Abstract
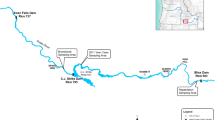
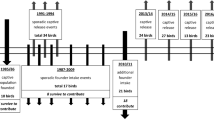
Because of recent environmental changes, a number of species have become critically endangered in the wild. To prevent extinction of these species, captive populations have been established and generally maintained in a manner that attempts to avoid detrimental genetic changes. In particular, wild founders should be unrelated and not inbred. The subsequent generations in captivity should make the effective population size as large as possible to retain the initial genetic variation. Pallid sturgeon (Scaphirhynchus albus) from the upper Missouri River USA have not successfully recruited in decades. Perpetuation of this stock is now accomplished through captive spawning of wild-caught fish with subsequent release of their offspring. In addition to the fish released into the wild, more than 2,000 offspring of the wild fish from 11 year-classes are captively housed. We examined the genetic risks associated with using these fish as a captive population for the future propagation of pallid sturgeon and concluded that the wild individuals are unrelated, non-inbred remnants of a formerly larger population. Further, there is a sufficiently large effective population size (N e) present in the captive broodstock for propagation provided that the effective population size can be increased in subsequent generations. This study shows how genetic and evolutionary principles and applications can be used to evaluate the potential founders of a captive population and make recommendations for the long-term evolutionary success of an endangered species.




Similar content being viewed by others
References
Araki H, Berejikian BA, Ford MJ, Blouin MS (2008) Fitness of hatchery-reared salmonids in the wild. Evol Appl 1:342–355
Ballou JD, Gilpin ME, Foose TJ (1995) Population management for survival and recovery. Columbia University Press, New York
Bergman HL, Boelter AM, Parady K, Fleming C, Keevin T, Latka DC, Korschgen C, Galat DL, Hill T, Jordan G, Krentz S, Nelson-Stastny W, Olsen M, Mestl GE, Rouse K, Berkley J (2008) Research needs and management strategies for pallid sturgeon recovery. Proceedings of a Workshop held July 31–August 2, 2007, St. Louis. Final Report to the U.S. Army Corps of Engineers. William D. Ruckelshaus Institute of Environment and Natural Resources, University of Wyoming, Laramie
Boyd L, Houpt FA (1994) Przewalski’s horse: the history and biology of an endangered species. State Univ. New York Press, Albany
Braaten PJ, Fuller DB, Holte LD, Lott RD, Viste W, Brandt TF, Legare RG (2008) Drift dynamics of larval pallid sturgeon and shovelnose sturgeon in a natural side channel of the upper Missouri River, Montana. N Am J Fish Manage 28:808–826
Braaten PJ, Fuller DB, Lott RD, Jordan GR (2009) An estimate of the historic population size of adult pallid sturgeon in the upper Missouri River Basin, Montana and North Dakota. J Appl Ichthyol 25:2–7
Christie MR, Marine ML, French RA, Blouin MS (2012) Genetic adaptation to captivity can occur in a single generation. Proc Natl Acad Sci USA 109:238–242
Crossman JA, Scribner KT, Duong TY, Davis CA, Forsythe PS, Baker EA (2011) Gamete and larval collection methods and hatchery rearing environments affect levels of genetic diversity in early life stages of lake sturgeon (Acipenser fulvescens). Aquaculture 310:312–324
Crow JF, Morton NE (1955) Measurement of gene frequency drift in small populations. Evolution 9:202–214
DeHaan PW, Jordan GR, Ardren WR (2008) Use of genetic tags to identify captive-bred pallid sturgeon (Scaphirhynchus albus) in the wild: improving abundance estimates for an endangered species. Conserv Genet 9:691–697
Drauch Schreier A, Rodzen J, Ireland SC, May B (2012) Genetic techniques inform conservation aquaculture of the endangered Kootenai River white sturgeon, Acipenser transmontanus. Endanger Spec Res 16:65–75
