most active against E. coli (MIC50 0.27 μg/ml).

Similar content being viewed by others
References
Kahveci, B.; Yılmaz, F.; Menteşe, E.; Ülker, S. Chem. Heterocycl. Compd. 2015, 51, 447. [Khim. Geterotsikl. Soedin. 2015, 51, 447.]
Ghate, M.; Manohar, D.; Kulkarni, V.; Shobha, R.; Kattimani, S. Y. Eur. J. Med. Chem. 2003, 38, 297.
Weigt, S.; Huebler, N.; Strecker, R.; Braunbeck, T.; Broschard, T. H. Reprod. Toxicol. 2012, 33, 133.
Zhou, P.; Takaishi, Y.; Duan, H.; Chen, B.; Honda, G.; Itoh, M.; Takeda, Y.; Kodzhimatov, O. K.; Lee, K. H. Phytochemistry 2000, 53, 689.
Žalubovskis, R. Chem. Heterocycl. Compd. 2015, 51, 607. [Khim. Geterotsikl. Soedin. 2015, 51, 607.]
Störl, K.; Störl, J.; Roth, M.; Zimmer, C. Biochim. Biophys. Acta, Gen. Subj. 1994, 1199, 143.
Roussaki, M.; Kontogiorgis, C. A.; Hadjipavlou-Litina, D.; Hamilakis, S.; Detsi, A. Bioorg. Med. Chem. Lett. 2010, 20, 3889.
Schmutz, E.; Mühlenweg, A.; Li, S.-M.; Heide, L. Antimicrob. Agents Chemother. 2003, 47, 869.
Xie, S.-S.; Wang, X.-B.; Li, J.-Y.; Yang, L.; Kong, L.-Y. Eur. J. Med. Chem. 2013, 64, 540.
Ren, J.-L.; Zhang, X.-Y.; Yu, B.; Wang, X.-X.; Shao, K.-P.; Zhu, X.-G.; Liu, H.-M. Eur. J. Med. Chem. 2015, 93, 321
Ren, J.-L.; Zheng, E.; Ye, X.-W.; Wang, M.-M.; Yu, B.; Wang, W.-H.; Guo, Y.-Z.; Liu, H.-M. Bioorg. Med. Chem. Lett. 2013, 23, 4154.
The authors are grateful for the financial support from the National Natural Science Foundation of China (No. 81273393).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information file to this article containing the 1H and 13C NMR spectra of the synthesized compounds is available at http://link.springer.com/journal/10593.
Published in Khimiya Geterotsiklicheskikh Soedinenii, 2016, 52(6), 374–378
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 719 kb)
Rights and permissions
About this article
Cite this article
Zhang, SY., Fu, DJ., Sun, HH. et al. Synthesis and bioactivity of novel coumarin derivatives. Chem Heterocycl Comp 52, 374–378 (2016). https://doi.org/10.1007/s10593-016-1898-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-016-1898-3