Dryer MP, Sandvol AJ (1993) Recovery plan for the pallid sturgeon (Scaphirhynchus albus). US Fish and Wildlife Service, Denver
Duchesne P, Bernatchez L (2002) An analytical investigation of the dynamics of inbreeding in multi-generation supportive breeding. Conserv Genet 3:47–60
Ford MJ (2002) Selection in captivity during supportive breeding may reduce fitness in the wild. Conserv Biol 16:815–825
Frankham R (2008) Genetic adaptation to captivity in species conservation programs. Mol Ecol 17:325–333
Franklin IR (1980) Evolutionary change in small populations. In: Soule ME, Wilcox BA (eds) Conservation biology: an evolutionary-ecological perspective. Sinauer Associates, Sunderland, pp 135–150
Fraser DJ (2008) How well can captive breeding programs conserve biodiversity? A review of salmonids. Evol Appl 1:535–586
Gilpin ME, Soule ME (1986) Minimum viable populations: processes of species extinction. In: Soule ME (ed) Conservation biology: the science of scarcity and diversity. Sinauer Associates, Sunderland, pp 19–34
Goodnight KF, Queller DC (1999) Computer software for performing likelihood tests of pedigree relationship using genetic markers. Mol Ecol 8:1231–1234
Hall N, Mercer L, Phillips D, Shaw J, Anderson AD Maximum likelihood estimation of individual inbreeding coefficients and null allele frequencies. Genet Res (in press)
Hedrick P (2011) Genetics of populations, 4th edn. Jones and Bartlett, Boston
Hedrick PW, Fredrickson RJ (2008) Captive breeding and the reintroduction of Mexican and red wolves. Mol Ecol 17:344–350
Hedrick PW, Fredrickson R (2010) Genetic rescue guidelines with examples from Mexican wolves and Florida panthers. Conserv Genet 11:615–626
Hedrick P, Hedgecock D, Hamelberg S (1995) Estimation of effective population size in winter-run chinook salmon. Conserv Biol 9:615–624
Hedrick PW, Miller PS, Geffen E, Wayne R (1997) Genetic evaluation of the three captive Mexican wolf lineages. Zoo Biol 16:47–69
Hedrick P, Dowling TE, Minckley WL, Tibbets CA, Demarais BD, Marsh PC (2000a) Establishing a captive broodstock for the endangered bonytail chub (Gila elegans). J Hered 91:35–39
Hedrick PW, Hedgecock D, Hamelberg S, Croci SJ (2000b) The impact of supplementation in winter-run chinook salmon on effective population size. J Hered 91:112–116
Ireland SC, Beamesderfer RCP, Paragamian VL, Wakkinen VD, Siple JT (2002) Success of hatchery-reared juvenile white sturgeon (Acipenser transmontanus) following release in the Kootenai River, Idaho, USA. J Appl Ichthyol 18:642–650
Johnson JE, Jensen BL (1991) Hatcheries for endangered freshwater fish. In: Minckley WL, Deacon JE (eds) Battle against extinction. University of Arizona Press, Tuscon
Jones OR, Wang J (2010) Molecular marker-based pedigrees for animal conservation biologists. Anim Conserv 13:26–34
Kalinowski ST, Taper ML, Marshall TC (2007) Revising how the computer program CERVUS accommodates genotyping error increases success in paternity assignment. Mol Ecol 16:1099–1106
Kalinowski ST, Van Doornik DM, Kozfkay CC, Waples RS (2012) Genetic diversity in the Snake River sockeye salmon captive broodstock program as estimated from broodstock records. Cons Genet (in press)
Keenlyne KD, Jenkins LG (1993) Age at sexual maturity of the pallid sturgeon. Trans Am Fish Soc 122:393–396
Lande R, Barrowclough GF (1987) Effective population size, genetic variation, and their use in population management In: Soule ME (ed) Viable populations for conservation. Cambridge University Press, Cambridge
Lippe C, Dumont P, Bernatchez L (2006) High genetic diversity and no inbreeding in the endangered copper redhorse, Moxostoma hubbsi (Catostomidae, Pisces): the positive sides of a long generation time. Mol Ecol 15:1769–1780
Lynch M, O’Hely (2001) Captive breeding and the genetic fitness of natural populations. Conserv Genet 2:363–378
McQuown EC, Sloss BL, Sheehan RJ, Rodzen J, Tranah GJ, May B (2000) Microsatellite analysis of genetic variation in sturgeon: new primer sequences for Scaphirhynchus and Acipenser. Trans Am Fish Soc 129:1380–1388
Murphy CE, Hoover JJ, George SG, Killgore KJ (2007) Morphometric variation among river sturgeons (Scaphirhynchus spp.) of the Middle and Lower Mississippi River. J Appl Ichthyol 23:313–323
Quattro JM, Greig TW, Coykendall DK, Bowen BW, Baldwin JD (2002) Genetic issues in aquatic species management: the shortnose sturgeon (Acipenser brevirostrum) in the southeastern United States. Conserv Genet 3:155–166
Ralls K, Ballou JD (2004) Genetic status and management of California Condors. Condor 106:215–228
Rousset F (2008) Genepop ‘007: a complete re-implementation of the GENEPOP software for Windows and Linux. Mol Ecol Res 8:103–106
Ryman N, Laikre L (1991) Effects of supportive breeding on the genetically effective population size. Conserv Biol 5:325–329
Schrey AW, Heist EJ (2007) Stock structure of pallid sturgeon analyzed with microsatellite loci. J Appl Ichthyol 23:297–303
Seal US, Thorne ET, Anderson SH, Bogan MA (eds) (1989) Conservation biology of the black-footed ferret. Yale Univ. Press, New Haven
Shuman DA, Klumb RA, Wilson RH, Jaeger ME, Haddix T, Gardner WM, Doyle WJ, Horner PT, Ruggles M, Steffensen KD, Stukel S, Wanner GA (2011) Pallid sturgeon size structure, condition, and growth in the Missouri River Basin. J Appl Ichthyol 27:269–281
Steffensen KD, Powell LA, Koch JD (2010) Assessment of Hatchery-Reared pallid sturgeon survival in the Lower Missouri River. N Am J Fish Manage 30:671–678
Steffensen KD, Powell LA, Pegg MA (2012) Population size of hatchery-reared and wild pallid sturgeon in the lower Missouri River. N Am J Fish Manage 32:159–166
Turner TF, Dowling TE, Marsh PC, Kesner BR, Kelsen AT (2007) Effective size, census size, and genetic monitoring of the endangered razorback sucker, Xyrauchen texanus. Conserv Genet 8:417–425
USFWS (2007) Pallid sturgeon (Scaphirhynchus albus) five year review: summary and evaluation. Billings, MT
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538
Wang JL, Ryman N (2001) Genetic effects of multiple generations of supportive breeding. Conserv Biol 15:1619–1631
Waples R (2002) Evaluating the effect of stage-specific survivorship on the Ne/N ratio. Mol Ecol 11:1029–1037
Webb MAH, Williams JE, Hildebrand LR (2005) Recovery program review for endangered pallid sturgeon in the Upper Missouri River Basin. Rev Fish Sci 13:165–176
Westemeier RL, Brawn JD, Simpson SA, Esker TL, Jansen RW, Walk JW, Kershner EL, Bouzat JL, Paige KN (1998) Tracking the long-term decline and recovery of an isolated population. Science 282:1695–1698
Acknowledgments
We would like to thank Jeff Powell, Kurt Schilling, and the staff at Gavin’s Point National Fish Hatchery for their assistance and Southern Illinois University Fisheries and Illinois Aquaculture center for providing equipment and assistance in the field. We would also like to thank our undergraduate assistant Josh Geltz, and the graduate students Emily Croteau, Jen Eichelberger, and Ryan Boley from the Southern Illinois University Carbondale. Rob Hopkins helped with the figures and Amy Anderson helped calculate inbreeding coefficients. This work was supported by the US Department of Energy Western Area Power Administration Upper Great Plains Customer Service Region Contract No. 08-UGPR-56, and Ullman Distinguished Professorship (PWH).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saltzgiver, M.J., Heist, E.J. & Hedrick, P.W. Genetic evaluation of the initiation of a captive population: the general approach and a case study in the endangered pallid sturgeon (Scaphirhynchus albus). Conserv Genet 13, 1381–1391 (2012). https://doi.org/10.1007/s10592-012-0381-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-012-0381-7